Chemistry:Triphosphorus pentanitride
| |
| Names | |
|---|---|
| IUPAC name
Triphosphorus pentanitride
| |
| Other names
Phosphorus(V) nitride, Phosphorus nitride
| |
| Identifiers | |
3D model (JSmol)
|
|
| EC Number |
|
PubChem CID
|
|
| |
| Properties | |
| P 3N 5 | |
| Molar mass | 162.955 g/mol |
| Appearance | White solid |
| Density | 2.77 g/cm3 (α-P 3N 5) |
| Melting point | 850 °C (1,560 °F; 1,120 K) decomposes |
| insoluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Triphosphorus pentanitride is an inorganic compound with the chemical formula P
3N
5. Containing only phosphorus and nitrogen, this material is classified as a binary nitride. While it has been investigated for various applications this has not led to any significant industrial uses. It is a white solid, although samples often appear colored owing to impurities.
Synthesis
Triphosphorus pentanitride can be produced by reactions between various phosphorus(V) and nitrogen anions (such as ammonia and sodium azide):[1]
- 3 PCl
5 + 5 NH
3 → P
3N
5 + 15 HCl - 3 PCl
5 + 15 NaN
3 → P
3N
5 + 15 NaCl + 20 N
2
The reaction of the elements is claimed to produce a related material.[2] Similar methods are used to prepared boron nitride (BN) and silicon nitride (Si
3N
4); however the products are generally impure and amorphous.[1][3]
Crystalline samples have been produced by the reaction of ammonium chloride and hexachlorocyclotriphosphazene[4] or phosphorus pentachloride.[1]
- (NPCl
2)
3 + 2 [NH
4]Cl → P
3N
5 + 8 HCl - 3 PCl
5 + 5 [NH
4]Cl → P
3N
5 + 20 HCl
P
3N
5 has also been prepared at room temperature, by a reaction between phosphorus trichloride and sodium amide.[5]
- 3 PCl
3 + 5 NaNH
2 → P
3N
5 + 5 NaCl + 4 HCl + 3 H
2
Reactions
P
3N
5 is thermally less stable than either BN or Si
3N
4, with decomposition to the elements occurring at temperatures above 850 °C:[1]
- P
3N
5 → 3 PN + N
2 - 4 PN → P
4 + 2 N
2
It is resistant to weak acids and bases, and insoluble in water at room temperature, however it hydrolyzes upon heating to form the ammonium phosphate salts [NH
4]
2HPO
4 and [NH
4]H
2PO
4.
Triphosphorus pentanitride reacts with lithium nitride and calcium nitride to form the corresponding salts of PN7−
4 and PN4−
3. Heterogenous ammonolyses of triphosphorus pentanitride gives imides such as HPN
2 and HP
4N
7. It has been suggested that these compounds may have applications as solid electrolytes and pigments.[6]
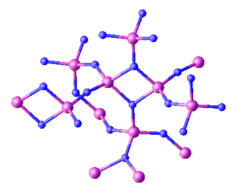
Structure and properties
Several polymorphs are known for triphosphorus pentanitride. The alpha‑form of triphosphorus pentanitride (α‑P
3N
5) is encountered at atmospheric pressure and exists at pressures up to 11 GPa, at which point it converts to the gamma‑variety (γ‑P
3N
5) of the compound.[7][8] Upon heating γ‑P
3N
5 to temperatures above 2000 K at pressures between 67 and 70 GPa, it transforms into δ-P
3N
5.[9] The release of pressure on the δ-P
3N
5 polymorph does not revert it back into γ‑P
3N
5 or α‑P
3N
5. Instead, at pressures below 7 GPa, δ-P
3N
5 converts into a fourth form of triphosphorus pentanitride, α′‑P
3N
5.[9]
| Polymorph | Density (g/cm3) |
|---|---|
| α‑P 3N 5 |
2.77 |
| α′‑P 3N 5 |
3.11 |
| γ‑P 3N 5 |
3.65 |
| δ‑P 3N 5 |
5.27 (at 72 GPa) |
The structure of all polymorphs of triphosphorus pentanitride was determined by single crystal X-ray diffraction. α‑P
3N
5 and α′‑P
3N
5 are formed of a network structure of PN
4 tetrahedra with 2- and 3-coordinated nitrides,[7][9] γ‑P
3N
5 is composed of both PN
4 and PN
5 polyhedra[8] while δ-P
3N
5 is composed exclusively of corner- and edge-sharing PN
6 octahedra.[9] δ-P
3N
5 is the most incompressible triphosphorus pentanitride, having a bulk modulus of 313 GPa.[9]
Potential applications
Triphosphorus pentanitride has no commercial applications, although it found use as a gettering material for incandescent lamps, replacing various mixtures containing red phosphorus in the late 1960s. The lighting filaments are dipped into a suspension of P
3N
5 prior to being sealed into the bulb. After bulb closure, but while still on the pump, the lamps are lit, causing the P
3N
5 to thermally decompose into its constituent elements. Much of this is removed by the pump but enough P
4 vapor remains to react with any residual oxygen inside the bulb. Once the vapor pressure of P
4 is low enough, either filler gas is admitted to the bulb prior to sealing off or, if a vacuum atmosphere is desired, the bulb is sealed off at that point. The high decomposition temperature of P
3N
5 allows sealing machines to run faster and hotter than was possible using red phosphorus.
Related halogen containing polymers, trimeric bromophosphonitrile (PNBr
2)
3 (melting point 192 °C) and tetrameric bromophosphonitrile (PNBr
2)
4 (melting point 202 °C) find similar lamp gettering applications for tungsten halogen lamps, where they perform the dual processies of gettering and precise halogen dosing.[10]
Triphosphorus pentanitride has also been investigated as a semiconductor for applications in microelectronics, particularly as a gate insulator in metal-insulator-semiconductor devices.[11][12]
As a fuel in pyrotechnic obscurant mixtures, it offers some benefits over the more commonly used red phosphorus, owing mainly to its higher chemical stability. Unlike red phosphorus, P
3N
5 can be safely mixed with strong oxidizers, even potassium chlorate. While these mixtures can burn up to 200 times faster than state-of-the-art red phosphorus mixtures, they are far less sensitive to shock and friction. Additionally, P
3N
5 is much more resistant to hydrolysis than red phosphorus, giving pyrotechnic mixtures based on it greater stability under long-term storage.[13]
Patents have been filed for the use of triphosphorus pentanitride in fire fighting measures.[14][15]
See also
References
- ↑ 1.0 1.1 1.2 1.3 Schnick, Wolfgang (1 June 1993). "Solid-State Chemistry with Nonmetal Nitrides". Angewandte Chemie International Edition in English 32 (6): 806–818. doi:10.1002/anie.199308061. https://epub.ub.uni-muenchen.de/3954/1/3954.pdf.
- ↑ Vepřek, S.; Iqbal, Z.; Brunner, J.; Schärli, M. (1 March 1981). "Preparation and properties of amorphous phosphorus nitride prepared in a low-pressure plasma". Philosophical Magazine B 43 (3): 527–547. doi:10.1080/01418638108222114. Bibcode: 1981PMagB..43..527V.
- ↑ Meng, Zhaoyu; Peng, Yiya; Yang, Zhiping; Qian, Yitai (1 January 2000). "Synthesis and Characterization of Amorphous Phosphorus Nitride.". Chemistry Letters 29 (11): 1252–1253. doi:10.1246/cl.2000.1252.
- ↑ Schnick, Wolfgang; Lücke, Jan; Krumeich, Frank (1996). "Phosphorus Nitride P3N5: Synthesis, Spectroscopic, and Electron Microscopic Investigations". Chemistry of Materials 8: 281–286. doi:10.1021/cm950385y.
- ↑ Chen, Luyang; Gu, Yunle; Shi, Liang; Yang, Zeheng; Ma, Jianhua; Qian, Yitai (2004). "Room temperature route to phosphorus nitride hollow spheres". Inorganic Chemistry Communications 7 (5): 643. doi:10.1016/j.inoche.2004.03.009.
- ↑ Schnick, Wolfgang (1993). "Phosphorus(V) Nitrides: Preparation, Properties, and Possible Applications of New Solid State Materials with Structural Analogies to Phosphates and Silicates". Phosphorus, Sulfur, and Silicon and the Related Elements 76 (1–4): 183–186. doi:10.1080/10426509308032389.
- ↑ 7.0 7.1 Horstmann, Stefan; Irran, Elisabeth; Schnick, Wolfgang (1997). "Synthesis and Crystal Structure of Phosphorus(V) Nitrideα-P3N5". Angewandte Chemie International Edition in English 36 (17): 1873–1875. doi:10.1002/anie.199718731.
- ↑ 8.0 8.1 Landskron, Kai; Huppertz, Hubert; Senker, Jürgen; Schnick, Wolfgang (2001). "High-Pressure Synthesis of γ-P3N5 at 11 GPa and 1500 °C in a Multianvil Assembly: A Binary Phosphorus(V) Nitride with a Three-Dimensional Network Structure from PN4 Tetrahedra and Tetragonal PN5 Pyramids". Angewandte Chemie 40 (14): 2643–2645. doi:10.1002/1521-3773(20010716)40:14<2643::AID-ANIE2643>3.0.CO;2-T. https://onlinelibrary.wiley.com/doi/10.1002/1521-3773(20010716)40:14%3C2643::AID-ANIE2643%3E3.0.CO;2-T.
- ↑ 9.0 9.1 9.2 9.3 9.4 Laniel, Dominique; Trybel, Florian; Néri, Adrien; Yin, Yuqing; Aslandukov, Andrey; Fedotenko, Timofey; Khandarkhaeva, Saiana; Tasnádi, Ferenc et al. (2022-11-07). "Revealing Phosphorus Nitrides up to the Megabar Regime: Synthesis of α′‐P 3 N 5, δ‐P 3 N 5 and PN 2" (in en). Chemistry – A European Journal 28 (62): e202201998. doi:10.1002/chem.202201998. ISSN 0947-6539. PMID 35997073.
- ↑ S.T. Henderson and A.M. Marsden, Lamps and Lighting 2nd Ed., Edward Arnlold Press, 1975, ISBN:0 7131 3267 1
- ↑ Hirota, Yukihiro (1982). "Chemical vapor deposition and characterization of phosphorus nitride (P3N5) gate insulators for InP metal-insulator-semiconductor devices". Journal of Applied Physics 53 (7): 5037–5043. doi:10.1063/1.331380. Bibcode: 1982JAP....53.5037H.
- ↑ Jeong, Yoon-Ha; Choi, Ki-Hwan; Jo, Seong-Kue; Kang, Bongkoo (1995). "Effects of Sulfide Passivation on the Performance of GaAs MISFETs with Photo-CVD Grown P3N5 Gate Insulators". Japanese Journal of Applied Physics 34 (Part 1, No. 2B): 1176–1180. doi:10.1143/JJAP.34.1176. Bibcode: 1995JaJAP..34.1176J.
- ↑ Koch, Ernst-Christian; Cudziło, Stanisław (2016), "Safer Pyrotechnic Obscurants Based on Phosphorus(V) Nitride", Angewandte Chemie International Edition 55 (49): 15439–15442, doi:10.1002/anie.201609532, PMID 27862760
- ↑ Phosphorus nitride agents to protect against fires and explosions, http://patentscope.wipo.int/search/en/WO1996010443
- ↑ Manufacture of flame-retardant regenerated cellulose fibres, December 20, 1977, http://www.patentgenius.com/patent/4063883.html
| NH3 | He(N2)11 | ||||||||||||||||
| Li3N | Be3N2 | BN | β-C3N4 g-C3N4 |
N2 | NxOy | NF3 | Ne | ||||||||||
| Na3N | Mg3N2 | AlN | Si3N4 | PN P3N5 |
SxNy SN S4N4 |
NCl3 | Ar | ||||||||||
| K3N | Ca3N2 | ScN | TiN | VN | CrN Cr2N |
MnxNy | FexNy | CoN | Ni3N | CuN | Zn3N2 | GaN | Ge3N4 | As | Se | NBr3 | Kr |
| Rb3N | Sr3N2 | YN | ZrN | NbN | β-Mo2N | Tc | Ru | Rh | PdN | Ag3N | CdN | InN | Sn | Sb | Te | NI3 | Xe |
| Cs3N | Ba3N2 | Hf3N4 | TaN | WN | Re | Os | Ir | Pt | Au | Hg3N2 | TlN | Pb | BiN | Po | At | Rn | |
| Fr3N | Ra3N | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | CeN | Pr | Nd | Pm | Sm | Eu | GdN | Tb | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | Th | Pa | UN | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||
 |

