Physics:Isotopes of beryllium
| ||||||||||||||||||||||||||||||
| Standard atomic weight Ar, standard(Be) |
| |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Beryllium (4Be) has 11 known isotopes and 3 known isomers, but only one of these isotopes (The element Chemistry:Beryllium does not exist.) is stable and a primordial nuclide. As such, beryllium is considered a monoisotopic element. It is also a mononuclidic element, because its other isotopes have such short half-lives that none are primordial and their abundance is very low. Beryllium is unique as being the only monoisotopic element with an even number of protons (even atomic number) and also has an odd number of neutrons;[2] the 25 other monoisotopic elements all have odd numbers of protons (odd atomic number), and even of neutrons, so the total mass number is still odd.
Of the 10 radioisotopes of beryllium, the most stable are The element Chemistry:Beryllium does not exist. with a half-life of 1.387 million years and The element Chemistry:Beryllium does not exist. with a half-life of 53.22 days. All other radioisotopes have half-lives shorter than 15 seconds.
The 1:1 neutron–proton ratio seen in stable isotopes of many light elements (up to oxygen, and in elements with even atomic number up to calcium) is prevented in beryllium by the extreme instability of The element Chemistry:Beryllium does not exist. toward splitting into two The element Chemistry:Helium does not exist. nuclei, which may be seen either alpha decay or a type of fission; in any case the half-life is only 8.2×10−17 s, short enough to normally be considered unbound. This, as with the relative instability of all lithium, beryllium, and boron isotopes, is favored due to the extremely tight binding of the helium-4 nucleus.
Beryllium is prevented from having a stable isotope with 4 protons and 6 neutrons by the very lopsided neutron–proton ratio for such a light element. Nevertheless, this isotope, beryllium-10, has a half-life above a million years and a decay energy less than 1 MeV, which indicates unusual stability given that condition.
Most beryllium present in the universe is thought to be formed by cosmic ray nucleosynthesis from cosmic ray spallation in the period between the Big Bang and the formation of the Solar System. The isotopes The element Chemistry:Beryllium does not exist. and The element Chemistry:Beryllium does not exist. are both cosmogenic nuclides because they are made, in the Solar System, continually at the rate they decay by spallation,[3] as is carbon-14.
List of isotopes
Template:Isotope table discovery
|-id=Beryllium-6
| The element Chemistry:Beryllium does not exist.[n 1]
| 4
| 2
| 6.019726(6)
| style="text-align:center" | 1958
| 5.0(3) zs
[91.6(5.6) keV]
| 2p
| The element Chemistry:Helium does not exist.
| 0+
|
|-
| The element Chemistry:Beryllium does not exist.[n 2]
| 4
| 3
| 7.01692871(8)
| style="text-align:center" | 1938
| 53.22(6) d
| ε
| The element Chemistry:Lithium does not exist.
| 3/2−
| Trace[n 3]
|-
| [[Beryllium-8|The element Chemistry:Beryllium does not exist.]][n 4]
| 4
| 4
| 8.00530510(4)
| style="text-align:center" | 1932
| 81.9(3.7) as
[5.58(25) eV]
| α[n 5]
| The element Chemistry:Helium does not exist.
| 0+
|
|-id=Beryllium-9
| The element Chemistry:Beryllium does not exist.[n 6]
| 4
| 5
| 9.01218306(8)
| style="text-align:center" | 1921
| colspan=3 align=center|Stable
| 3/2−
| 1
|-id=Beryllium-10
| The element Chemistry:Beryllium does not exist.
| 4
| 6
| 10.01353469(9)
| style="text-align:center" | 1935
| 1.387(12)×106 y[nb 1]
| β−
| The element Chemistry:Boron does not exist.
| 0+
| Trace[n 3]
|-id=Beryllium-11
| rowspan=3|The element Chemistry:Beryllium does not exist.[n 7]
| rowspan=3|4
| rowspan=3|7
| rowspan=3|11.02166108(26)
| rowspan=3 style="text-align:center" | 1958
| rowspan=3|13.76(7) s
| β− (96.7(1)%)
| The element Chemistry:Boron does not exist.
| rowspan=3|1/2+
| rowspan=3|
|-
| β−α (3.3(1)%)
| The element Chemistry:Lithium does not exist.
|-
| β−p (0.0013(3)%)
| The element Chemistry:Beryllium does not exist.
|-id=Beryllium-12
| rowspan=2|The element Chemistry:Beryllium does not exist.
| rowspan=2|4
| rowspan=2|8
| rowspan=2|12.0269221(20)
| rowspan=2 style="text-align:center" | 1966
| rowspan=2|21.46(5) ms | β− (99.50(3)%) | The element Chemistry:Boron does not exist. | rowspan=2|0+ | rowspan=2| |- | β−n (0.50(3)%) | The element Chemistry:Boron does not exist. |-id=Beryllium-12m | style="text-indent:1em" | The element Chemistry:Beryllium does not exist. | colspan="3" style="text-indent:2em" | 2251(1) keV | style="text-align:center" | 2003 | 233(7) ns | IT | The element Chemistry:Beryllium does not exist. | 0+ | |-id=Beryllium-13 | The element Chemistry:Beryllium does not exist. | 4 | 9 | 13.036135(11) | style="text-align:center" | 1983 | 1.0(7) zs | n ? | The element Chemistry:Beryllium does not exist. | (1/2−) | |-id=Beryllium-14 | rowspan=5|The element Chemistry:Beryllium does not exist.[n 8] | rowspan=5|4 | rowspan=5|10 | rowspan=5|14.04289(14) | rowspan=5 style="text-align:center" | 1973
| rowspan=5|4.53(27) ms
| β−n (86(6)%)
| The element Chemistry:Boron does not exist.
| rowspan=5|0+
| rowspan=5|
|-
| β− (> 9.0(6.3)%)
| The element Chemistry:Boron does not exist.
|-
| β−2n (5(2)%)
| The element Chemistry:Boron does not exist.
|-
| β−t (0.02(1)%)
| The element Chemistry:Beryllium does not exist.
|-
| β−α (< 0.004%)
| The element Chemistry:Lithium does not exist.
|-id=Beryllium-15
| The element Chemistry:Beryllium does not exist.
| 4
| 11
| 15.05349(18)
| style="text-align:center" | 2013
| 790(270) ys
| n
| The element Chemistry:Beryllium does not exist.
| (5/2+)
|
|-id=Beryllium-16
| The element Chemistry:Beryllium does not exist.
| 4
| 12
| 16.06167(18)
| style="text-align:center" | 2012
| 650(130) ys
[0.73(18) MeV]
| 2n
| The element Chemistry:Beryllium does not exist.
| 0+
|
|}
- ↑ Intermediate in the proton–proton chain (final step of PP I).
- ↑ Produced in Big Bang nucleosynthesis, but not primordial, as it all quickly decayed to 7Li
- ↑ 3.0 3.1 cosmogenic nuclide
- ↑ Intermediate product of triple alpha process in stellar nucleosynthesis
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedBe8 - ↑ Borromean nucleus
- ↑ Has 1 halo neutron
- ↑ Has 4 halo neutrons
Beryllium-7
Beryllium-7 is an isotope with a half-life of 53.22 days that is generated naturally as a cosmogenic nuclide.[3] It is also a nuisance byproduct in nuclear reactors and accelerators. The rate at which the short-lived The element Chemistry:Beryllium does not exist. is transferred from the air to the ground is controlled in part by the weather. The element Chemistry:Beryllium does not exist. decay in the Sun (the half-life in stars can be greatly different from the normal as it is a free rather than a bound electron that is captured) is one of the sources of solar neutrinos, and the first type ever detected using the Homestake experiment. Presence of The element Chemistry:Beryllium does not exist. in sediments is often used to establish that they are fresh, i.e. less than about 3–4 months in age, or about two half-lives of The element Chemistry:Beryllium does not exist..[4]

Beryllium-8
Beryllium-8 decays immediately into two alpha particles as its total energy is about 92 keV greater than that of the two alpha particles, and the Coulomb barrier to decay is negligible. This is unusual among light N = Z nuclides and creates a bottleneck in stellar nucleosynthesis, which requires that a third alpha be immediately captured, known as the fusion of three alpha particles, to form stable carbon-12 and thence all heavier elements.
Beryllium-10
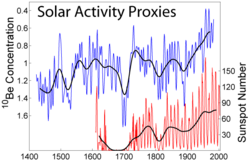
Beryllium-10 (10Be) has a half-life of 1.387 million years and beta decays to stable Boron-10 with a maximum energy of 556.0 keV:
- 10Be → 10B + e−.
Beryllium-10 is formed in the Earth's atmosphere mainly by cosmic ray spallation of nitrogen and oxygen.[5][6][3]
Because beryllium tends to exist in solutions below about pH 5.5 (and rainwater above many industrialized areas can have a pH less than 5), it will dissolve and be transported to the Earth's surface via rainwater. As the precipitation quickly becomes more alkaline, beryllium drops out of solution. Cosmogenic 10Be thereby accumulates at the soil surface, where its relatively long half-life does not limit its residence time there.
10Be and its daughter product have been used in surface exposure dating to examine soil erosion, soil formation from regolith, the development of lateritic soils and the age of ice cores.[7] It is also formed in nuclear explosions by a reaction of fast neutrons with 13C in the carbon dioxide in air, and is one of the historical indicators of past activity at nuclear test sites. 10Be decay is a significant isotope used as a proxy data measure for cosmogenic nuclides to characterize solar and extra-solar attributes of the past from terrestrial samples.[8]
The rate of production of beryllium-10 depends on the activity of the sun. When solar activity is low (low numbers of sunspots and low solar wind), the barrier against cosmic rays that exists beyond the termination shock is weakened (see Cosmic ray). This means more beryllium-10 is produced, and it can be detected millennia later. Beryllium-10 can thus serve as a marker of Miyake events, such as the 774–775 carbon-14 spike. There can be an effect on climate[9] (see Homeric Minimum). While other cosmogenic isotopes experience similar cycles, the high rate of production, long half-life, and relative immobility in the environment make this most suitable for this purpose.
Decay chains
Isotopes of beryllium heavier than the stable 9Be decay via beta decay or a combination of beta decay and neutron emission. However, The element Chemistry:Beryllium does not exist. splits in two to result in The element Chemistry:Helium does not exist.. Then, The element Chemistry:Beryllium does not exist. decays only via electron capture, an exceptional occurrence in such a light element. For this reason, its half-life can be artificially lowered by 0.83% via endohedral enclosure (7Be@C60).[10] Finally even lighter isotopes decay exclusively by emitting protons and are also (like 8Be) unbound. The decay of all known beryllium isotopes is summarized as follows:
See also
Daughter products other than beryllium
Notes
- ↑ Note that NUBASE2020 uses the tropical year to convert between years and other units of time, not the Gregorian year. The relationship between years and other time units in NUBASE2020 is as follows: 1 y = 365.2422 d = 31 556 926 s
References
- ↑ Meija, Juris; Coplen, Tyler B.; Berglund, Michael; Brand, Willi A.; De Bièvre, Paul; Gröning, Manfred; Holden, Norman E.; Irrgeher, Johanna et al. (2016). "Atomic weights of the elements 2013 (IUPAC Technical Report)". Pure and Applied Chemistry 88 (3): 265–91. doi:10.1515/pac-2015-0305.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedCIAAWberyllium - ↑ 3.0 3.1 3.2 S.V. Poluianov; G.A. Kovaltsov; A.L. Mishev; I.G. Usoskin (2016). "Production of cosmogenic isotopes 7Be, 10Be, 14C, 22Na, and 36Cl in the atmosphere: Altitudinal profiles of yield functions". J. Geophys. Res. Atmos. 121 (13): 8125–8136. doi:10.1002/2016JD025034. Bibcode: 2016JGRD..121.8125P.
- ↑ 4.0 4.1 Yamamoto, Masayoshi; Sakaguchi, Aya; Sasaki, Keiichi; Hirose, Katsumi; Igarashi, Yasuhito; Kim, Chang Kyu (January 2006). "Seasonal and spatial variation of atmospheric 210Pb and 7Be deposition: features of the Japan Sea side of Japan". Journal of Environmental Radioactivity 86 (1): 110–131. doi:10.1016/j.jenvrad.2005.08.001. PMID 16181712.
- ↑ G.A. Kovaltsov; I.G. Usoskin (2010). "A new 3D numerical model of cosmogenic nuclide 10Be production in the atmosphere". Earth Planet. Sci. Lett. 291 (1–4): 182–199. doi:10.1016/j.epsl.2010.01.011. Bibcode: 2010E&PSL.291..182K.
- ↑ J. Beer; K. McCracken; R. von Steiger (2012). Cosmogenic radionuclides: theory and applications in the terrestrial and space environments. Physics of Earth and Space Environments. 26. Physics of Earth and Space Environments, Springer, Berlin. doi:10.1007/978-3-642-14651-0. ISBN 978-3-642-14650-3.
- ↑ Balco, Greg; Shuster, David L. (2009). "26Al-10Be–21Ne burial dating". Earth and Planetary Science Letters 286 (3–4): 570–575. doi:10.1016/j.epsl.2009.07.025. Bibcode: 2009E&PSL.286..570B. http://www.bgc.org/shuster/BalcoShuster(2009b)_Al_Be_Ne_burial_dating.pdf. Retrieved 2012-12-10.
- ↑ Paleari, Chiara I.; F. Mekhaldi; F. Adolphi; M. Christl; C. Vockenhuber; P. Gautschi; J. Beer; N. Brehm et al. (2022). "Cosmogenic radionuclides reveal an extreme solar particle storm near a solar minimum 9125 years BP". Nat. Commun. 13 (214): 214. doi:10.1038/s41467-021-27891-4. PMID 35017519. Bibcode: 2022NatCo..13..214P.
- ↑ Philip Ball (Dec 19, 2001). "Flickering sun switched climate". Nature. doi:10.1038/news011220-9.
- ↑ Ohtsuki, T.; Yuki, H.; Muto, M.; Kasagi, J.; Ohno, K. (9 September 2004). "Enhanced Electron-Capture Decay Rate of 7Be Encapsulated in C60 Cages". Physical Review Letters 93 (11). doi:10.1103/PhysRevLett.93.112501. PMID 15447332. Bibcode: 2004PhRvL..93k2501O. https://journals.aps.org/prl/abstract/10.1103/PhysRevLett.93.112501. Retrieved 23 February 2022.
Lua error: Internal error: The interpreter has terminated with signal "24".
Lua error: Internal error: The interpreter has terminated with signal "24".