Biology:Polyomaviridae
| Polyomaviridae | |
|---|---|
| |
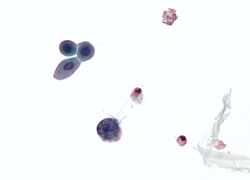
| Micrograph showing a polyomavirus infected cell—large (blue) cell below-center-left. Urine cytology specimen. | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Monodnaviria |
| Kingdom: | Shotokuvirae |
| Phylum: | Cossaviricota |
| Class: | Papovaviricetes |
| Order: | Sepolyvirales |
| Family: | Polyomaviridae |
| Genera | |
| |
Polyomaviridae is a family of viruses whose natural hosts are primarily mammals and birds.[1][2] As of 2020, there are six recognized genera and 117 species, five of which are unassigned to a genus.[3] 14 species are known to infect humans, while others, such as Simian Virus 40, have been identified in humans to a lesser extent.[4][5] Most of these viruses are very common and typically asymptomatic in most human populations studied.[6][7] BK virus is associated with nephropathy in renal transplant and non-renal solid organ transplant patients,[8][9] JC virus with progressive multifocal leukoencephalopathy,[10] and Merkel cell virus with Merkel cell cancer.[11]
Structure and genome
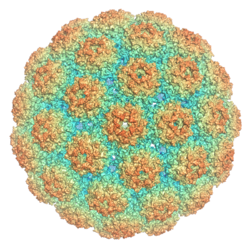
Polyomaviruses are non-enveloped double-stranded DNA viruses with circular genomes of around 5000 base pairs. The genome is packaged in a viral capsid of about 40-50 nanometers in diameter, which is icosahedral in shape (T=7 symmetry).[2][12] The capsid is composed of 72 pentameric capsomeres of a protein called VP1, which is capable of self-assembly into a closed icosahedron;[13] each pentamer of VP1 is associated with one molecule of one of the other two capsid proteins, VP2 or VP3.[5]

The genome of a typical polyomavirus codes for between 5 and 9 proteins, divided into two transcriptional regions called the early and late regions due to the time during infection in which they are transcribed. Each region is transcribed by the host cell's RNA polymerase II as a single pre-messenger RNA containing multiple genes. The early region usually codes for two proteins, the small and large tumor antigens, produced by alternative splicing. The late region contains the three capsid structural proteins VP1, VP2, and VP3, produced by alternative translational start sites. Additional genes and other variations on this theme are present in some viruses: for example, rodent polyomaviruses have a third protein called middle tumor antigen in the early region, which is extremely efficient at inducing cellular transformation; SV40 has an additional capsid protein VP4; some examples have an additional regulatory protein called agnoprotein expressed from the late region. The genome also contains a non-coding control or regulatory region containing the early and late regions' promoters, transcriptional start sites, and the origin of replication.[2][12][5][15]
Replication and life cycle

The polyomavirus life cycle begins with entry into a host cell. Cellular receptors for polyomaviruses are sialic acid residues of glycans, commonly gangliosides. The attachment of polyomaviruses to host cells is mediated by the binding of VP1 to sialylated glycans on the cell surface.[2][12][15][16] In some particular viruses, additional cell-surface interactions occur; for example, the JC virus is believed to require interaction with the 5HT2A receptor and the Merkel cell virus with heparan sulfate.[15][17] However, in general virus-cell interactions are mediated by commonly occurring molecules on the cell surface, and therefore are likely not a major contributor to individual viruses' observed cell-type tropism.[15] After binding to molecules on the cell surface, the virion is endocytosed and enters the endoplasmic reticulum - a behavior unique among known non-enveloped viruses[18] - where the viral capsid structure is likely to be disrupted by action of host cell disulfide isomerase enzymes.[2][12][19]
The details of transit to the nucleus are not clear and may vary among individual polyomaviruses. It has been frequently reported that an intact, albeit distorted, virion particle is released from the endoplasmic reticulum into the cell cytoplasm, where the genome is released from the capsid, possibly due to the low calcium concentration in the cytoplasm.[18] Both expression of viral genes and replication of the viral genome occur in the nucleus using host cell machinery. The early genes - comprising at minimum the small tumor antigen (ST) and large tumor antigen (LT) - are expressed first, from a single alternatively spliced messenger RNA strand. These proteins serve to manipulate the host's cell cycle - dysregulating the transition from G1 phase to S phase, when the host cell's genome is replicated - because host cell DNA replication machinery is needed for viral genome replication.[2][12][15] The precise mechanism of this dysregulation depends on the virus; for example, SV40 LT can directly bind host cell p53, but murine polyomavirus LT does not.[20] LT induces DNA replication from the viral genome's non-coding control region (NCCR), after which expression of the early mRNA is reduced and expression of the late mRNA, which encodes the viral capsid proteins, begins.[19] As these interactions begin, the LTs belonging to several polyomaviruses, including Merkel cell polyomavirus, present oncogenic potential.[21] Several mechanisms have been described for regulating the transition from early to late gene expression, including the involvement of the LT protein in repressing the early promoter,[19] the expression of un-terminated late mRNAs with extensions complementary to early mRNA,[15] and the expression of regulatory microRNA.[15] Expression of the late genes results in accumulation of the viral capsid proteins in the host cell cytoplasm. Capsid components enter the nucleus in order to encapsidate new viral genomic DNA. New virions may be assembled in viral factories.[2][12] The mechanism of viral release from the host cell varies among polyomaviruses; some express proteins that facilitate cell exit, such as the agnoprotein or VP4.[19] In some cases high levels of encapsidated virus result in cell lysis, releasing the virions.[15]
Viral proteins
Tumor antigens
The large tumor antigen plays a key role in regulating the viral life cycle by binding to the viral origin of DNA replication where it promotes DNA synthesis. Also as the polyomavirus relies on the host cell machinery to replicate the host cell needs to be in s-phase for this to begin. Due to this, large T-antigen also modulates cellular signaling pathways to stimulate progression of the cell cycle by binding to a number of cellular control proteins.[22] This is achieved by a two prong attack of inhibiting tumor suppressing genes p53 and members of the retinoblastoma (pRB) family,[23] and stimulating cell growth pathways by binding cellular DNA, ATPase-helicase, DNA polymerase α association, and binding of transcription preinitiation complex factors.[24] This abnormal stimulation of the cell cycle is a powerful force for oncogenic transformation.[citation needed]
The small tumor antigen protein is also able to activate several cellular pathways that stimulate cell proliferation. Polyomavirus small T antigens commonly target protein phosphatase 2A (PP2A),[25] a key multisubunit regulator of multiple pathways including Akt, the mitogen-activated protein kinase (MAPK) pathway, and the stress-activated protein kinase (SAPK) pathway.[26][27] Merkel cell polyomavirus small T antigen encodes a unique domain, called the LT-stabilization domain (LSD), that binds to and inhibits the FBXW7 E3 ligase regulating both cellular and viral oncoproteins.[28] Unlike for SV40, the MCV small T antigen directly transforms rodent cells in vitro.[29]
The middle tumor antigen is used in model organisms developed to study cancer, such as the MMTV-PyMT system where middle T is coupled to the MMTV promoter. There it functions as an oncogene, while the tissue where the tumor develops is determined by the MMTV promoter.[citation needed]
Capsid proteins
The polyomavirus capsid consists of one major component, major capsid protein VP1, and one or two minor components, minor capsid proteins VP2 and VP3. VP1 pentamers form the closed icosahedral viral capsid, and in the interior of the capsid each pentamer is associated with one molecule of either VP2 or VP3.[5][30] Some polyomaviruses, such as Merkel cell polyomavirus, do not encode or express VP3.[31] The capsid proteins are expressed from the late region of the genome.[5]
Agnoprotein
The agnoprotein is a small multifunctional phospho-protein found in the late coding part of the genome of some polyomaviruses, most notably BK virus, JC virus, and SV40. It is essential for proliferation in the viruses that express it and is thought to be involved in regulating the viral life cycle, particularly replication and viral exit from the host cell, but the exact mechanisms are unclear.[32][33]
Taxonomy
The polyomaviruses are members of group I (dsDNA viruses). The classification of polyomaviruses has been the subject of several proposed revisions as new members of the group are discovered. Formerly, polyomaviruses and papillomaviruses, which share many structural features but have very different genomic organizations, were classified together in the now-obsolete family Papovaviridae.[34] (The name Papovaviridae derived from three abbreviations: Pa for Papillomavirus, Po for Polyomavirus, and Va for "vacuolating.")[35] The polyomaviruses were divided into three major clades (that is, genetically-related groups): the SV40 clade, the avian clade, and the murine polyomavirus clade.[36] A subsequent proposed reclassification by the International Committee on Taxonomy of Viruses (ICTV) recommended dividing the family of Polyomaviridae into three genera:[37]
- Genus Orthopolyomavirus (type species SV40)
- Genus Wukipolyomavirus (type species KI polyomavirus)
- Genus Avipolyomavirus (type species Avian polyomavirus)
The current ICTV classification system recognises six genera and 117 species, of which five could not be assigned a genus. This system retains the distinction between avian and mammalian viruses, grouping the avian subset into the genus Gammapolyomavirus. The six genera are:[3]
- Alphapolyomavirus
- Betapolyomavirus
- Deltapolyomavirus
- Epsilonpolyomavirus
- Gammapolyomavirus
- Zetapolyomavirus
The following species are unassigned to a genus:[3]
- Centropristis striata polyomavirus 1
- Rhynchobatus djiddensis polyomavirus 1
- Sparus aurata polyomavirus 1
- Trematomus bernacchii polyomavirus 1
- Trematomus pennellii polyomavirus 1
Description of additional viruses is ongoing. These include the sea otter polyomavirus 1[38] and Alpaca polyomavirus[39] Another virus is the giant panda polyomavirus 1.[40] Another virus has been described from sigmodontine rodents.[41] Another - tree shrew polyomavirus 1 - has been described in the tree shrew.[42]
Human polyomaviruses
Most polyomaviruses do not infect humans. Of the polyomaviruses cataloged as of 2017, a total of 14 were known with human hosts.[4] However, some polyomaviruses are associated with human disease, particularly in immunocompromised individuals. MCV is highly divergent from the other human polyomaviruses and is most closely related to murine polyomavirus. Trichodysplasia spinulosa-associated polyomavirus (TSV) is distantly related to MCV. Two viruses—HPyV6 and HPyV7—are most closely related to KI and WU viruses, while HPyV9 is most closely related to the African green monkey-derived lymphotropic polyomavirus (LPV).[citation needed]
A fourteenth virus has been described.[43] Lyon IARC polyomavirus is related to raccoon polyomavirus.[citation needed]
List of human polyomaviruses
The following 14 polyomaviruses with human hosts had been identified and had their genomes sequenced as of 2017:[4]
| Species | Proposed genus | Virus name | Abbreviation | NCBI RefSeq | Year of discovery | Clinical correlate (if any) | References |
|---|---|---|---|---|---|---|---|
| 5 Human polyomavirus 5 | Alpha | Merkel cell polyomavirus | MCPyV | NC_010277 | 2008 | Merkel cell cancer[5] | [44][11][45] |
| 8 Human polyomavirus 8 | Alpha | Trichodysplasia spinulosa polyomavirus | TSPyV | NC_014361 | 2010 | Trichodysplasia spinulosa[5] | [46][47] |
| 9 Human polyomavirus 9 | Alpha | Human polyomavirus 9 | HPyV9 | NC_015150 | 2011 | None known | [48] |
| 12 Human polyomavirus 12 | Alpha | Human polyomavirus 12 | HPyV12 | NC_020890 | 2013 | None known | [49] |
| 13 Human polyomavirus 13 | Alpha | New Jersey polyomavirus | NJPyV | NC_024118 | 2014 | None known | [50] |
| 1 Human polyomavirus 1 | Beta | BK polyomavirus | BKPyV | NC_001538 | 1971 | Polyomavirus-associated nephropathy; haemorrhagic cystitis[5] | [51] |
| 2 Human polyomavirus 2 | Beta | JC polyomavirus | JCPyV | NC_001699 | 1971 | Progressive multifocal leukoencephalopathy[5] | [52] |
| 3 Human polyomavirus 3 | Beta | KI polyomavirus | KIPyV | NC_009238 | 2007 | None known | [53] |
| 4 Human polyomavirus 4 | Beta | WU polyomavirus | WUPyV | NC_009539 | 2007 | None known | [14] |
| 6 Human polyomavirus 6 | Delta | Human polyomavirus 6 | HPyV6 | NC_014406 | 2010 | HPyV6 associated pruritic and dyskeratotic dermatosis (H6PD)[54] | [31] |
| 7 Human polyomavirus 7 | Delta | Human polyomavirus 7 | HPyV7 | NC_014407 | 2010 | HPyV7-related epithelial hyperplasia[54][55][56] | [31] |
| 11 Human polyomavirus 11 | Delta | STL polyomavirus | STLPyV | NC_020106 | 2013 | None known | [57] |
| Human polyomavirus 14 | Alpha | Lyon IARC polyomavirus | LIPyV | NC_034253.1 | 2017 | None known | [58][59] |
Deltapolyomavirus contains only the four human viruses shown in the above table. The Alpha and Beta groups contain viruses that infect a variety of mammals. The Gamma group contains the avian viruses.[4] Clinically significant disease associations are shown only where causality is expected.[5][60]
Antibodies to the monkey lymphotropic polyomavirus have been detected in humans suggesting that this virus - or a closely related virus - can infect humans.[61]
Clinical relevance
All the polyomaviruses are highly common childhood and young adult infections.[62] Most of these infections appear to cause little or no symptoms. These viruses are probably lifelong persistent among almost all adults. Diseases caused by human polyomavirus infections are most common among immunocompromised people; disease associations include BK virus with nephropathy in renal transplant and non-renal solid organ transplant patients,[8][9] JC virus with progressive multifocal leukoencephalopathy,[10] and Merkel cell virus (MCV) with Merkel cell cancer.[11]
SV40
SV40 replicates in the kidneys of monkeys without causing disease, but can cause cancer in rodents under laboratory conditions. In the 1950s and early 1960s, well over 100 million people may have been exposed to SV40 due to previously undetected SV40 contamination of polio vaccine, prompting concern about the possibility that the virus might cause disease in humans.[63][64] Although it has been reported as present in some human cancers, including brain tumors, bone tumors, mesotheliomas, and non-Hodgkin's lymphomas,[65] accurate detection is often confounded by high levels of cross-reactivity for SV40 with widespread human polyomaviruses.[64] Most virologists dismiss SV40 as a cause for human cancers.[63][66][67]
Diagnosis
The diagnosis of polyomavirus almost always occurs after the primary infection as it is either asymptomatic or sub-clinical. Antibody assays are commonly used to detect presence of antibodies against individual viruses.[68] Competition assays are frequently needed to distinguish among highly similar polyomaviruses.[69]
In cases of progressive multifocal leucoencephalopathy (PML), a cross-reactive antibody to SV40 T antigen (commonly Pab419) is used to stain tissues directly for the presence of JC virus T antigen. PCR can be used on a biopsy of the tissue or cerebrospinal fluid to amplify the polyomavirus DNA. This allows not only the detection of polyomavirus but also which sub type it is.[70]
There are three main diagnostic techniques used for the diagnosis of the reactivation of polyomavirus in polyomavirus nephropathy (PVN): urine cytology, quantification of the viral load in both urine and blood, and a renal biopsy.[68] The reactivation of polyomavirus in the kidneys and urinary tract causes the shedding of infected cells, virions, and/or viral proteins in the urine. This allows urine cytology to examine these cells, which if there is polyomavirus inclusion of the nucleus, is diagnostic of infection.[71] Also as the urine of an infected individual will contain virions and/or viral DNA, quantitation of the viral load can be done through PCR.[72] This is also true for the blood.
Renal biopsy can also be used if the two methods just described are inconclusive or if the specific viral load for the renal tissue is desired. Similarly to the urine cytology, the renal cells are examined under light microscopy for polyomavirus inclusion of the nucleus, as well as cell lysis and viral partials in the extra cellular fluid. The viral load as before is also measure by PCR.[citation needed]
Tissue staining using a monoclonal antibody against MCV T antigen shows utility in differentiating Merkel cell carcinoma from other small, round cell tumors.[73] Blood tests to detect MCV antibodies have been developed and show that infection with the virus is widespread although Merkel cell carcinoma patients have exceptionally higher antibody responses than asymptomatically infected persons.[7][74][75][76]
Use in tracing human migration
The JC virus offers a promising genetic marker for human evolution and migration.[77] It is carried by 70–90 percent of humans and is usually transmitted from parents to offspring. This method does not appear to be reliable for tracing the recent African origin of modern humans.[citation needed]
History
Murine polyomavirus was the first polyomavirus discovered, having been reported by Ludwik Gross in 1953 as an extract of mouse leukemias capable of inducing parotid gland tumors.[78] The causative agent was identified as a virus by Sarah Stewart and Bernice Eddy, after whom it was once called "SE polyoma".[79][80][81] The term "polyoma" refers to the viruses' ability to produce multiple (poly-) tumors (-oma) under certain conditions. The name has been criticized as a "meatless linguistic sandwich" ("meatless" because both morphemes in "polyoma" are affixes) giving little insight into the viruses' biology; in fact, subsequent research has found that most polyomaviruses rarely cause clinically significant disease in their host organisms under natural conditions.[82]
Dozens of polyomaviruses have been identified and sequenced as of 2017, infecting mainly birds and mammals. Two polyomaviruses are known to infect fish, the black sea bass[83] and gilthead seabream.[84] A total of fourteen polyomaviruses are known to infect humans.[4]
References
- ↑ "ICTV Virus Taxonomy Profile: Polyomaviridae". The Journal of General Virology 98 (6): 1159–1160. June 2017. doi:10.1099/jgv.0.000839. PMID 28640744.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "ICTV Report Polyomaviridae". http://www.ictv.global/report/polyomaviridae.
- ↑ 3.0 3.1 3.2 "Virus Taxonomy: 2020 Release". International Committee on Taxonomy of Viruses (ICTV). March 2021. https://ictv.global/taxonomy.
- ↑ 4.0 4.1 4.2 4.3 4.4 "A taxonomy update for the family Polyomaviridae". Archives of Virology 161 (6): 1739–50. June 2016. doi:10.1007/s00705-016-2794-y. PMID 26923930.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 5.8 5.9 "A cornucopia of human polyomaviruses". Nature Reviews. Microbiology 11 (4): 264–76. April 2013. doi:10.1038/nrmicro2992. PMID 23474680.
- ↑ "Seroepidemiology of Human Polyomaviruses in a US Population". American Journal of Epidemiology 183 (1): 61–9. January 2016. doi:10.1093/aje/kwv155. PMID 26667254.
- ↑ 7.0 7.1 "Seroepidemiology of human polyomaviruses". PLOS Pathogens 5 (3): e1000363. March 2009. doi:10.1371/journal.ppat.1000363. PMID 19325891.
- ↑ 8.0 8.1 "BK virus nephropathy in renal transplant recipients". Nephrology 21 (8): 647–54. August 2016. doi:10.1111/nep.12728. PMID 26780694.
- ↑ 9.0 9.1 "BK polyoma virus infection and renal disease in non-renal solid organ transplantation". Clinical Kidney Journal 9 (2): 310–8. April 2016. doi:10.1093/ckj/sfv143. PMID 26985385.
- ↑ 10.0 10.1 "Progressive Multifocal Leukoencephalopathy". F1000Research 4: 1424. 2015. doi:10.12688/f1000research.7071.1. PMID 26918152.
- ↑ 11.0 11.1 11.2 "Clonal integration of a polyomavirus in human Merkel cell carcinoma". Science 319 (5866): 1096–100. February 2008. doi:10.1126/science.1152586. PMID 18202256. Bibcode: 2008Sci...319.1096F.
- ↑ "Self-assembly of purified polyomavirus capsid protein VP1". Cell 46 (6): 895–904. September 1986. doi:10.1016/0092-8674(86)90071-1. PMID 3019556.
- ↑ 14.0 14.1 "Identification of a novel polyomavirus from patients with acute respiratory tract infections". PLOS Pathogens 3 (5): e64. May 2007. doi:10.1371/journal.ppat.0030064. PMID 17480120.
- ↑ 15.0 15.1 15.2 15.3 15.4 15.5 15.6 15.7 International Agency for Research on Cancer (2013). "Introduction to Polyomaviruses". IARC Monographs on the Evaluation of Carcinogenic Risks to Humans 104: 121–131. https://monographs.iarc.fr/ENG/Monographs/vol104/mono104-0GI.pdf.
- ↑ 16.0 16.1 "Structural and Functional Analysis of Murine Polyomavirus Capsid Proteins Establish the Determinants of Ligand Recognition and Pathogenicity". PLOS Pathogens 11 (10): e1005104. October 2015. doi:10.1371/journal.ppat.1005104. PMID 26474293.
- ↑ "Glycosaminoglycans and sialylated glycans sequentially facilitate Merkel cell polyomavirus infectious entry". PLOS Pathogens 7 (7): e1002161. July 2011. doi:10.1371/journal.ppat.1002161. PMID 21829355.
- ↑ 18.0 18.1 "How viruses use the endoplasmic reticulum for entry, replication, and assembly". Cold Spring Harbor Perspectives in Biology 5 (1): a013250. January 2013. doi:10.1101/cshperspect.a013250. PMID 23284050.
- ↑ 19.0 19.1 19.2 19.3 "Update on human polyomaviruses and cancer". Advances in Cancer Research 106: 1–51. 2010. doi:10.1016/S0065-230X(10)06001-X. ISBN 9780123747716. PMID 20399955.
- ↑ "Comparisons between Murine Polyomavirus and Simian Virus 40 Show Significant Differences in Small T Antigen Function". Journal of Virology 85 (20): 10649–10658. 2011. doi:10.1128/JVI.05034-11. PMID 21835797.
- ↑ "Merkel Cell Carcinomas Arising in Autoimmune Disease Affected Patients Treated with Biologic Drugs, Including Anti-TNF". Clinical Cancer Research 23 (14): 3929–3934. July 2017. doi:10.1158/1078-0432.CCR-16-2899. PMID 28174236. https://clincancerres.aacrjournals.org/content/23/14/3929.
- ↑ "Human polyomaviruses and brain tumors". Brain Research. Brain Research Reviews 50 (1): 69–85. December 2005. doi:10.1016/j.brainresrev.2005.04.007. PMID 15982744.
- ↑ "Polyomavirus-associated Trichodysplasia spinulosa involves hyperproliferation, pRB phosphorylation and upregulation of p16 and p21". PLOS ONE 9 (10): e108947. 2014. doi:10.1371/journal.pone.0108947. PMID 25291363. Bibcode: 2014PLoSO...9j8947K.
- ↑ "The T/t common exon of simian virus 40, JC, and BK polyomavirus T antigens can functionally replace the J-domain of the Escherichia coli DnaJ molecular chaperone". Proceedings of the National Academy of Sciences of the United States of America 94 (8): 3679–84. April 1997. doi:10.1073/pnas.94.8.3679. PMID 9108037. Bibcode: 1997PNAS...94.3679K.
- ↑ "Polyoma small and middle T antigens and SV40 small t antigen form stable complexes with protein phosphatase 2A". Cell 60 (1): 167–76. January 1990. doi:10.1016/0092-8674(90)90726-u. PMID 2153055.
- ↑ "The interaction of SV40 small tumor antigen with protein phosphatase 2A stimulates the map kinase pathway and induces cell proliferation". Cell 75 (5): 887–97. December 1993. doi:10.1016/0092-8674(93)90533-V. PMID 8252625.
- ↑ "Induction of cyclin D1 by simian virus 40 small tumor antigen". Proceedings of the National Academy of Sciences of the United States of America 93 (23): 12861–6. November 1996. doi:10.1073/pnas.93.23.12861. PMID 8917510. Bibcode: 1996PNAS...9312861W.
- ↑ "Merkel cell polyomavirus small T antigen controls viral replication and oncoprotein expression by targeting the cellular ubiquitin ligase SCFFbw7". Cell Host & Microbe 14 (2): 125–35. August 2013. doi:10.1016/j.chom.2013.06.008. PMID 23954152.
- ↑ "Human Merkel cell polyomavirus small T antigen is an oncoprotein targeting the 4E-BP1 translation regulator". The Journal of Clinical Investigation 121 (9): 3623–34. September 2011. doi:10.1172/JCI46323. PMID 21841310.
- ↑ "Interaction of polyomavirus internal protein VP2 with the major capsid protein VP1 and implications for participation of VP2 in viral entry" (in en). The EMBO Journal 17 (12): 3233–40. June 1998. doi:10.1093/emboj/17.12.3233. PMID 9628860.
- ↑ 31.0 31.1 31.2 "Merkel cell polyomavirus and two previously unknown polyomaviruses are chronically shed from human skin". Cell Host & Microbe 7 (6): 509–15. June 2010. doi:10.1016/j.chom.2010.05.006. PMID 20542254.
- ↑ "Infection by agnoprotein-negative mutants of polyomavirus JC and SV40 results in the release of virions that are mostly deficient in DNA content". Virology Journal 8: 255. May 2011. doi:10.1186/1743-422X-8-255. PMID 21609431.
- ↑ "Emerging From the Unknown: Structural and Functional Features of Agnoprotein of Polyomaviruses". Journal of Cellular Physiology 231 (10): 2115–27. October 2016. doi:10.1002/jcp.25329. PMID 26831433.
- ↑ "ICTV Taxonomy Website". https://ictv.global/taxonomy.
- ↑ International Agency for Research on Cancer (2013). "IARC Working Group on the Evaluation of Carcinogenic Risk to Humans. Malaria and Some Polyomaviruses (SV40, BK, JC, and Merkel Cell Viruses).". IARC Monographs on the Evaluation of Carcinogenic Risks to Humans 104. https://www.ncbi.nlm.nih.gov/books/NBK294248/.
- ↑ "Comparing phylogenetic codivergence between polyomaviruses and their hosts". Journal of Virology 80 (12): 5663–9. June 2006. doi:10.1128/JVI.00056-06. PMID 16731904.
- ↑ "Taxonomical developments in the family Polyomaviridae". Archives of Virology 156 (9): 1627–34. September 2011. doi:10.1007/s00705-011-1008-x. PMID 21562881.
- ↑ "Endemic infection of stranded southern sea otters (Enhydra lutris nereis) with novel parvovirus, polyomavirus, and adenovirus". Journal of Wildlife Diseases 53 (3): 532–542. July 2017. doi:10.7589/2016-04-082. PMID 28192039.
- ↑ "A novel pulmonary polyomavirus in alpacas (Vicugna pacos)". Veterinary Microbiology 201: 49–55. 2017. doi:10.1016/j.vetmic.2017.01.005. PMID 28284622.
- ↑ "A novel polyomavirus from the nasal cavity of a giant panda (Ailuropoda melanoleuca)". Virology Journal 14 (1): 207. 2017. doi:10.1186/s12985-017-0867-5. PMID 29078783.
- ↑ "A novel polyomavirus in sigmodontine rodents from São Paulo State, Brazil". Archives of Virology 163 (10): 2913–2915. 2018. doi:10.1007/s00705-018-3913-8. PMID 29931397.
- ↑ "Detection and genome characterization of two novel papillomaviruses and a novel polyomavirus in tree shrew (Tupaia belangeri chinensis) in China". Virol J 16 (1): 35. 2019. doi:10.1186/s12985-019-1141-9. PMID 30885224.
- ↑ "Isolation and characterization of a novel putative human polyomavirus". Virology 506: 45–54. 2017. doi:10.1016/j.virol.2017.03.007. PMID 28342387.
- ↑ Altman, Lawreence K. (2008-01-18). "Virus Is Linked to a Powerful Skin Cancer". The New York Times. https://www.nytimes.com/2008/01/18/health/research/18virus.html.
- ↑ "Human Merkel cell polyomavirus infection I. MCV T antigen expression in Merkel cell carcinoma, lymphoid tissues and lymphoid tumors". International Journal of Cancer 125 (6): 1243–9. September 2009. doi:10.1002/ijc.24510. PMID 19499546.
- ↑ "Discovery of a new human polyomavirus associated with trichodysplasia spinulosa in an immunocompromized patient". PLOS Pathogens 6 (7): e1001024. July 2010. doi:10.1371/journal.ppat.1001024. PMID 20686659.
- ↑ "The trichodysplasia spinulosa-associated polyomavirus: virological background and clinical implications". APMIS 121 (8): 770–82. August 2013. doi:10.1111/apm.12092. PMID 23593936.
- ↑ "A novel human polyomavirus closely related to the african green monkey-derived lymphotropic polyomavirus". Journal of Virology 85 (9): 4586–90. May 2011. doi:10.1128/jvi.02602-10. PMID 21307194.
- ↑ "Identification of a novel human polyomavirus in organs of the gastrointestinal tract". PLOS ONE 8 (3): e58021. 2013. doi:10.1371/journal.pone.0058021. PMID 23516426. Bibcode: 2013PLoSO...858021K.
- ↑ "Identification of a novel polyomavirus in a pancreatic transplant recipient with retinal blindness and vasculitic myopathy". The Journal of Infectious Diseases 210 (10): 1595–9. November 2014. doi:10.1093/infdis/jiu250. PMID 24795478.
- ↑ "New human papovavirus (B.K.) isolated from urine after renal transplantation". Lancet 1 (7712): 1253–7. June 1971. doi:10.1016/s0140-6736(71)91776-4. PMID 4104714.
- ↑ "Cultivation of papova-like virus from human brain with progressive multifocal leucoencephalopathy". Lancet 1 (7712): 1257–60. June 1971. doi:10.1016/S0140-6736(71)91777-6. PMID 4104715.
- ↑ "Identification of a third human polyomavirus". Journal of Virology 81 (8): 4130–6. April 2007. doi:10.1128/JVI.00028-07. PMID 17287263.
- ↑ 54.0 54.1 "Human polyomavirus 6 and 7 are associated with pruritic and dyskeratotic dermatoses". Journal of the American Academy of Dermatology 76 (5): 932–940.e3. May 2017. doi:10.1016/j.jaad.2016.11.035. PMID 28040372. PMC 5392424. https://zenodo.org/record/995723.
- ↑ "Human polyomavirus 7-associated pruritic rash and viremia in transplant recipients". The Journal of Infectious Diseases 211 (10): 1560–5. May 2015. doi:10.1093/infdis/jiu524. PMID 25231015.
- ↑ "Survey for human polyomaviruses in cancer". JCI Insight 1 (2). February 2016. doi:10.1172/jci.insight.85562. PMID 27034991.
- ↑ "Discovery of STL polyomavirus, a polyomavirus of ancestral recombinant origin that encodes a unique T antigen by alternative splicing". Virology 436 (2): 295–303. February 2013. doi:10.1016/j.virol.2012.12.005. PMID 23276405.
- ↑ Gheit, Tarik; Dutta, Sankhadeep; Oliver, Javier; Robitaille, Alexis; Hampras, Shalaka; Combes, Jean-Damien; McKay-Chopin, Sandrine; Calvez-Kelm, Florence Le et al. (2017). "Isolation and characterization of a novel putative human polyomavirus". Virology 506: 45–54. doi:10.1016/j.virol.2017.03.007. PMID 28342387.
- ↑ "Human polyomaviruses and cancer: an overview". Clinics (Sao Paulo) 73 (suppl 1): e558s. 2018. doi:10.6061/clinics/2018/e558s. PMID 30328951.
- ↑ "Human polyomaviruses in disease and cancer". Virology 437 (2): 63–72. March 2013. doi:10.1016/j.virol.2012.12.015. PMID 23357733.
- ↑ "Genome analysis of the new human polyomaviruses". Reviews in Medical Virology 22 (6): 354–77. November 2012. doi:10.1002/rmv.1711. PMID 22461085.
- ↑ "Prevalence of polyomavirus BK and JC infection and replication in 400 healthy blood donors". The Journal of Infectious Diseases 199 (6): 837–46. March 2009. doi:10.1086/597126. PMID 19434930.
- ↑ 63.0 63.1 "Is there a role for SV40 in human cancer?". Journal of Clinical Oncology 24 (26): 4356–65. September 2006. doi:10.1200/JCO.2005.03.7101. PMID 16963733.
- ↑ 64.0 64.1 "SV40 in human cancers--an endless tale?". International Journal of Cancer 107 (5): 687. December 2003. doi:10.1002/ijc.11517. PMID 14566815.
- ↑ "SV40 and human tumours: myth, association or causality?". Nature Reviews. Cancer 2 (12): 957–64. December 2002. doi:10.1038/nrc947. PMID 12459734.
- ↑ "Thirty-five year mortality following receipt of SV40- contaminated polio vaccine during the neonatal period". British Journal of Cancer 85 (9): 1295–7. November 2001. doi:10.1054/bjoc.2001.2065. PMID 11720463.
- ↑ "SV40 and human cancer: a review of recent data". International Journal of Cancer 120 (2): 215–23. January 2007. doi:10.1002/ijc.22425. PMID 17131333.
- ↑ 68.0 68.1 "Polyomavirus disease in renal transplantation: review of pathological findings and diagnostic methods". Human Pathology 36 (12): 1245–55. December 2005. doi:10.1016/j.humpath.2005.08.009. PMID 16311117.
- ↑ Viscidi, Raphael P.; Clayman, Barbara (2006). "Serological Cross Reactivity between Polyomavirus Capsids". in Ahsan, Nasimul. Polyomaviruses and Human Diseases. Advances in Experimental Medicine and Biology. 577. pp. 73–84. doi:10.1007/0-387-32957-9_5. ISBN 978-0-387-29233-5. https://books.google.com/books?id=Wz2aOQvHEPQC&pg=PA73.
- ↑ "Quantification of human polyomavirus JC in brain tissue and cerebrospinal fluid of patients with progressive multifocal leukoencephalopathy by competitive PCR". Journal of Virological Methods 84 (1): 23–36. January 2000. doi:10.1016/S0166-0934(99)00128-7. PMID 10644084.
- ↑ "Polyomavirus infection of renal allograft recipients: from latent infection to manifest disease". Journal of the American Society of Nephrology 10 (5): 1080–9. May 1999. doi:10.1681/ASN.V1051080. PMID 10232695. http://jasn.asnjournals.org/cgi/pmidlookup?view=long&pmid=10232695.
- ↑ "Quantitation of viral DNA in renal allograft tissue from patients with BK virus nephropathy". Transplantation 74 (4): 485–8. August 2002. doi:10.1097/00007890-200208270-00009. PMID 12352906.
- ↑ "Merkel cell polyomavirus expression in merkel cell carcinomas and its absence in combined tumors and pulmonary neuroendocrine carcinomas". The American Journal of Surgical Pathology 33 (9): 1378–85. September 2009. doi:10.1097/PAS.0b013e3181aa30a5. PMID 19609205.
- ↑ "Human Merkel cell polyomavirus infection II. MCV is a common human infection that can be detected by conformational capsid epitope immunoassays". International Journal of Cancer 125 (6): 1250–6. September 2009. doi:10.1002/ijc.24509. PMID 19499548.
- ↑ "Quantitation of human seroresponsiveness to Merkel cell polyomavirus". PLOS Pathogens 5 (9): e1000578. September 2009. doi:10.1371/journal.ppat.1000578. PMID 19750217.
- ↑ "Association of Merkel cell polyomavirus-specific antibodies with Merkel cell carcinoma". Journal of the National Cancer Institute 101 (21): 1510–22. November 2009. doi:10.1093/jnci/djp332. PMID 19776382.
- ↑ Elizabeth Matisoo-Smith; K. Ann Horsburgh (2012). DNA for Archaeologists. Routledge. ISBN 978-1598746815.
- ↑ "A filterable agent, recovered from Ak leukemic extracts, causing salivary gland carcinomas in C3H mice". Proceedings of the Society for Experimental Biology and Medicine 83 (2): 414–21. June 1953. doi:10.3181/00379727-83-20376. PMID 13064287.
- ↑ "Neoplasms in mice inoculated with a tumor agent carried in tissue culture". Journal of the National Cancer Institute 20 (6): 1223–43. June 1958. doi:10.1093/jnci/20.6.1223. PMID 13549981.
- ↑ "Characteristics of the SE polyoma virus". American Journal of Public Health and the Nation's Health 49 (11): 1486–92. November 1959. doi:10.2105/AJPH.49.11.1486. PMID 13819251.
- ↑ "Glycosaminoglycans and sialylated glycans sequentially facilitate Merkel cell polyomavirus infectious entry". PLOS Pathogens 7 (7): e1002161. July 2011. doi:10.1371/journal.ppat.1002161. PMID 21829355.
- ↑ "Natural biology of polyomavirus middle T antigen". Microbiology and Molecular Biology Reviews 65 (2): 288–318; second and third pages, table of contents. June 2001. doi:10.1128/mmbr.65.2.288-318.2001. PMID 11381103.
- ↑ "Genome Sequence of a Fish-Associated Polyomavirus, Black Sea Bass (Centropristis striata) Polyomavirus 1". Genome Announcements 3 (1): e01476-14. January 2015. doi:10.1128/genomeA.01476-14. PMID 25635011.
- ↑ "Concurrence of Iridovirus, Polyomavirus, and a Unique Member of a New Group of Fish Papillomaviruses in Lymphocystis Disease-Affected Gilthead Sea Bream". Journal of Virology 90 (19): 8768–79. October 2016. doi:10.1128/JVI.01369-16. PMID 27440877.
External links
Wikidata ☰ Q18670504 entry
 |