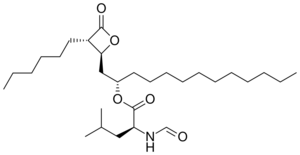
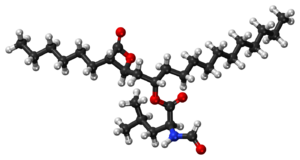
Biology:Orlistat
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Xenical, Alli |
| Other names | tetrahydrolipstatin |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601244 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Negligible[8] |
| Protein binding | >99% |
| Metabolism | In the GI tract |
| Elimination half-life | 1 to 2 hours |
| Excretion | Fecal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C29H53NO5 |
| Molar mass | 495.745 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Orlistat, sold under the brand name Xenical among others, is a medication used to treat obesity. Its primary function is preventing the absorption of fats from the human diet by acting as a lipase inhibitor, thereby reducing caloric intake. It is intended for use in conjunction with a healthcare provider-supervised reduced-calorie diet.[4]
Orlistat is the saturated derivative of lipstatin, a potent natural inhibitor of pancreatic lipases isolated from the bacterium Streptomyces toxytricini.[9] However, due to its relative simplicity and stability, orlistat was chosen over lipstatin for development as an anti-obesity drug.[10]
The effectiveness of orlistat in promoting weight loss is definite but modest. Pooled data from clinical trials suggest that people given orlistat in addition to lifestyle modifications, such as diet and exercise, lose about 2–3 kilograms (4–7 lb) more than those not taking the drug over the course of a year.[11] Orlistat also modestly reduces blood pressure and appears to prevent the onset of type 2 diabetes, whether from the weight loss itself or other effects. It reduces the incidence of diabetes type II in people who are obese around the same amount that lifestyle changes do.[12]
Benefits aside, however, orlistat is noted for its gastrointestinal side effects (sometimes referred to as treatment effects), which can include steatorrhea (oily, loose stools). They decrease with time, however, and are the most frequently reported adverse effects of the drug.[4] In Australia, the United States and the European Union, orlistat is available for sale without a prescription.[13] Over-the-counter approval was controversial in the United States, with consumer advocacy group Public Citizen repeatedly opposing it on safety and efficacy grounds.[14] Generic formulations of orlistat are available in some countries. In Australia it has been listed as an S3 medication, available from a pharmacist without a prescription, since 2000.[15]
Medical uses
Orlistat is used for the treatment of obesity. The amount of weight loss achieved with orlistat varies. In one-year clinical trials, between 35.5% and 54.8% of subjects achieved a 5% or greater decrease in body mass, although not all of this mass was necessarily fat. Between 16.4% and 24.8% achieved at least a 10% decrease in body fat.[4] After orlistat was stopped, a significant number of subjects regained weight—up to 35% of the weight they had lost.[4] It reduces the incidence of diabetes type II in people who are obese around the same amount that lifestyle changes do.[12] Long-term use of orlistat also leads to a very modest reduction in blood pressure (mean reductions of 2.5 and 1.9 mmHg in systolic and diastolic blood pressure respectively).[16]
Contraindications
Orlistat is contraindicated in:[4]
- Malabsorption
- Hypersensitivity to orlistat
- Reduced gallbladder function (e.g. after cholecystectomy)
- Pregnancy and breastfeeding
- Anorexia and Bulimia
- Use caution with: obstructed bile duct, impaired liver function, and pancreatic disease
Side effects
The primary side effects of the drug are gastrointestinal-related, and include steatorrhea (oily, loose stools with excessive flatus due to unabsorbed fats reaching the large intestine), fecal incontinence and frequent or urgent bowel movements.[17] To minimize these effects, foods with high fat content should be avoided; the manufacturer advises consumers to follow a low-fat, reduced-calorie diet. Oily stools and flatulence can be controlled by reducing the dietary fat content to somewhere in the region of 15 grams per meal.[18] The manual for Alli makes it clear that orlistat treatment involves aversion therapy, encouraging the user to associate eating fat with unpleasant treatment effects.[19]
Side effects are most severe when beginning therapy and may decrease in frequency with time;[4] It has also been suggested that the decrease in side effects over time may be associated with long-term compliance with a low-fat diet.[20]
On 26 May 2010, the U.S. Food and Drug Administration (FDA) approved a revised label for Xenical to include new safety information about cases of severe liver injury that have been reported rarely with the use of this medication.[21]
An analysis of over 900 orlistat users in Ontario showed that their rate of acute kidney injury was more than triple that of non-users.[22]
A study from 2013 looked at 94,695 participants receiving orlistat in the UK between 1999 and 2011.[23] The study showed no evidence of an increased risk of liver injury during treatment.[23] They concluded:[23]
The incidence of acute liver injury was higher in the periods both immediately before and immediately after the start of orlistat treatment. This suggests that the observed increased risks of liver injury linked to the start of treatment may reflect changes in health status associated with the decision to begin treatment rather than any causal effect of the drug.
Long-term
Despite a higher incidence of breast cancer amongst those taking orlistat in early, pooled clinical trial data—the analysis of which delayed FDA review of orlistat[24]—a two-year study published in 1999 found similar rates between orlistat and placebo (0.54% versus 0.51%), and evidence that tumors predated treatment in 3 of the 4 participants who had them.[25] There is evidence from an in vitro study to suggest that the introduction of specific varied preparations containing orlistat, namely the concurrent administration of orlistat and the monoclonal antibody trastuzumab, can induce cell death in breast cancer cells and block their growth.[26]
Fecal fat excretion promotes colon carcinogenesis. In 2006 the results of 30-day study were published indicating that orlistat at a dosage of 200 mg/kg chow administered to rats consuming a high-fat chow and receiving two 25 mg/kg doses of the potent carcinogen 1,2-dimethylhydrazine produced significantly higher numbers of aberrant crypt foci (ACF) colon lesions than did the carcinogen plus high-fat chow without orlistat.[27] ACF lesions are believed to be one of the earliest precursors of colon cancer.[28]
Precautions
Absorption of fat-soluble vitamins and other fat-soluble nutrients is inhibited by the use of orlistat.[4]
Interactions
Orlistat may reduce plasma levels of ciclosporin (also known as "cyclosporin" or "cyclosporine", trade names Sandimmune, Gengraf, Neoral, etc.), an immunosuppressive drug frequently used to prevent transplant rejection; the two drugs should therefore not be administered concomitantly.[4] Orlistat can also impair absorption of the antiarrhythmic amiodarone.[29] The Medicines and Healthcare products Regulatory Agency (MHRA) has suggested the possibility that orlistat could reduce the absorption of antiretroviral HIV medications.[30]
Mechanism of action
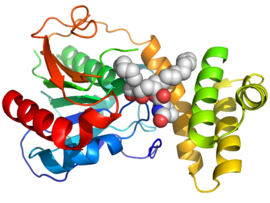
Orlistat works by inhibiting gastric and pancreatic lipases, the enzymes that break down triglycerides in the intestine.[32][33] When lipase activity is blocked, triglycerides from the diet are not hydrolyzed into absorbable free fatty acids, and instead are excreted unchanged. Only trace amounts of orlistat are absorbed systemically; the primary effect is local lipase inhibition within the GI tract after an oral dose. The primary route of elimination is through the feces.
Orlistat was also found to inhibit the thioesterase domain of fatty acid synthase (FAS), an enzyme involved in the proliferation of cancer cells but not normal cells. However, potential side effects of orlistat, such as inhibition of other cellular off-targets or poor bioavailability, might hamper its application as an effective antitumor agent. One profiling study undertook a chemical proteomics approach to look for new cellular targets of orlistat, including its off-targets.[34] Orlistat also shows potential activity against the Trypanosoma brucei parasite.[35]
Orlistat prevents approximately 30% of dietary fat from being absorbed.[36]
Legal status
Orlistat is available both with and without a prescription.[37][4][5][38]
Australia and New Zealand
In Australia and New Zealand, orlistat is available as a "Pharmacist Only Medicine".[15] In 2007, the Committee decided to keep orlistat as a Schedule 3 drug, but withdrew its authorization of direct-to-consumer Xenical advertising, stating this "increased pressure on pharmacists to provide orlistat to consumers...this in turn had the potential to result in inappropriate patterns of use".[39]
United States
Orlistat was initially approved by the U.S. Food and Drug Administration in April 1999 as a prescription-only medication.[40] On 23 January 2006, an FDA advisory panel voted 11 to 3 to recommend the approval of an OTC formulation of orlistat, to be sold under the brand name Alli by GlaxoSmithKline.[41] Approval was granted on 7 February 2007,[42] and Alli became the first weight loss drug officially sanctioned by the U.S. government for over-the-counter use.[43] Consumer advocacy organization Public Citizen opposed over-the-counter approval for orlistat.[14]
Alli became available in the U.S. in June 2007. It is sold as 60 mg capsules—half the dosage of prescription orlistat.[14][43]
European Union and Switzerland
On 21 January 2009, the European Medicines Agency granted approval for the sale of orlistat without a prescription.[37][44]
At least since September 2017, tablets with 60 mg orlistat can be freely sold in Swiss drugstores. Formulations with 120 mg per tablet require a prescription, but can be sold without one in pharmacies under an exemption rule, which is based on a list of easily diagnosable diseases.[45]
Generic formulations
U.S. patent protection for Xenical, originally to end on 18 June 2004, was extended by five years (until 2009) by the U.S. Patent and Trademark Office. The extension was granted on 20 July 2002,[46] and expired on 18 June 2009.[47]
Generic orlistat is available in Iran under the brand Venustat manufactured by Aburaihan Pharmaceutical co., in India, under the brands Orlean (Eris), Vyfat, Olistat, Obelit, Orlica and Reeshape.[48] In Russia, orlistat is available under the brand names Xenical (Hoffmann–La Roche), Orsoten/Orsoten Slim (KRKA d. d.) and Xenalten (OBL-Pharm). In Austria, orlistat is available under the brand name Slimox. In Malaysia, orlistat is available under the brand name Cuvarlix and is marketed by Pharmaniaga. In the Philippines orlistat is available under the brand name RedoXfat Plus manufactured by ATC Healthcare
Society and culture
Cost
At times, such as in spring 2012, orlistat has come into short supply, with consequent price increases because of nonavailability of one of the drug's components.[49]
Counterfeit products
In January 2010, the U.S. Food and Drug Administration issued an alert stating that some counterfeit versions of Alli sold over the Internet contain no orlistat, and instead contain the weight-loss drug sibutramine. The concentration of sibutramine in these counterfeit products is at least twice the amount recommended for weight loss.[50]
References
- ↑ "Xenical 120 mg hard capsules - Summary of Product Characteristics (SmPC)". 18 May 2017. https://www.medicines.org.uk/emc/product/2592/smpc.
- ↑ "Beacita 120mg Capsules, hard - Summary of Product Characteristics (SmPC)". 11 November 2020. https://www.medicines.org.uk/emc/product/2934/smpc.
- ↑ "alli 60 mg hard capsules - Summary of Product Characteristics (SmPC)". 11 June 2021. https://www.medicines.org.uk/emc/product/6533/smpc.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 "Xenical- orlistat capsule". 9 December 2021. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=6240792b-9224-2d10-e053-2a91aa0a2c3e.
- ↑ 5.0 5.1 "Alli- orlistat capsule". 9 November 2020. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=a2d3bd73-f3af-4ea5-a57c-66b0004cfe4f.
- ↑ "Xenical EPAR". 17 September 2018. https://www.ema.europa.eu/en/medicines/human/EPAR/xenical.
- ↑ "Alli EPAR". 17 September 2018. https://www.ema.europa.eu/en/medicines/human/EPAR/alli.
- ↑ "Review of limited systemic absorption of orlistat, a lipase inhibitor, in healthy human volunteers". Journal of Clinical Pharmacology 35 (11): 1103–1108. November 1995. doi:10.1002/j.1552-4604.1995.tb04034.x. PMID 8626884.
- ↑ "Syntheses of tetrahydrolipstatin and absolute configuration of tetrahydrolipstatin and lipstatin". Helvetica Chimica Acta 70 (1): 196–202. 1987. doi:10.1002/hlca.19870700124.
- ↑ "The first total synthesis of (−)-lipstatin". Journal of Organic Chemistry 60 (22): 7334–7339. 1995. doi:10.1021/jo00127a045.
- ↑ "Long-term pharmacotherapy for obesity and overweight". The Cochrane Database of Systematic Reviews 2003 (3): CD004094. 2004. doi:10.1002/14651858.CD004094.pub2. PMID 15266516.
- ↑ 12.0 12.1 "Pharmacological and lifestyle interventions to prevent or delay type 2 diabetes in people with impaired glucose tolerance: systematic review and meta-analysis". BMJ 334 (7588): 299. February 2007. doi:10.1136/bmj.39063.689375.55. PMID 17237299.
- ↑ "POISONS STANDARD JUNE 2017". Therapeutic Goods Administration. June 2017. https://www.legislation.gov.au/Details/F2017L00605.
- ↑ 14.0 14.1 14.2 "FDA OKs First Nonprescription Diet Pill". USA Today. 9 February 2007. https://www.usatoday.com/news/health/2007-02-09-diet-pill_x.htm.
- ↑ 15.0 15.1 "Orlistat 120mg capsule blister pack". TGA. 11 April 2000. https://www.ebs.tga.gov.au/servlet/xmlmillr6?dbid=ebs/PublicHTML/pdfStore.nsf&docid=61598&agid=(PrintDetailsPublic)&actionid=1.
- ↑ "Long-term effects of weight-reducing drugs in people with hypertension". The Cochrane Database of Systematic Reviews 1 (1): CD007654. January 2021. doi:10.1002/14651858.CD007654.pub5. PMID 33454957.
- ↑ "Treating Obesity". NHS. http://www.nhs.uk/Conditions/Obesity/Pages/Treatment.aspx.
- ↑ "FDA Approves alli (orlistat 60 mg capsules) Over-The-Counter" (PDF) (Press release). PR Newswire. 7 February 2007. Archived from the original (PDF) on 24 August 2007. Retrieved 8 April 2007.
- ↑ From page 12 of the Alli Companion Guide, 2007 edition: "They can be an incentive to keep from eating more fat than you really intend to."
- ↑ "Pharmacological treatment of obesity". Arquivos Brasileiros de Endocrinologia e Metabologia 50 (2): 377–389. April 2006. doi:10.1590/S0004-27302006000200024. PMID 16767304.
- ↑ "FDA Drug Safety Communication: Completed safety review of Xenical/Alli (orlistat) and severe liver injury". U.S. Food and Drug Administration (FDA). 28 June 2019. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/fda-drug-safety-communication-completed-safety-review-xenicalalli-orlistat-and-severe-liver-injury.
- ↑ "Orlistat and acute kidney injury: an analysis of 953 patients". Archives of Internal Medicine 171 (7): 703–704. April 2011. doi:10.1001/archinternmed.2011.103. PMID 21482850.
- ↑ 23.0 23.1 23.2 "Orlistat and the risk of acute liver injury: self controlled case series study in UK Clinical Practice Research Datalink". BMJ 346: f1936. April 2013. doi:10.1136/bmj.f1936. PMID 23585064.
- ↑ "Obesity Drug Can Lead to Modest Weight Loss, Study Finds". The New York Times. 20 January 1999. https://www.nytimes.com/1999/01/20/us/obesity-drug-can-lead-to-modest-weight-loss-study-finds.html?pagewanted=all.
- ↑ "Weight control and risk factor reduction in obese subjects treated for 2 years with orlistat: a randomized controlled trial". JAMA 281 (3): 235–242. January 1999. doi:10.1001/jama.281.3.235. PMID 9918478.
- ↑ "Antitumoral actions of the anti-obesity drug orlistat (XenicalTM) in breast cancer cells: blockade of cell cycle progression, promotion of apoptotic cell death and PEA3-mediated transcriptional repression of Her2/neu (erbB-2) oncogene". Annals of Oncology 16 (8): 1253–1267. August 2005. doi:10.1093/annonc/mdi239. PMID 15870086.
- ↑ "The anti-obesity agent Orlistat is associated to increase in colonic preneoplastic markers in rats treated with a chemical carcinogen". Cancer Letters 240 (2): 221–224. August 2006. doi:10.1016/j.canlet.2005.09.011. PMID 16377080.
- ↑ "Aberrant crypt foci of the colon as precursors of adenoma and cancer". The New England Journal of Medicine 339 (18): 1277–1284. October 1998. doi:10.1056/NEJM199810293391803. PMID 9791143.
- ↑ "Effects of orlistat, a lipase inhibitor, on the pharmacokinetics of three highly lipophilic drugs (amiodarone, fluoxetine, and simvastatin) in healthy volunteers". Journal of Clinical Pharmacology 43 (4): 428–435. April 2003. doi:10.1177/0091270003252236. PMID 12723464.
- ↑ "Orlistat: theoretical interaction with antiretroviral HIV medicines". Medicines and Healthcare products Regulatory Agency (MHRA). 13 March 2014. http://www.mhra.gov.uk/Safetyinformation/DrugSafetyUpdate/CON392868.
- ↑ PDB: 2PX6; "Crystal structure of the thioesterase domain of human fatty acid synthase inhibited by Orlistat". Nature Structural & Molecular Biology 14 (8): 704–709. August 2007. doi:10.1038/nsmb1265. PMID 17618296.
- ↑ "Orlistat, a new lipase inhibitor for the management of obesity". Pharmacotherapy 20 (3): 270–279. March 2000. doi:10.1592/phco.20.4.270.34882. PMID 10730683.
- ↑ "Orlistat & Xenical: Do Weight Loss Pills Work? | e-Surgery". 5 June 2020. https://e-surgery.com/orlistat-xenical-do-weight-loss-pills-work/.
- ↑ "Activity-based proteome profiling of potential cellular targets of Orlistat--an FDA-approved drug with anti-tumor activities". Journal of the American Chemical Society 132 (2): 656–666. January 2010. doi:10.1021/ja907716f. PMID 20028024. https://www.researchgate.net/publication/40758526. Retrieved 19 April 2019.
- ↑ "Parasite-based screening and proteome profiling reveal orlistat, an FDA-approved drug, as a potential anti Trypanosoma brucei agent". Chemistry: A European Journal 18 (27): 8403–8413. July 2012. doi:10.1002/chem.201200482. PMID 22674877. https://pubmed.ncbi.nlm.nih.gov/22674877/?from_single_result=orlistat+mycobacteria+Trypanosoma+brucei. Retrieved 9 June 2020.
- ↑ 2006 Physicians' Desk Reference (PDR). Thomson PDR. 2006. ISBN 978-1-56363-527-4. https://archive.org/details/physiciandeskref00mont.
- ↑ 37.0 37.1 "Chemists to provide obesity pill". BBC News Online. 21 January 2009. http://news.bbc.co.uk/2/hi/health/7843061.stm.
- ↑ "Orlistat". https://www.ema.europa.eu/en/medicines/human/referrals/orlistat.
- ↑ "Scheduling of orlistat" (Press release). Australian Therapeutic Goods Administration. 22 February 2007. Archived from the original on 29 December 2007.
- ↑ Stolberg, Sheryl (27 April 1999). "F.D.A. Approves Fat-Blocking Anti-Obesity Drug". https://www.nytimes.com/1999/04/27/us/fda-approves-fat-blocking-anti-obesity-drug.html.
- ↑ "Panel Supports Offering Diet Pill Orlistat Over the Counter". The Washington Post: pp. A02. 24 January 2006. https://www.washingtonpost.com/wp-dyn/content/article/2006/01/23/AR2006012301507.html.
- ↑ "FDA Approves Orlistat for Over-the-Counter Use" (Press release). U.S. Food and Drug Administration (FDA). 7 February 2007. Archived from the original on 13 May 2009. Retrieved 7 February 2007.
- ↑ 43.0 43.1 "Weight-Loss Drug to Be Sold Over the Counter". The New York Times. 7 February 2007. https://www.nytimes.com/2007/02/07/health/07cnd-diet.html.
- ↑ "GlaxoSmithKline receives European Commission approval to market alli (orlistat 60mg)" (Press release). GlaxoSmithKline. 21 January 2009. Archived from the original on 27 January 2009. Retrieved 22 January 2009.
- ↑ http://www.compendium.ch , directory of drugs approved in Switzerland
- ↑ "Certificate Extending Patent Term Under 35 U.S.C. § 156". United States Patent and Trademark Office. 30 July 2002. http://www.uspto.gov/web/offices/pac/dapp/opla/term/certs/4598089.pdf.
- ↑ "Drug Patent Expirations in June 2009". DrugPatentWatch.com, in "Drug Patent Expirations in June 2009". Biotech Blog. 1 June 2009. http://www.biotechblog.com/2009/06/01/drug-patent-expirations-in-june-2009.
- ↑ "Fatty issues". The Deccan Chronicle. 1 March 2009. http://www.deccanchronicle.com/sunday-debate/fatty-issues-694.
- ↑ Jeanne Whalen (20 April 2012). "Glaxo Sells Bulk of Over-the-Counter Drugs". The Wall Street Journal. https://www.wsj.com/articles/SB10001424052702303513404577355810163684518. "Glaxo said the issue wasn't a lack of interested buyers, but manufacturing problems that have led to shortages of the diet pill and forced the company to delay the product's sale."
- ↑ "Fake Alli diet pills can pose health risks". CNN. 23 January 2010. http://www.cnn.com/2010/HEALTH/01/23/fake.diet.drug/index.html.
Further reading
- "Synthesis of high specific activity [3H]-9-cis-retinoic acid and its application for identifying retinoids with unusual binding properties". Journal of Medicinal Chemistry 37 (3): 408–414. February 1994. doi:10.1021/jm00029a013. PMID 8308867.
- "Total synthesis of (−)-tetrahydrolipstatin". The Journal of Organic Chemistry 58 (27): 7768. 1993. doi:10.1021/jo00079a022.
- "Activity-based proteome profiling of potential cellular targets of Orlistat--an FDA-approved drug with anti-tumor activities". Journal of the American Chemical Society 132 (2): 656–666. January 2010. doi:10.1021/ja907716f. PMID 20028024.
- "Parasite-based screening and proteome profiling reveal orlistat, an FDA-approved drug, as a potential anti Trypanosoma brucei agent". Chemistry: A European Journal 18 (27): 8403–8413. July 2012. doi:10.1002/chem.201200482. PMID 22674877.
External links
- "Orlistat". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/orlistat.
 |
