Chemistry:Maraviroc
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /məˈrævɪrɒk/ mə-RAV-i-rok Selzentry: /sɛlˈzɛntri/ |
| Trade names | Selzentry, Celsentri |
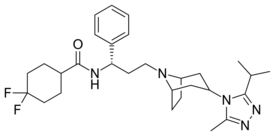
| Other names | UK-427857, 4,4-Difluoro-N-[(1S)-3-{(1R,3s,5S)-3-[3-methyl-5-(propan-2-yl)-4H-1,2,4-triazol-4-yl]-8-azabicyclo[3.2.1]octan-8-yl}-1-phenylpropyl] cyclohexanecarboxamide |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607076 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 23%[3] |
| Protein binding | ~76%[1] |
| Metabolism | Liver (CYP, predominantly CYP3A)[1] |
| Metabolites | Secondary amine formed by N-dealkylation (major) |
| Elimination half-life | 14–18 hours[1] (mean 16 hours)[4] |
| Excretion | Feces (76%), urine (20%)[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| Chemical and physical data | |
| Formula | C29H41F2N5O |
| Molar mass | 513.678 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Maraviroc, sold under the brand names Selzentry (US) and Celsentri (EU), is an antiretroviral medication used to treat HIV infection.[1][2] It is taken by mouth.[1][2] It is in the CCR5 receptor antagonist class.[1][2]
It was approved for medical use in the United States in August 2007,[1] and in the European Union in September 2007.[2]
Medical uses
Maraviroc is indicated, in combination with other antiretroviral medications, for the treatment of only CCR5-tropic HIV-1 infection.[1][2]
Side effects
Maraviroc can cause serious, life-threatening side effects. These include liver problems, skin reactions, and allergic reactions. An allergic reaction may happen before liver problems occur.[5] Official labeling of Selzentry has black box warning for hepatotoxicity.[1] The MOTIVATE trials showed no clinically relevant differences in safety between the maraviroc and placebo groups.[6]
Mechanism of action
Maraviroc is an entry inhibitor. Specifically, maraviroc is a negative allosteric modulator of the CCR5 receptor, which is found on the surface of certain human cells. The chemokine receptor CCR5 is an essential co-receptor for most HIV strains and necessary for the entry process of the virus into the host cell. The drug binds to CCR5, thereby blocking the HIV protein gp120 from associating with the receptor. HIV is then unable to enter human macrophages and T cells.[7] Because HIV can also use other coreceptors, such as CXCR4, an HIV tropism test such as a trofile assay must be performed to determine if the drug will be effective.[8]
History
Maraviroc, originally designated UK-427857, was developed by the drug company Pfizer in its UK labs located in Sandwich. On 24 April 2007 the U.S. Food and Drug Administration advisory panel reviewing maraviroc's New Drug Application unanimously recommended approval for the new drug,[9] and the drug received full FDA approval on 6 August 2007 for use in treatment experienced patients.[10]
Two randomized, placebo-controlled clinical trials, compared 209 people receiving optimized therapy plus a placebo to 426 people receiving optimized therapy plus 150 mg maraviroc once daily and 414 patients receiving optimized therapy plus 150 mg maraviroc twice daily. At 48 weeks, 55% of participants receiving maraviroc once daily and 60% of participants receiving the drug twice daily achieved a viral load of less than 400 copies/mL compared with 26% of those taking placebo; about 44% of the once-daily and 45% of the twice-daily maraviroc group had a viral load of less than 50 copies/mL compared with about 23% of those who received placebo. In addition, those who received the entry inhibitor had a mean increase in CD4+ cells of 110 cells/μL in the once-daily group, 106 cells/μL in the twice-daily group, and 56 cells/μL in the placebo group.[6][11][12] Maraviroc was approved for medical use in the European Union in September 2007.[2]
Names
Maraviroc is the International nonproprietary name (INN).[13]
Research
Maraviroc appears to reduce graft-versus-host disease in people treated with allogeneic bone marrow transplantation for leukemia, in a Phase I/II study.[14][15]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Selzentry- maraviroc tablet, film coated Selzentry- maraviroc solution". 18 July 2018. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=46f30ac5-c96b-429e-976d-8c5ee1c0761b.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Celsentri EPAR". 17 September 2018. https://www.ema.europa.eu/en/medicines/human/EPAR/celsentri.
- ↑ "Assessment of the absorption, metabolism and absolute bioavailability of maraviroc in healthy male subjects". British Journal of Clinical Pharmacology 65 (Suppl 1): 60–7. April 2008. doi:10.1111/j.1365-2125.2008.03137.x. PMID 18333867.
- ↑ "Maraviroc: pharmacokinetics and drug interactions". Antiviral Therapy 14 (5): 607–18. 2009. doi:10.1177/135965350901400514. PMID 19704163.
- ↑ "Maraviroc (HIV treatment) Dosage, Side Effects". https://aidsinfo.nih.gov/drugs/408/maraviroc--hiv-treatment/0/patient.
- ↑ 6.0 6.1 Stephenson J (April 2007). "Researchers buoyed by novel HIV drugs: will expand drug arsenal against resistant virus". JAMA 297 (14): 1535–6. doi:10.1001/jama.297.14.1535. PMID 17426263.
- ↑ Levy JA (January 2009). "HIV pathogenesis: 25 years of progress and persistent challenges". AIDS 23 (2): 147–60. doi:10.1097/QAD.0b013e3283217f9f. PMID 19098484.
- ↑ "Access denied? The status of co-receptor inhibition to counter HIV entry". Expert Opinion on Pharmacotherapy 8 (7): 923–33. May 2007. doi:10.1517/14656566.8.7.923. PMID 17472538.
- ↑ Gay News From 365Gay.com
- ↑ Krauskopf, Lewis (6 August 2007). "Pfizer wins U.S. approval for new HIV drug". Reuters. https://www.reuters.com/article/ousiv/idUSN0642522320070806.
- ↑ "CCR5 antagonists: comparison of efficacy, side effects, pharmacokinetics and interactions--review of the literature". European Journal of Medical Research 12 (9): 409–17. October 2007. PMID 17933722.
- ↑ "Maraviroc reduces viral load in naive patients at 48 weeks". AIDS Patient Care and STDs 21 (9): 703–4. September 2007. PMID 17941136.
- ↑ "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 53". WHO Drug Information 19 (1): 84–5. 2005. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Blockade of lymphocyte chemotaxis in visceral graft-versus-host disease". N. Engl. J. Med. 367 (2): 135–45. July 2012. doi:10.1056/NEJMoa1201248. PMID 22784116.
- ↑ "HIV Drug Reduces Graft-versus-Host Disease in Bone Marrow Transplant Patients, Penn Study Shows". Penn Medicine (Press release).
Further reading
- "Maraviroc Therapy and CCR5 Genotype". Medical Genetics Summaries. National Center for Biotechnology Information (NCBI). 2015. Bookshelf ID: NBK279895. https://www.ncbi.nlm.nih.gov/books/NBK279895/.
External links
- "Maraviroc". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/maraviroc.
- Maraviroc at the US National Library of Medicine Medical Subject Headings (MeSH)
 |
