Biology:Fatty acid synthase
| Fatty acid synthase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 2.3.1.85 | ||||||||
| CAS number | 9045-77-6 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
 Generic protein structure example |
Fatty acid synthase (FAS)[1] is an enzyme that in humans is encoded by the FASN gene.[2][3][4][5]
Fatty acid synthase is a multi-enzyme protein that catalyzes fatty acid synthesis. It is not a single enzyme but a whole enzymatic system composed of two identical 272 kDa multifunctional polypeptides, in which substrates are handed from one functional domain to the next.[1][6][7][8][9]
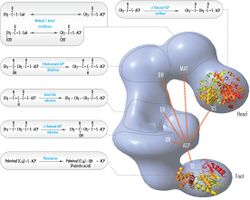
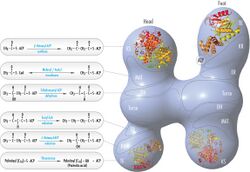
Its main function is to catalyze the synthesis of palmitate (C16:0, a long-chain saturated fatty acid) from acetyl-CoA and malonyl-CoA, in the presence of NADPH.[5]
The fatty acids are synthesized by a series of decarboxylative Claisen condensation reactions from acetyl-CoA and malonyl-CoA. Following each round of elongation the beta keto group is reduced to the fully saturated carbon chain by the sequential action of a ketoreductase (KR), dehydratase (DH), and enoyl reductase (ER). The growing fatty acid chain is carried between these active sites while attached covalently to the phosphopantetheine prosthetic group of an acyl carrier protein (ACP), and is released by the action of a thioesterase (TE) upon reaching a carbon chain length of 16 (palmitic acid).[1]
Classes
There are two principal classes of fatty acid synthases.
- Type I systems utilise a single large, multifunctional polypeptide and are common to both animals and fungi (although the structural arrangement of fungal and animal syntheses differ). A Type I fatty acid synthase system is also found in the CMN group of bacteria (corynebacteria, mycobacteria, and nocardia). In these bacteria, the FAS I system produces palmitic acid, and cooperates with the FAS II system to produce a greater diversity of lipid products.[10]
- Type II is found in archaea, bacteria and plant plastids, and is characterized by the use of discrete, monofunctional enzymes for fatty acid synthesis. Inhibitors of this pathway (FASII) are being investigated as possible antibiotics.[11]
The mechanism of FAS I and FAS II elongation and reduction is the same, as the domains of the FAS II enzymes are largely homologous to their domain counterparts in FAS I multienzyme polypeptides. However, the differences in the organization of the enzymes - integrated in FAS I, discrete in FAS II - gives rise to many important biochemical differences.[12]
The evolutionary history of fatty acid synthases are very much intertwined with that of polyketide synthases (PKS). Polyketide synthases use a similar mechanism and homologous domains to produce secondary metabolite lipids. Furthermore, polyketide synthases also exhibit a Type I and Type II organization. FAS I in animals is thought to have arisen through modification of PKS I in fungi, whereas FAS I in fungi and the CMN group of bacteria seem to have arisen separately through the fusion of FAS II genes.[10]
Structure
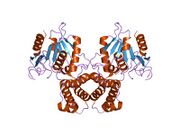
Mammalian FAS consists of a homodimer of two identical protein subunits, in which three catalytic domains in the N-terminal section (-ketoacyl synthase (KS), malonyl/acetyltransferase (MAT), and dehydrase (DH)), are separated by a core region (known as the interdomain) of 600 residues from four C-terminal domains (enoyl reductase (ER), -ketoacyl reductase (KR), acyl carrier protein (ACP) and thioesterase (TE)).[13][14] The interdomain region allows the two monomeric domains to form a dimer.[13]
The conventional model for organization of FAS (see the 'head-to-tail' model on the right) is largely based on the observations that the bifunctional reagent 1,3-dibromopropanone (DBP) is able to crosslink the active site cysteine thiol of the KS domain in one FAS monomer with the phosphopantetheine prosthetic group of the ACP domain in the other monomer.[15][16] Complementation analysis of FAS dimers carrying different mutations on each monomer has established that the KS and MAT domains can cooperate with the ACP of either monomer.[17][18] and a reinvestigation of the DBP crosslinking experiments revealed that the KS active site Cys161 thiol could be crosslinked to the ACP 4'-phosphopantetheine thiol of either monomer.[19] In addition, it has been recently reported that a heterodimeric FAS containing only one competent monomer is capable of palmitate synthesis.[20]
The above observations seemed incompatible with the classical 'head-to-tail' model for FAS organization, and an alternative model has been proposed, predicting that the KS and MAT domains of both monomers lie closer to the center of the FAS dimer, where they can access the ACP of either subunit (see figure on the top right).[21]
A low resolution X-ray crystallography structure of both pig (homodimer)[22] and yeast FAS (heterododecamer)[23] along with a ~6 Å resolution electron cryo-microscopy (cryo-EM) yeast FAS structure [24] have been solved.
Substrate shuttling mechanism
The solved structures of yeast FAS and mammalian FAS show two distinct organizations of highly conserved catalytic domains/enzymes in this multi-enzyme cellular machine. Yeast FAS has a highly efficient rigid barrel-like structure with 6 reaction chambers which synthesize fatty acids independently, while the mammalian FAS has an open flexible structure with only two reaction chambers. However, in both cases the conserved ACP acts as the mobile domain responsible for shuttling the intermediate fatty acid substrates to various catalytic sites. A first direct structural insight into this substrate shuttling mechanism was obtained by cryo-EM analysis, where ACP is observed bound to the various catalytic domains in the barrel-shaped yeast fatty acid synthase.[24] The cryo-EM results suggest that the binding of ACP to various sites is asymmetric and stochastic, as also indicated by computer-simulation studies[25]
Regulation
Metabolism and homeostasis of fatty acid synthase is transcriptionally regulated by Upstream Stimulatory Factors (USF1 and USF2) and sterol regulatory element binding protein-1c (SREBP-1c) in response to feeding/insulin in living animals.[26][27]
Although liver X receptors (LXRs) modulate the expression of sterol regulatory element binding protein-1c (SREBP-1c) in feeding, regulation of FAS by SREBP-1c is USF-dependent.[27][28][29][30]
Acylphloroglucinols isolated from the fern Dryopteris crassirhizoma show a fatty acid synthase inhibitory activity.[31]
Clinical significance
The FASN gene has been investigated as a possible oncogene.[32] FAS is upregulated in breast and gastric cancers, as well as being an indicator of poor prognosis, and so may be worthwhile as a chemotherapeutic target.[33][34][35] FAS inhibitors are therefore an active area of drug discovery research.[36][37][38][39][40]
FAS may also be involved in the production of an endogenous ligand for the nuclear receptor PPARalpha, the target of the fibrate drugs for hyperlipidemia,[41] and is being investigated as a possible drug target for treating the metabolic syndrome.[42] Orlistat which is a gastrointestinal lipase inhibitor also inhibits FAS and has a potential as a medicine for cancer.[43][44]
In some cancer cell lines, this protein has been found to be fused with estrogen receptor alpha (ER-alpha), in which the N-terminus of FAS is fused in-frame with the C-terminus of ER-alpha.[5]
An association with uterine leiomyomata has been reported.[45]
See also
- Discovery and development of gastrointestinal lipase inhibitors
- Fatty acid synthesis
- Fatty acid metabolism
- Fatty acid degradation
- Enoyl-acyl carrier protein reductase
- List of fatty acid metabolism disorders
References
- ↑ 1.0 1.1 1.2 Paiva, Pedro; Medina, Fabiola E.; Viegas, Matilde; Ferreira, Pedro; Neves, Rui P. P.; Sousa, João P. M.; Ramos, Maria J.; Fernandes, Pedro A. (2021-08-11). "Animal Fatty Acid Synthase: A Chemical Nanofactory" (in en). Chemical Reviews 121 (15): 9502–9553. doi:10.1021/acs.chemrev.1c00147. ISSN 0009-2665. PMID 34156235. https://pubs.acs.org/doi/10.1021/acs.chemrev.1c00147.
- ↑ "Isolation and chromosomal mapping of genomic clones encoding the human fatty acid synthase gene". Genomics 23 (2): 420–424. February 1995. doi:10.1006/geno.1994.1518. PMID 7835891.
- ↑ "Human fatty acid synthase: properties and molecular cloning". Proceedings of the National Academy of Sciences of the United States of America 92 (19): 8695–8699. Oct 1995. doi:10.1073/pnas.92.19.8695. PMID 7567999. Bibcode: 1995PNAS...92.8695J.
- ↑ "The SDR (short-chain dehydrogenase/reductase and related enzymes) nomenclature initiative". Chemico-Biological Interactions 178 (1–3): 94–98. Feb 2009. doi:10.1016/j.cbi.2008.10.040. PMID 19027726.
- ↑ 5.0 5.1 5.2 "Entrez Gene: FASN fatty acid synthase". https://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=2194.
- ↑ "Regulation of synthesis of hepatic fatty acid synthetase: binding of fatty acid synthetase antibodies to polysomes". Proceedings of the National Academy of Sciences of the United States of America 72 (10): 3956–3960. October 1975. doi:10.1073/pnas.72.10.3956. PMID 1060077. Bibcode: 1975PNAS...72.3956A.
- ↑ "Presence of two polypeptide chains comprising fatty acid synthetase". Proceedings of the National Academy of Sciences of the United States of America 72 (5): 1940–1944. May 1975. doi:10.1073/pnas.72.5.1940. PMID 1098047. Bibcode: 1975PNAS...72.1940S.
- ↑ "Specific release of the thioesterase component of the fatty acid synthetase multienzyme complex by limited trypsinization". Proceedings of the National Academy of Sciences of the United States of America 73 (4): 1184–1188. April 1976. doi:10.1073/pnas.73.4.1184. PMID 1063400. Bibcode: 1976PNAS...73.1184S.
- ↑ "Structural and functional organization of the animal fatty acid synthase". Progress in Lipid Research 42 (4): 289–317. July 2003. doi:10.1016/S0163-7827(02)00067-X. PMID 12689621.
- ↑ 10.0 10.1 "Evolutionary implications of bacterial polyketide synthases". Molecular Biology and Evolution 22 (10): 2027–2039. October 2005. doi:10.1093/molbev/msi193. PMID 15958783.
- ↑ Fulmer T (March 2009). "Not so FAS". Science-Business EXchange 2 (11): 430. doi:10.1038/scibx.2009.430.
- ↑ Fundamentals of enzymology: the cell and molecular biology of catalytic proteins. Oxford [Oxfordshire]: Oxford University Press. 1999. ISBN 978-0-19-850229-6.
- ↑ 13.0 13.1 "Human fatty acid synthase: role of interdomain in the formation of catalytically active synthase dimer". Proceedings of the National Academy of Sciences of the United States of America 98 (6): 3104–3108. March 2001. doi:10.1073/pnas.051635998. PMID 11248039. Bibcode: 2001PNAS...98.3104C.
- ↑ Smith S (December 1994). "The animal fatty acid synthase: one gene, one polypeptide, seven enzymes". FASEB Journal 8 (15): 1248–1259. doi:10.1096/fasebj.8.15.8001737. PMID 8001737.
- ↑ "Animal fatty acid synthetase. A novel arrangement of the beta-ketoacyl synthetase sites comprising domains of the two subunits". Journal of Biological Chemistry 256 (10): 5128–5133. May 1981. doi:10.1016/S0021-9258(19)69376-2. PMID 6112225.
- ↑ "Animal fatty acid synthetase. Identification of the residues comprising the novel arrangement of the beta-ketoacyl synthetase site and their role in its cold inactivation". Journal of Biological Chemistry 257 (6): 3230–3235. March 1982. doi:10.1016/S0021-9258(19)81100-6. PMID 7061475.
- ↑ "Differential affinity labeling of the two subunits of the homodimeric animal fatty acid synthase allows isolation of heterodimers consisting of subunits that have been independently modified". Journal of Biological Chemistry 273 (9): 4937–4943. February 1998. doi:10.1074/jbc.273.9.4937. PMID 9478938.
- ↑ "Mapping the functional topology of the animal fatty acid synthase by mutant complementation in vitro". Biochemistry 40 (36): 10792–18799. September 2001. doi:10.1021/bi015535z. PMID 11535054.
- ↑ "Dibromopropanone cross-linking of the phosphopantetheine and active-site cysteine thiols of the animal fatty acid synthase can occur both inter- and intrasubunit. Reevaluation of the side-by-side, antiparallel subunit model". Journal of Biological Chemistry 274 (17): 11557–11563. April 1999. doi:10.1074/jbc.274.17.11557. PMID 10206962.
- ↑ "Engineering of an active animal fatty acid synthase dimer with only one competent subunit". Chemistry and Biology 10 (2): 169–173. February 2003. doi:10.1016/S1074-5521(03)00023-1. PMID 12618189.
- ↑ "Structure and molecular organization of mammalian fatty acid synthase". Nature Structural and Molecular Biology 12 (3): 225–232. March 2005. doi:10.1038/nsmb899. PMID 15711565.
- ↑ "The crystal structure of a mammalian fatty acid synthase". Science 321 (5894): 1315–1322. September 2008. doi:10.1126/science.1161269. PMID 18772430. Bibcode: 2008Sci...321.1315M.
- ↑ "The crystal structure of yeast fatty acid synthase, a cellular machine with eight active sites working together". Cell 129 (2): 319–332. April 2007. doi:10.1016/j.cell.2007.03.013. PMID 17448991.
- ↑ 24.0 24.1 "Direct structural insight into the substrate-shuttling mechanism of yeast fatty acid synthase by electron cryomicroscopy". Proceedings of the National Academy of Sciences of the United States of America 107 (20): 9164–9169. May 2010. doi:10.1073/pnas.0913547107. PMID 20231485. Bibcode: 2010PNAS..107.9164G.
- ↑ "Mechanism of substrate shuttling by the acyl-carrier protein within the fatty acid mega-synthase". Journal of the American Chemical Society 132 (35): 12357–12364. September 2010. doi:10.1021/ja103354w. PMID 20704262.
- ↑ "Hormonal regulation of mouse fatty acid synthase gene transcription in liver". Journal of Biological Chemistry 264 (1): 574–577. January 1989. doi:10.1016/S0021-9258(17)31298-X. PMID 2535847.
- ↑ 27.0 27.1 "Occupancy and function of the -150 sterol regulatory element and -65 E-box in nutritional regulation of the fatty acid synthase gene in living animals". Molecular and Cellular Biology 23 (16): 5896–5907. August 2003. doi:10.1128/MCB.23.16.5896-5907.2003. PMID 12897158.
- ↑ "Direct interaction between USF and SREBP-1c mediates synergistic activation of the fatty-acid synthase promoter". Journal of Biological Chemistry 282 (8): 5453–5467. February 2007. doi:10.1074/jbc.M610566200. PMID 17197698.
- ↑ "Identification of liver X receptor-retinoid X receptor as an activator of the sterol regulatory element-binding protein 1c gene promoter". Molecular and Cellular Biology 21 (9): 2991–3000. May 2001. doi:10.1128/MCB.21.9.2991-3000.2001. PMID 11287605.
- ↑ "Regulation of mouse sterol regulatory element-binding protein-1c gene (SREBP-1c) by oxysterol receptors, LXRalpha and LXRbeta". Genes & Development 14 (22): 2819–2830. November 2000. doi:10.1101/gad.844900. PMID 11090130.
- ↑ "Fatty acid synthase inhibitory activity of acylphloroglucinols isolated from Dryopteris crassirhizoma". Bioorganic & Medicinal Chemistry Letters 16 (18): 4738–4742. September 2006. doi:10.1016/j.bmcl.2006.07.018. PMID 16870425.
- ↑ "Fatty acid synthase: a metabolic oncogene in prostate cancer?". Journal of Cellular Biochemistry 91 (1): 47–53. January 2004. doi:10.1002/jcb.10708. PMID 14689581.
- ↑ "MRNA stability and overexpression of fatty acid synthase in human breast cancer cell lines". Anticancer Research 27 (1A): 27–34. 2007. PMID 17352212.
- ↑ "Increased expression of fatty acid synthase (OA-519) in ovarian neoplasms predicts shorter survival". Human Pathology 28 (6): 686–692. June 1997. doi:10.1016/S0046-8177(97)90177-5. PMID 9191002.
- ↑ "Clinical importance of FASN in relation to HIF-1α and SREBP-1c in gastric adenocarcinoma". Life Sciences 224: 169–176. May 2019. doi:10.1016/j.lfs.2019.03.056. PMID 30914315. https://www.sciencedirect.com/science/article/abs/pii/S0024320519302206.
- ↑ "First Human Study Taking Place With Fatty Acid Synthase Inhibitor". oncotherapynetwork.com. April 7, 2017. http://www.oncotherapynetwork.com/breast-cancer-targets/first-human-study-taking-place-fatty-acid-synthase-inhibitor.
- ↑ "Design and synthesis of a series of bioavailable fatty acid synthase (FASN) KR domain inhibitors for cancer therapy". Bioorganic & Medicinal Chemistry Letters 28 (12): 2159–2164. May 2018. doi:10.1016/j.bmcl.2018.05.014. PMID 29779975.
- ↑ "A human fatty acid synthase inhibitor binds β-ketoacyl reductase in the keto-substrate site". Nature Chemical Biology 10 (9): 774–779. September 2014. doi:10.1038/nchembio.1603. PMID 25086508.
- ↑ "Understanding the Intersections between Metabolism and Cancer Biology". Cell 168 (4): 657–669. February 2017. doi:10.1016/j.cell.2016.12.039. PMID 28187287.
- ↑ Sgro, Christopher David (2009-01-01). An investigation into the interdomain region of Caenorhabditis elegans fatty acid synthase and its implications as a drug target (thesis thesis). La Trobe.
- ↑ "Identification of a physiologically relevant endogenous ligand for PPARalpha in liver.". Cell 138 (3): 476–488. August 2009. doi:10.1016/j.cell.2009.05.036. PMID 19646743.
- ↑ "Antidiabetic and antisteatotic effects of the selective fatty acid synthase (FAS) inhibitor platensimycin in mouse models of diabetes.". Proceedings of the National Academy of Sciences of the United States of America 108 (13): 5378–5383. March 2011. doi:10.1073/pnas.1002588108. PMID 21389266. Bibcode: 2011PNAS..108.5378W.
- ↑ "Fatty acid synthase as a potential therapeutic target in cancer". Future Oncology 6 (4): 551–562. April 2010. doi:10.2217/fon.10.11. PMID 20373869.
- ↑ "Synthesis of novel beta-lactone inhibitors of fatty acid synthase". Journal of Medicinal Chemistry 51 (17): 5285–5296. September 2008. doi:10.1021/jm800321h. PMID 18710210.
- ↑ "Genome-wide linkage and association analyses implicate FASN in predisposition to uterine leiomyomata". American Journal of Human Genetics 91 (4): 621–628. 2012. doi:10.1016/j.ajhg.2012.08.009. PMID 23040493.
Further reading
- Wakil SJ (1989). "Fatty acid synthase, a proficient multifunctional enzyme". Biochemistry 28 (11): 4523–4530. doi:10.1021/bi00437a001. PMID 2669958.
- "Fatty acid synthase: a metabolic oncogene in prostate cancer?". Journal of Cellular Biochemistry 91 (1): 47–53. 2004. doi:10.1002/jcb.10708. PMID 14689581.
- Lejin D (1978). "[Viscosimetry in clinical practice]". Medicinski Pregled 30 (9–10): 477–482. PMID 600212.
- Wronkowski Z (1976). "[Cancer diagnosis of the respiratory system]". Pielȩgniarka I Połozna (12): 7–8. PMID 1044453.
- "Human fatty acid synthase mRNA: tissue distribution, genetic mapping, and kinetics of decay after glucose deprivation". Journal of Lipid Research 36 (7): 1507–1521. 1995. doi:10.1016/S0022-2275(20)39738-8. PMID 7595075.
- "Fatty acid synthesis: a potential selective target for antineoplastic therapy". Proceedings of the National Academy of Sciences of the United States of America 91 (14): 6379–6383. 1994. doi:10.1073/pnas.91.14.6379. PMID 8022791. Bibcode: 1994PNAS...91.6379K.
- "Human fatty-acid synthase gene. Evidence for the presence of two promoters and their functional interaction". Journal of Biological Chemistry 271 (23): 13584–13592. 1996. doi:10.1074/jbc.271.23.13584. PMID 8662758.
- "Expression of fatty acid synthase is closely linked to proliferation and stromal decidualization in cycling endometrium". International Journal of Gynecological Pathology 16 (1): 45–51. 1997. doi:10.1097/00004347-199701000-00008. PMID 8986532.
- "Human fatty acid synthase: assembling recombinant halves of the fatty acid synthase subunit protein reconstitutes enzyme activity". Proceedings of the National Academy of Sciences of the United States of America 94 (23): 12326–12330. 1997. doi:10.1073/pnas.94.23.12326. PMID 9356448. Bibcode: 1997PNAS...9412326J.
- "Fatty acid synthase is expressed mainly in adult hormone-sensitive cells or cells with high lipid metabolism and in proliferating fetal cells". Journal of Histochemistry and Cytochemistry 48 (5): 613–622. 2000. doi:10.1177/002215540004800505. PMID 10769045.
- "Identification of a novel FAS/ER-alpha fusion transcript expressed in human cancer cells". Biochimica et Biophysica Acta (BBA) - Gene Structure and Expression 1493 (3): 373–377. 2000. doi:10.1016/s0167-4781(00)00202-5. PMID 11018265.
- "FIST/HIPK3: a Fas/FADD-interacting serine/threonine kinase that induces FADD phosphorylation and inhibits fas-mediated Jun NH(2)-terminal kinase activation". Journal of Experimental Medicine 192 (8): 1165–1174. 2000. doi:10.1084/jem.192.8.1165. PMID 11034606.
- "Human fatty acid synthase: role of interdomain in the formation of catalytically active synthase dimer". Proceedings of the National Academy of Sciences of the United States of America 98 (6): 3104–3108. 2001. doi:10.1073/pnas.051635998. PMID 11248039. Bibcode: 2001PNAS...98.3104C.
- "Quaternary structure of human fatty acid synthase by electron cryomicroscopy". Proceedings of the National Academy of Sciences of the United States of America 99 (1): 138–143. 2002. doi:10.1073/pnas.012589499. PMID 11756679. Bibcode: 2002PNAS...99..138B.
- "Direct and indirect mechanisms for regulation of fatty acid synthase gene expression by liver X receptors". Journal of Biological Chemistry 277 (13): 11019–11025. 2002. doi:10.1074/jbc.M111041200. PMID 11790787.
- "Domain movements in human fatty acid synthase by quantized elastic deformational model". Proceedings of the National Academy of Sciences of the United States of America 99 (12): 7895–7899. 2002. doi:10.1073/pnas.112222299. PMID 12060737. Bibcode: 2002PNAS...99.7895M.
- "Polyunsaturated fatty acids decrease the expression of sterol regulatory element-binding protein-1 in CaCo-2 cells: effect on fatty acid synthesis and triacylglycerol transport". Biochemical Journal 368 (Pt 3): 855–864. 2003. doi:10.1042/BJ20020731. PMID 12213084.
External links
- Fatty+Acid+Synthase at the US National Library of Medicine Medical Subject Headings (MeSH)
- Fatty Acid Synthesis: Rensselaer Polytechnic Institute
- Fatty Acid Synthase: RCSB PDB Molecule of the Month
- 3D electron microscopy structures of fatty acid synthase from the EM Data Bank(EMDB)
- PDBe-KB provides an overview of all the structure information available in the PDB for Human Fatty acid synthase
 |