Chemistry:Osmium dioxide
| |
 Osmium dioxide dihydrate
| |
| Names | |
|---|---|
| IUPAC name
Osmium dioxide
| |
| Other names
Osmium(IV) oxide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| OsO2 | |
| Molar mass | 222.229 g/mol |
| Appearance | black or yellow brown |
| Density | 11.4 g/cm3 |
| Melting point | 500 °C (932 °F; 773 K) (decomposes) |
| insoluble[1] | |
| Solubility | dissolves in HCl |
| Related compounds[2] | |
| Osmium tetroxide | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Osmium dioxide is an inorganic compound with the formula OsO
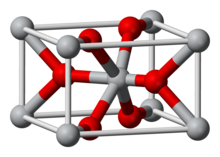
2. It exists as brown to black crystalline powder, but single crystals are golden and exhibit metallic conductivity. The compound crystallizes in the rutile structural motif, i.e. the connectivity is very similar to that in the mineral rutile.
Preparation
OsO
2 can be obtained by the reaction of osmium with a variety of oxidizing agents, including, sodium chlorate, osmium tetroxide, and nitric oxide at about 600 °C.[3][4] Using chemical transport, one can obtain large crystals of OsO
2, sized up to 7x5x3 mm3. Single crystals show metallic resistivity of ~15 μΩ cm. A typical transport agent is O2 via the reversible formation of volatile OsO
4:[5]
- OsO
2 + O
2 ⇌ OsO
4
It can also be prepared by reducing osmium in higher oxidation states with alcohol, in which it forms a dihydrate. As opposed to the anhydrous dioxide, the dihydrate possesses a bluish black appearance.[6][7]
- K
2[OsO
2(OH)
4] + C
2H
5OH → OsO
2•2H
2O + 2KOH + CH
3CHO
Adding strong alkali to chloroosmic acid or its salts also yields the dihydrate.[6][7][8]
- K
2OsCl
6 + 4KOH → 6KCl + OsO
2•2H
2O
Properties
Osmium dioxide does not dissolve in water, but it can be dissolved by strong acids such as hydrochloric acid.[9][10]
The crystals have rutile structure.[11] Unlike osmium tetroxide, OsO
2 is not toxic.[12]
Hexavalent osmium
Compounds of osmium in the +6 oxidation state are dominated by the osmyl species, in which OsO
2 exists as a radical trans-dioxo moiety. These osmyl compounds are all diamagnetic, and stabilized by strong σ-donor and π-donor ligands. Most osmyl compounds are mononuclear and have a linear O=Os=O structure.[6][13]
Examples of osmyl compounds include K
2[OsO
2(OH)
4], [OsO
2(NH
3)
4]Cl
2, and K
2(OsO
2)(C
2O
4)
2, but many others are known.
References
- ↑ Comey, Arthur Messinger (1896) (in en). A Dictionary of Chemical Solubilities: Inorganic. Macmillan and Company. p. 275. https://books.google.com/books?id=H9bPAAAAMAAJ&pg=PA275. Retrieved 3 July 2025.
- ↑ OsO2 at webelements
- ↑ A. F. Holleman; E. Wiberg (2001). Inorganic chemistry. Academic Press. p. 1465. ISBN 0-12-352651-5.
- ↑ Thiele G.; Woditsch P. (1969). "Neutronenbeugungsuntersuchungen am Osmium(IV)-oxid". Journal of the Less Common Metals 17 (4): 459. doi:10.1016/0022-5088(69)90074-5.
- ↑ Rogers, D. B.; Butler, S. R.; Shannon, R. D. (1972). "Single Crystals of Transition-Metal Dioxides". Inorganic Syntheses. XIII. pp. 135–145. doi:10.1002/9780470132449.ch27. ISBN 9780470132449.
- ↑ 6.0 6.1 6.2 J. Newton Friend (1920). "Osmium and its compounds". A textbook of inorganic chemistry, vol.IX Part I Cobalt, Nickel, and The Elements of The Platinum Group. London: Charles Griffin and Company, Limited. pp. 12, 216-219, 222. https://dn790003.ca.archive.org/0/items/textbookinorgani00libg_516/textbookinorgani00libg_516.pdf. Retrieved 3 July 2025.
- ↑ 7.0 7.1 Prakash Satya (2013). "Platinum Metals-IV:Osmium". Advanced Chemistry of Rare Elements, 5th Ed.. India: S Chand and Company Limited. pp. 611. https://books.google.com/books?id=WB_4DwAAQBAJ&dq=osmium+tetrasulfide+OsS4&pg=PA611#v=onepage&q&f=false. Retrieved 24 July 2025.
- ↑ Stopinski, Orin (September 1977). "Platinum-Group Metals". Environmental Health Effects Research Series (Washington, DC: Environmental Protection Agency): 85–86. https://nepis.epa.gov/Exe/ZyPDF.cgi/91013EPF.PDF?Dockey=91013EPF.PDF. Retrieved 3 July 2025.
- ↑ J. E. Greedan; D. B. Willson; T. E. Haas (1968). "Metallic nature of osmium dioxide". Inorg. Chem. 7 (11): 2461–2463. doi:10.1021/ic50069a059.
- ↑ Yen, P (2004). "Growth and characterization of OsO2 single crystals". Journal of Crystal Growth 262 (1–4): 271. doi:10.1016/j.jcrysgro.2003.10.021.
- ↑ Boman C.E.; Danielsen, Jacob; Haaland, Arne; Jerslev, Bodil; Schäffer, Claus Erik; Sunde, Erling; Sørensen, Nils Andreas (1970). "Precision Determination of the Crystal Structure of Osmium Dioxide". Acta Chemica Scandinavica 24: 123–128. doi:10.3891/acta.chem.scand.24-0123.
- ↑ Smith, I.C., B.L. Carson, and T.L. Ferguson (1974). "Osmium: An appraisal of environmental exposure.". Env Health Perspect (National Institute of Environmental Health Sciences) 8: 201–213. doi:10.2307/3428200. PMID 4470919.
- ↑ McFadzean, Belinda (December 2007). The Kinetics and Associated Equilibra of High Oxidation State Osmium Complexes (PDF) (PhD thesis). Port Elizabeth, South Africa: Nelson Mandela Metropolitan University. Retrieved 3 July 2025.
 |
