Chemistry:Raltegravir
Raltegravir, sold under the brand name Isentress, is an antiretroviral medication used, together with other medication, to treat HIV/AIDS.[1] It may also be used, as part of post-exposure prophylaxis, to prevent HIV infection following potential exposure.[2] It is taken by mouth.[1]
Common side effects include trouble sleeping, feeling tired, nausea, high blood sugar, and headaches.[2] Severe side effects may include allergic reactions including Stevens–Johnson syndrome, muscle breakdown, and liver problems.[2] It is unclear if use during pregnancy or breastfeeding is safe.[2] Raltegravir is an HIV integrase strand transfer inhibitor which blocks the functioning of HIV integrase which is needed for viral replication.[2]
Raltegravir was approved for medical use in the United States in 2007.[2] It is on the World Health Organization's List of Essential Medicines.[3] Lamivudine/raltegravir, a combination with lamivudine, is also available.[2]
Medical uses

Raltegravir was initially approved only for use in individuals whose infection has proven resistant to other HAART drugs.[4] However, in July 2009, the U.S. Food and Drug Administration (FDA) granted expanded approval for raltegravir for use in all patients.[5] As with any HAART medication, raltegravir is unlikely to show durability if used as monotherapy, due to the highly mutagenic nature of HIV.
In December 2011, it approval for use in children over the age of two, taken in pill form orally twice a day by prescription with two other antiretroviral medications to form the cocktail (most anti-HIV drugs regimens for adults and children use these cocktails).[citation needed] Raltegravir is available in chewable form, but because the two tablet formulations are not interchangeable, the chewable pills are only approved for use in children two to 11.[citation needed] Older adolescents will use the adult formulation.[6]
Efficacy
In a study of the drug as part of combination therapy, raltegravir exhibited potent and durable antiretroviral activity similar to that of efavirenz at 24 and 48 weeks but achieved HIV-1 RNA levels below detection at a more rapid rate. After 24 and 48 weeks of treatment, raltegravir did not result in increased serum levels of total cholesterol, low-density lipoprotein cholesterol, or triglycerides.[7][8]
Side effects
Raltegravir was generally well tolerated when used in combination with optimized background therapy regimens in treatment-experienced patients with HIV-1 infection in trials of up to 48 weeks' duration.[9]
Mechanism of action
As an integrase inhibitor, raltegravir targets integrase, an enzyme common to retroviruses that integrates the viral genetic material into human chromosomes, a critical step in the HIV infection model. The drug is metabolized away via glucuronidation.[10]
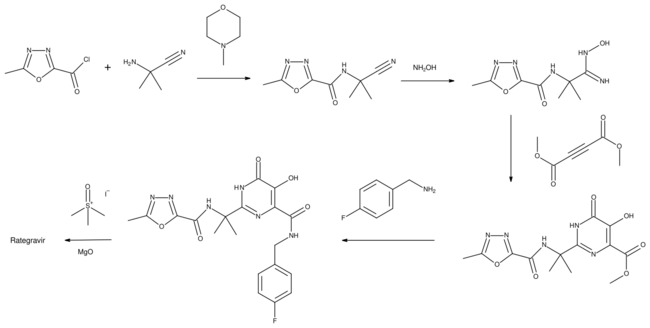
Chemistry
Raltegravir has been synthesized in several ways, which have been reviewed.[11][12]
In one method used for its manufacture, 2-amino-2-methylpropanenitrile is reacted with the acid chloride of 5-methyl-1,3,4-oxadiazole-2-carboxylic acid using N-methylmorpholine as base. The product is treated with aqueous hydroxylamine to form an amidoxime. The central pyrimidone ring of the drug is then created when the amidoxime reacts with dimethyl acetylenedicarboxylate. The synthesis is completed by conversion of the remaining methyl ester of the intermediate to an amide with 4-fluorobenzylamine, followed by methylation using trimethylsulfoxonium iodide. Use of that reagent ensures the required chemoselectivity so that methylation occurs on the nitrogen atom of the pyrimidone.[13]
History
Raltegravir was the first integrase inhibitor to receive approval in the United States in October 2007.[14][4][15] It was developed by Merck and reported by Summa et al. in the Journal of Medicinal Chemistry.[16]
In July 2024, the UK's Medicines and Healthcare products Regulatory Agency authorised the first generic raltegravir medicines for adult and paediatric patients with HIV who weigh at least 40kg. Authorisation was granted to Lupin Healthcare (UK) Limited and Zentiva Pharma UK Limited.[17]
Research
Raltegravir significantly alters HIV viral dynamics and decay and further research in this area is ongoing. In clinical trials patients taking raltegravir achieved viral loads less than 50 copies per millitre sooner than those taking similarly potent non-nucleoside reverse transcriptase inhibitors or protease inhibitors. This statistically significant difference in viral load reduction has caused some HIV researchers to begin questioning long held paradigms about HIV viral dynamics and decay.[18] Research into raltegravir's ability to affect latent viral reservoirs and possibly aid in the eradication of HIV is currently ongoing.[19]
Research results were published in July 2008. The authors concluded that "raltegravir plus optimized background therapy provided better viral suppression than optimized background therapy alone for at least 48 weeks."[20]
Research on human cytomegalovirus (HCMV) terminase proteins demonstrated that raltegravir may block viral replication of the herpesviruses.[21]
In January 2013, a phase II trial was initiated to evaluate the therapeutic benefit of raltegravir in treating multiple sclerosis (MS).[22] The drug is active against Human Endogenous Retroviruses (HERVs) and possibly Epstein–Barr virus, which have been suggested in the pathogenesis of relapsing-remitting MS.[citation needed]
References
- ↑ 1.0 1.1 British national formulary : BNF 69 (69 ed.). British Medical Association. 2015. p. 429. ISBN 978-0-85711-156-2.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Raltegravir Potassium". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/raltegravir-potassium.html.
- ↑ World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 4.0 4.1 "Isentress Drug Approval Package". U.S. Food and Drug Administration (FDA). 22 February 2008. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2007/022145_Isentress.cfm.
- ↑ "UPDATE 2-FDA OKs widened use of Merck's Isentress HIV drug". Reuters. 10 July 2009. https://www.reuters.com/article/idUSN0948398720090710.
- ↑ "FDA Okays Raltegravir for Kids, Teens with HIV". 15 October 2024. http://www.everydayhealth.com/hiv-aids/1222/fda-okays-raltegravir-for-kids-teens-with-hiv.aspx?xid=aol_eh-hiv_6_20111219_&aolcat=HLT&icid=maing-grid7%7Cmain5%7Cdl10%7Csec3_lnk2%26pLid%3D122480.
- ↑ "Rapid and durable antiretroviral effect of the HIV-1 Integrase inhibitor raltegravir as part of combination therapy in treatment-naive patients with HIV-1 infection: results of a 48-week controlled study". Journal of Acquired Immune Deficiency Syndromes 46 (2): 125–133. October 2007. doi:10.1097/QAI.0b013e318157131c. PMID 17721395.
- ↑ "Researchers buoyed by novel HIV drugs: will expand drug arsenal against resistant virus". JAMA 297 (14): 1535–1536. April 2007. doi:10.1001/jama.297.14.1535. PMID 17426263.
- ↑ "Raltegravir: a review of its use in the management of HIV infection in treatment-experienced patients". Drugs 69 (8): 1059–1075. May 2009. doi:10.2165/00003495-200969080-00007. PMID 19496631.
- ↑ "HIV Antiretroviral Agents in Development". 30 March 2006. https://www.thebody.com/content/art1352.html.
- ↑ "Raltegravir (Isentress): The First-in-Class HIV-1 Integrase Inhibitor". Modern Drug Synthesis. 2010. pp. 1–15. doi:10.1002/9780470768594.ch1. ISBN 978-0-470-76859-4.
- ↑ "34: Antiviral Drugs". Synthesis of Best-Seller Drugs. 2016. pp. 719–720. doi:10.1016/B978-0-12-411492-0.00034-1. ISBN 978-0-12-411492-0.
- ↑ Gurjar MK, Sonawane SP, Maikap GS, Patil GD, Shinde SB, Patil PS Mehta SS, "Synthesis of raltegravir", WO patent 2013098854, published 2013-07-04, assigned to Emcure Pharmaceuticals Ltd
- ↑ "FDA approval of Isentress (raltegravir)". U.S. Food and Drug Administration (FDA). 25 June 2009. https://www.fda.gov/ForConsumers/ByAudience/ForPatientAdvocates/HIVandAIDSActivities/ucm124040.htm.
- ↑ "Molecular dynamics simulations and drug discovery". BMC Biology 9 (1). October 2011. doi:10.1186/1741-7007-9-71. PMID 22035460.
- ↑ "Discovery of raltegravir, a potent, selective orally bioavailable HIV-integrase inhibitor for the treatment of HIV-AIDS infection". Journal of Medicinal Chemistry 51 (18): 5843–5855. September 2008. doi:10.1021/jm800245z. PMID 18763751.
- ↑ "Generic HIV raltegravir medicines approved" (in en). https://www.europeanpharmaceuticalreview.com/news/231638/generic-hiv-raltegravir-medicines-approved/.
- ↑ "Faster Viral Decay With Raltegravir". 24 July 2007. https://www.thebodypro.com/article/faster-viral-decay-raltegravir.
- ↑ Clinical trial number NCT00554398 for "Impact of MK-0518 (Raltegravir) Intensification on HIV-1 Viral Latency in Patients With Previous Complete Viral Suppression" at ClinicalTrials.gov
- ↑ "Raltegravir with optimized background therapy for resistant HIV-1 infection". The New England Journal of Medicine 359 (4): 339–354. July 2008. doi:10.1056/NEJMoa0708975. PMID 18650512. https://www.arca.fiocruz.br/handle/icict/30576.
- ↑ "Drug against AIDS could be effective against herpesvirus". https://www.sciencedaily.com/releases/2010/09/100923081859.htm.
- ↑ Raltegravir (Isentress) Pilot Study in Relapsing Multiple Sclerosis - Full Text View - ClinicalTrials.gov. 24 May 2017. https://clinicaltrials.gov/ct2/show/NCT01767701.
External links
 |