Biology:MTAP
 Generic protein structure example |
| S-methyl-5-thioadenosine phosphorylase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
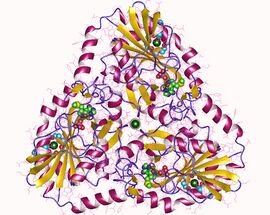 S-methyl-5'-thioadenosine phosphorylase trimer, Human | |||||||||
| Identifiers | |||||||||
| EC number | 2.4.2.28 | ||||||||
| CAS number | 61970-06-7 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
S-methyl-5'-thioadenosine phosphorylase (MTAP) is an enzyme responsible for polyamine metabolism. In humans, it is encoded by the methylthioadenosine phosphorylase (MTAP) gene on chromosome 9.[1] Multiple alternatively spliced transcript variants have been described for this gene, but their full-length natures remain unknown.[2]
This gene encodes an enzyme that plays a major role in polyamine metabolism and is important for the salvage of both adenine and methionine. It is responsible for the first step in this pathway, where it catalyzes the reversible phosphorylation of MTA to adenine and 5-methylthioribose-1-phosphate. This takes place after MTA is generated from S-adenosylmethionine.[1]
An additional role of MTAP has been found in the protozoan parasite Trypanosoma brucei, which causes African trypanosomiasis (sleeping sickness). The T. brucei MTAP has an unusually broad specificity and can cleave MTA as well as adenosine and deoxyadenosine.[3] The cleavage of deoxyadenosine serves as a protection mechanism to avoid the accumulation of toxic levels of dATP in the parasite (dATP is formed form deoxyadenosine). The cleavage activity has also consequences for drug discovery against African trypanosomiasis. It is important that adenosine analogues developed against the parasite are resistant to cleavage to be effective.[4]
MTAP was identified for the first time and characterized likely as a phosphorylase in 1969 by Pegg and Williams-Ashman.[5] The first purification that allowed characterization was by a group in 1986.[6] This purification allowed researchers to investigate why there is the lower expression of MTAP in some types of cancer.
Increased levels of MTA in tumor cells along with lower expression of MTAP.[7] The enzyme is deficient in many cancers because this gene and the tumor-suppressive p16 gene are co-deleted.[7][8][9][10][11]
Classification
This enzyme belongs to the family of glycosyltransferases, specifically the pentosyltransferases. The systematic name of this enzyme class is S-methyl-5-thioadenosine:phosphate S-methyl-5-thio-alpha-D-ribosyl-transferase. Other names in common use include 5'-methylthioadenosine nucleosidase, 5'-deoxy-5'-methylthioadenosine phosphorylase, MTA phosphorylase, MeSAdo phosphorylase, MeSAdo/Ado phosphorylase, methylthioadenosine phosphorylase, methylthioadenosine nucleoside phosphorylase, 5'-methylthioadenosine:phosphate methylthio-D-ribosyl-transferase, and S-methyl-5-thioadenosine phosphorylase. This enzyme participates in methionine metabolism.
Gene
The MTAP gene location is 9p21.3 which is chromosome 9, p arm, band 2, sub-band1, and sub-sub-band 3.[12][13] The MTAP gene has seven isomers which are created when mRNA's of the same locus have different transcription start sites.[14] Due to the nature of the MTAP gene and the surrounding genes of chromosome 9, deletion of the genes around p21, and gene p21 are common.[15] Particularly the deletion of the gene p16 in conjunction with the whole or partial deletion of MTAP has been indicated in some cancer types.[15] Genes p15 and p16 of chromosome nine are closely linked to the MTAP gene, because of this, MTAP is commonly cross-deleted.[8][9][15] This deletion is found in many cancerous tissues.[8][9]
Structure

MTAP is a trimer enzyme that shares a similar structure and functions with mammalian purine nucleoside phosphorylases (PNPs) which are also trimeric enzymes.[16] MTAP's subunits are identical in structure and composed of 283 amino acid residues that form to the size of about 32 kDa each.[6][16] The main structure of an MTAP subunit consists of eleven beta-sheets with six alpha-helices intermixed.[16] The active site of the enzyme is made up of beta-sheets five and 11, as well as alpha-helix 5, and four separate residue loop structures.[16] Within MTAP, helix six is a 12-residue C-terminal helix that arranges for the leucine residue 279 of one subunit to be a part of the active site of another subunit.[16] The active site of each subunit includes two residues (His137 and Leu279) from a neighboring subunit, relying on the interactions between the subunits for proper enzymatic activity.[16] MTAP contains an active site with three regions that correspond to a base, methylthioribose, and sulfate/phosphate binding site.[16]
Function
S-methyl-5'-thioadenosine phosphorylase, MTAP, primarily functions to salvage adenine and methionine from molecule methylthioadenosine (MTA), a byproduct of the polyamine pathway. MTAP is a phosphorylase, which is an enzyme that catalyzes the addition of an inorganic phosphate to another molecule. MTAP is responsible for the cleaving of its substrate, MTA, into adenine and 5-methylthioribose-1-phosphate by the addition of the inorganic phosphate to the 1-prime carbon of the ribose sugar unit MTA.[16] The 5-methylthioribose-1-phosphate is then cycled into the salvage pathway and metabolized into methionine.[18][19] The MTAP enzyme is responsible for nearly all the adenine synthesis in the human body.[16] Adenine is one of the purine bases of nucleic acids, which build both DNA and RNA. Through the recovery of adenine, MTAP plays an indirect role in the synthesis of DNA and RNA.
Cancer
In recent years a connection between tumor growth, cancer developments, and the enzyme MTAP. Research studies show that tumor cells have lower expression of MTAP enzymes and a higher concentration of the MTA molecule.[7] This trend can be easily understood through the polyamine pathway where MTAP functions to cleave its substrate MTA.[7][20] In healthy cells, the molecule MTA is believed to have tumor suppressing properties and regulate cell proliferation.[20] However, when MTA levels were recorded above optimal working conditions, these MTA molecules appeared to have an inverse relation, promoting tumor growth and significantly increasing the proliferation of tumor cells.[7] These increased levels of MTA in tumor cells is in direct correlation to a down regulation or complete deletion of the gene encoding the MTAP enzyme.[7]
References
- ↑ 1.0 1.1 1.2 "MTAP - S-methyl-5'-thioadenosine phosphorylase - Homo sapiens (Human) - MTAP gene & protein" (in en). https://www.uniprot.org/uniprot/Q13126.
- ↑ "MTAP methylthioadenosine phosphorylase [Homo sapiens (human) - Gene - NCBI"]. https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=ShowDetailView&TermToSearch=4507.
- ↑ "Methylthioadenosine Phosphorylase Protects the Parasite from the Antitrypanosomal Effect of Deoxyadenosine: implications for the pharmacology of adenosine antimetabolites". Journal of Biological Chemistry 291 (22): 11717–26. 2016. doi:10.1074/jbc.M116.715615. PMID 27036940.
- ↑ "9-(2'-Deoxy-2'-Fluoro-β-d-Arabinofuranosyl) Adenine Is a Potent Antitrypanosomal Adenosine Analogue That Circumvents Transport-Related Drug Resistance". Antimicrobial Agents and Chemotherapy 61 (6): e02719-16. 2016. doi:10.1128/AAC.02719-16. PMID 28373184.
- ↑ "Phosphate-stimulated breakdown of 5'-methylthioadenosine by rat ventral prostate". The Biochemical Journal 115 (2): 241–247. November 1969. doi:10.1042/bj1150241. PMID 5378381.
- ↑ 6.0 6.1 "Purification and characterization of 5'-deoxy-5'-methylthioadenosine phosphorylase from human placenta". The Journal of Biological Chemistry 261 (26): 12324–12329. September 1986. doi:10.1016/S0021-9258(18)67242-4. PMID 3091600.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 "Down-regulation of methylthioadenosine phosphorylase (MTAP) induces progression of hepatocellular carcinoma via accumulation of 5'-deoxy-5'-methylthioadenosine (MTA)". The American Journal of Pathology 178 (3): 1145–1152. March 2011. doi:10.1016/j.ajpath.2010.11.059. PMID 21356366.
- ↑ 8.0 8.1 8.2 "Homozygous MTAP deletion in primary human glioblastoma is not associated with elevation of methylthioadenosine". Nature Communications 12 (1). July 2021. doi:10.1038/s41467-021-24240-3. PMID 34244484. Bibcode: 2021NatCo..12.4228B.
- ↑ 9.0 9.1 9.2 "Loss of 5'-Methylthioadenosine Phosphorylase (MTAP) is Frequent in High-Grade Gliomas; Nevertheless, it is Not Associated with Higher Tumor Aggressiveness". Cells 9 (2): 492. February 2020. doi:10.3390/cells9020492. PMID 32093414.
- ↑ "Targeting the insulin-like growth factor-1 receptor in MTAP-deficient renal cell carcinoma". Signal Transduction and Targeted Therapy 4. 2019-01-25. doi:10.1038/s41392-019-0035-z. PMID 30701095.
- ↑ "MTAP Loss Promotes Stemness in Glioblastoma and Confers Unique Susceptibility to Purine Starvation". Cancer Research 79 (13): 3383–3394. July 2019. doi:10.1158/0008-5472.CAN-18-1010. PMID 31040154.
- ↑ "MTAP Gene". https://www.genecards.org/cgi-bin/carddisp.pl?gene=MTAP#genomic_location.
- ↑ "MLA CE Course Manual: Molecular Biology Information Resources (Genetics Review: Chromosome Band Numbers)". https://www.ncbi.nlm.nih.gov/Class/MLACourse/Original8Hour/Genetics/chrombanding.html.
- ↑ "UniProtKB - Q13126 (MTAP_Human)". https://www.uniprot.org/uniprot/Q13126#sequences.
- ↑ 15.0 15.1 15.2 "Genomic cloning of methylthioadenosine phosphorylase: a purine metabolic enzyme deficient in multiple different cancers". Proceedings of the National Academy of Sciences of the United States of America 93 (12): 6203–6208. June 1996. doi:10.1073/pnas.93.12.6203. PMID 8650244. Bibcode: 1996PNAS...93.6203N.
- ↑ 16.00 16.01 16.02 16.03 16.04 16.05 16.06 16.07 16.08 16.09 "The structure of human 5'-deoxy-5'-methylthioadenosine phosphorylase at 1.7 A resolution provides insights into substrate binding and catalysis". Structure 7 (6): 629–641. June 1999. doi:10.1016/S0969-2126(99)80084-7. PMID 10404592.
- ↑ Bank, RCSB Protein Data. "RCSB PDB - 3OZE: Crystal Structure of human 5'-deoxy-5'-methyladenosine phosphorylase" (in en-US). https://www.rcsb.org/structure/3OZE.
- ↑ 18.0 18.1 "Methylthioadenosine phosphorylase regulates ornithine decarboxylase by production of downstream metabolites". The Journal of Biological Chemistry 278 (50): 49868–49873. December 2003. doi:10.1074/jbc.M308451200. PMID 14506228.
- ↑ "Targeting tumors that lack methylthioadenosine phosphorylase (MTAP) activity: current strategies". Cancer Biology & Therapy 11 (7): 627–632. April 2011. doi:10.4161/cbt.11.7.14948. PMID 21301207.
- ↑ 20.0 20.1 "5'-Methylthioadenosine and Cancer: old molecules, new understanding". Journal of Cancer 10 (4): 927–936. January 2019. doi:10.7150/jca.27160. PMID 30854099.
Further reading
- "Assignment of the gene for methylthioadenosine phosphorylase to human chromosome 9 by mouse-human somatic cell hybridization". Proceedings of the National Academy of Sciences of the United States of America 81 (9): 2665–2668. May 1984. doi:10.1073/pnas.81.9.2665. PMID 6425836. Bibcode: 1984PNAS...81.2665C.
- "Construction of a 2.8-megabase yeast artificial chromosome contig and cloning of the human methylthioadenosine phosphorylase gene from the tumor suppressor region on 9p21". Proceedings of the National Academy of Sciences of the United States of America 92 (14): 6489–6493. July 1995. doi:10.1073/pnas.92.14.6489. PMID 7604019. Bibcode: 1995PNAS...92.6489O.
- "Purification and characterization of recombinant human 5'-methylthioadenosine phosphorylase: definite identification of coding cDNA". Biochemical and Biophysical Research Communications 223 (3): 514–519. June 1996. doi:10.1006/bbrc.1996.0926. PMID 8687427. Bibcode: 1996BBRC..223..514D.
- "Identification of a 1.2 Kb cDNA fragment from a region on 9p21 commonly deleted in multiple tumor types". Cancer Genetics and Cytogenetics 129 (2): 93–101. September 2001. doi:10.1016/S0165-4608(01)00444-7. PMID 11566337.
- "Methylthioadenosine phosphorylase, a gene frequently codeleted with p16(cdkN2a/ARF), acts as a tumor suppressor in a breast cancer cell line". Cancer Research 62 (22): 6639–6644. November 2002. PMID 12438261.
- "MSAP is a novel MIR-interacting protein that enhances neurite outgrowth and increases myosin regulatory light chain". The Journal of Biological Chemistry 278 (37): 35412–35420. September 2003. doi:10.1074/jbc.M306271200. PMID 12826659.
- "Characterization of methylthioadenosin phosphorylase (MTAP) expression in malignant melanoma". The American Journal of Pathology 163 (2): 683–690. August 2003. doi:10.1016/S0002-9440(10)63695-4. PMID 12875987.
- "Large-scale characterization of HeLa cell nuclear phosphoproteins". Proceedings of the National Academy of Sciences of the United States of America 101 (33): 12130–12135. August 2004. doi:10.1073/pnas.0404720101. PMID 15302935. Bibcode: 2004PNAS..10112130B.
- "Strong expression of methylthioadenosine phosphorylase (MTAP) in human colon carcinoma cells is regulated by TCF1/[beta]-catenin". Laboratory Investigation; A Journal of Technical Methods and Pathology 85 (1): 124–136. January 2005. doi:10.1038/labinvest.3700192. PMID 15492751.
- "Methylthioadenosine phosphorylase gene expression is impaired in human liver cirrhosis and hepatocarcinoma". Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 1690 (3): 276–284. November 2004. doi:10.1016/j.bbadis.2004.08.002. PMID 15511635.
- "Loss of methylthioadenosine phosphorylase and elevated ornithine decarboxylase is common in pancreatic cancer". Clinical Cancer Research 10 (21): 7290–7296. November 2004. doi:10.1158/1078-0432.CCR-04-0972. PMID 15534104.
- "Homozygous deletion of the MTAP gene in invasive adenocarcinoma of the pancreas and in periampullary cancer: a potential new target for therapy". Cancer Biology & Therapy 4 (1): 83–86. January 2005. doi:10.4161/cbt.4.1.1380. PMID 15662124.
- "Concordant loss of MTAP and p16/CDKN2A expression in pancreatic intraepithelial neoplasia: evidence of homozygous deletion in a noninvasive precursor lesion". Modern Pathology 18 (7): 959–963. July 2005. doi:10.1038/modpathol.3800377. PMID 15832197.
- "Promoter-hypermethylation is causing functional relevant downregulation of methylthioadenosine phosphorylase (MTAP) expression in hepatocellular carcinoma". Carcinogenesis 27 (1): 64–72. January 2006. doi:10.1093/carcin/bgi201. PMID 16081515.
- "5'-Methylthioadenosine phosphorylase from Caldariella acidophila Purification and properties". Eur. J. Biochem. 101 (2): 317–24. 1979. doi:10.1111/j.1432-1033.1979.tb19723.x. PMID 118001.
- Garbers DL (1978). "Demonstration of 5'-methylthioadenosine phosphorylase activity in various rat tissues. Some properties of the enzyme from rat lung". Biochim. Biophys. Acta 523 (1): 82–93. doi:10.1016/0005-2744(78)90011-6. PMID 415762.
- "Phosphate-stimulated breakdown of 5'-methylthioadenosine by rat ventral prostate". Biochem. J. 115 (2): 241–7. 1969. doi:10.1042/bj1150241. PMID 5378381.
External links
Structures include:
 |
