Chemistry:Potassium osmate
| |
| File:Potassium osmate(VI) dihydrate.jpg | |
| Names | |
|---|---|
| Other names
Potassium osmate(VI) dihydrate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
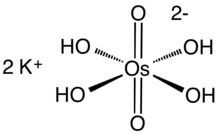
| K 2[OsO 2(OH) 4] | |
| Molar mass | 368.42 g/mol |
| Appearance | Garnet red[1] |
| Soluble[2] | |
| Solubility | Insoluble in alcohol or ether |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Danger |
| H301, H311, H330, H331 | |
| P260, P261, P264, P270, P271, P280, P284, P301+310, P302+352, P304+340, P310, P311, P312, P320, P321, P322, P330, P361, P363, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Potassium osmate is the inorganic compound with the formula K2[OsO2(OH)4]. This diamagnetic purple salt contains osmium in the VI (6+) oxidation state.[4] It gained attention as a catalyst for the asymmetric dihydroxylation of olefins.[5]
Properties
Solid potassium osmate is stable in air. It yields a red solution when dissolved in water, with which it slowly decomposes producing osmium tetroxide, however when prepared by reducing potassium perosmate with alcohol it yields a pink solution.[6][7] It is more stable in cold water under alkaline conditions, but hot water or acid accelerates decomposition. When exposed to sunlight for prolonged periods of time, it decomposes and produces a sooty powder along with the odor of the tetroxide. It is insoluble in alcohol and ether.[1][8][9]
Structure
The complex anion is octahedral. Like related d2 dioxo complexes, the oxo ligands are trans.[10] The Os=O and Os-OH distances are 1.75(2) and 1.99(2) Å, respectively. It is a relatively rare example of a metal oxo complex that obeys the 18e rule.
Preparation
The compound was first reported by Edmond Frémy in 1844.[11] Potassium osmate is prepared by reducing osmium tetroxide with ethanol:[12]
- 2 OsO4 + C2H5OH + 5 KOH → CH3CO2K + 2 K2[OsO2(OH)4]
Alkaline oxidative fusion of osmium metal also affords this salt.[4]
Reactions
Potassium osmate reacts with acids to produce osmyl salts, such as potassium osmyl chloride and the osmyl bromide. It reacts with oxalic acid to produce potassium osmyl oxalate.[9][1]
- K
2[OsO
2(OH)
4] + 4 HCl → K
2[OsO
2Cl
4] + 4 H
2O - K
2[OsO
2(OH)
4] + 2 H
2C
2O
4 → K
2[OsO
2(C
2O
4)
2 + 4 H
2O
- K
Potassium osmate reacts with potassium nitrite to yield potassium osmyl nitrite, and it reacts with ammonium salts to produce osmyl tetra-ammine compounds such as [OsO
2(NH
3)
4]SO
4 and [OsO
2(NH
3)
4]Cl
2.[9][13]
Potassium osmate can be reduced to osmium dioxide with alcohol.[1][9]
- K
2[OsO
2(OH)
4] + C
2H
5OH → OsO
2(H
2O)
2 + 2KOH + CH
3CHO
Potassium osmate can be used to prepare many other compounds. If potassium osmate is refluxed with HCl, it can be reduced to hexachloroosmic acid. When refluxed with HBr, it can be reduced to hexabromoosmic acid. It reacts with hydrogen sulfide to produce osmium disulfide, OsS
2, and with sulfurous acid to form potassium osmium sulfite.[9][8]
Applications
Potassium osmate is used as a catalyst for the Sharpless dihydroxylation of olefins. In this application, it shuttles to Os(VIII) derivative, which adds to the olefin. Osmium tetroxide functions equivalently.
Related compounds
Other osmates are known, such as the highly water soluble sodium osmate, and insoluble osmates such as those of barium, strontium, calcium, and lead.[9][8]
When osmium tetroxide is treated with an alkali hydroxide, or even when potassium osmate is oxidized in alkaline medium, the perosmate anion is formed, [OsO
4(OH)
2]2-, which possesses a yellow color in solution. When treated with alcohol, perosmates are reduced to osmates, [OsO
2(OH)
4]2-, yielding a pink color in solution. Perosmate salts share the same valency as osmium tetroxide, as opposed to perruthenates. The perosmates of sodium, potassium, rubidium, cesium, and barium are known.[14][15][16]
- OsO
4 + 2KOH → K
2[OsO
4(OH)
2][17]
When osmium tetroxide is dissolved in anhydrous methanol containing potassium hydroxide, a blue solution of dipotassium tetramethyl osmate (DTMO) is obtained. DTMO was used as a staining agent for electron microscopy.[18]
- OsO
4 + 5CH
3OH + 2KOH → K
2[OsO
2(OCH
3)
4] + H
2CO + 4H
2O
See also
References
- ↑ 1.0 1.1 1.2 1.3 J. Newton Friend (1920). "Osmium and its compounds". A textbook of inorganic chemistry, vol.IX Part I Cobalt, Nickel, and The Elements of The Platinum Group. London: Charles Griffin and Company, Limited. pp. 219–225, 231. https://dn790003.ca.archive.org/0/items/textbookinorgani00libg_516/textbookinorgani00libg_516.pdf. Retrieved 3 July 2025.
- ↑ Comey, Arthur Messinger (1896) (in en). A Dictionary of Chemical Solubilities: Inorganic. Macmillan and Company. p. 275. https://books.google.com/books?id=H9bPAAAAMAAJ&pg=PA275. Retrieved 6 July 2025.
- ↑ "Potassium osmate(VI) dihydrate". Chemical Book. https://www.chemicalbook.com/ChemicalProductProperty_EN_CB3421530.htm.
- ↑ 4.0 4.1 F. Albert Cotton; Geoffrey Wilkinson (1966). Advanced Inorganic Chemistry: A Comprehensive Treatise. New York, Interscience Publishers. p. 1007. https://archive.org/details/advancedinorgani00cott.
- ↑ Li, Guigen; Chang, Han-Ting; Sharpless, K. Barry (1996). "Catalytic Asymmetric Aminohydroxylation (AA) of Olefins". Angew. Chem. Int. Ed. Engl. 35 (4): 451–4. doi:10.1002/anie.199604511.
- ↑ Fremy, Edmund. (1900). "Osmium". Encyclopédie chimique.Tome III.- Métaux. 17e cahier : platine et métaux qui l'accompagnent. Paris : Dunod. pp. 64. https://archive.org/details/BIUSante_pharma_018935x30x1/page/n39/mode/2up. Retrieved 20 November 2025.
- ↑ Sidgwick, N.V. (1950). The Elements and Their Compounds. 2. London: Oxford University Press. pp. 1499-1500. http://www.sciencemadness.org/library/books/the_chemical_elements_and_their_compounds_v2.pdf. Retrieved 20 November 2025.
- ↑ 8.0 8.1 8.2 J.W. Mellor (May 1936). "Osmium". A comprehensive treatise on inorganic and theoretical chemistry, Volume 15. London: Longmans, Green and Co. pp. 706. https://archive.org/details/Mellor13/Mellor_15/page/n713/mode/2up?q=%5BOsO%28NH3%292%5D%28OH%292. Retrieved 22 June 2025.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 Prakash Satya (2013). "Platinum Metals-IV:Osmium". Advanced Chemistry of Rare Elements, 5th Ed.. India: S Chand and Company Limited. pp. 611–622. https://books.google.com/books?id=WB_4DwAAQBAJ&dq=osmium+tetrasulfide+OsS4&pg=PA611#v=onepage&q&f=false. Retrieved 24 July 2025.
- ↑ R. K. Murmann, C. L. Barnes "Redetermination of the crystal structure of potassium trans-(dioxo)-tetra(hydroxo)osmate(VI), K2[Os(OH)4(O)2]" Z. Kristallogr. NCS 217, 2002, pp. 303–304. doi:10.1524/ncrs.2002.217.jg.303
- ↑ Frémy, E. "Ueber das Osmium" Journal für Praktische Chemie 1844 vol.33, 406-416. doi: 10.1002/prac.18440330160
- ↑ John M. Malin (1980). "Potassium Tetrahydroxodioxoosmate(VI) and trans ‐Bis(Ethylenediamine)Dioxoosmium(VI) Chloride". Potassium Tetrahydroxodioxoosmate(VI) and trans-Bis(Ethylenediamine)Dioxoosmium(VI) Chloride. Inorganic Syntheses. 20. pp. 61–63. doi:10.1002/9780470132517.ch18. ISBN 9780470132517.
- ↑ Burvikova, J. N.; Lin'Ko, I. V.; Venskovskiy, N. U.; Rybakov, V. B. (2007). "Crystal structure and properties of [OsO2(NH3)4]SO4 · H2O". Crystallography Reports 52 (2): 221–225. doi:10.1134/S1063774507020095.
- ↑ Williamson, J.; Savage, J. (January 1965). "The Determination of Osmiridium in Witwatersrand Ores". Journal of the South African Institute of Mining and Metallurgy: 350. https://journals.co.za/doi/pdf/10.10520/AJA0038223X_4905. Retrieved 7 July 2025.
- ↑ Stopinski, Orin (September 1977). "Platinum-Group Metals". Environmental Health Effects Research Series (Washington, DC: Environmental Protection Agency): 85–87. https://nepis.epa.gov/Exe/ZyPDF.cgi/91013EPF.PDF?Dockey=91013EPF.PDF. Retrieved 19 June 2025.
- ↑ Griffth, W.P. (1964). "Infrared Spectra and Structures of Osmium Oxy-complexes". Journal of the Chemical Society 40: 245-246. https://pubs.rsc.org/en/content/articlelanding/1964/jr/jr9640000245. Retrieved 21 November 2025.
- ↑ Cotton, S. A. (1997). "Ruthenium and Osmium". Chemistry of Precious Metals. Rutland, UK: Blackie Academic & Professional. pp. 20. https://tech.chemistrydocs.com/Books/General%20Chemistry/Chemistry-of-Precious-Metals-by-Dr-S-A-Cotton.pdf. Retrieved 11 July 2025.
- ↑ Hinckley, C.; Murphy, Judith (13 August 1974). "Dipotassium Tetramethyl Osmate: A Stain for Electron Microscopy". Journal of Histochemistry and Cytochemistry 23 (2): 123-127. https://journals.sagepub.com/doi/pdf/10.1177/23.2.1167876. Retrieved 6 July 2025.
 |

