Chemistry:Β-Methylamino-L-alanine
| |
| Names | |
|---|---|
| IUPAC name
3-(Methylamino)-L-alanine
| |
| Systematic IUPAC name
(2S)-2-Amino-3-(methylamino)propanoic acid[1] | |
| Other names
2-Amino-3-methylaminopropanoic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
| MeSH | alpha-amino-beta-methylaminopropionate |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C4H10N2O2 | |
| Molar mass | 118.136 g·mol−1 |
| log P | −0.1 |
| Acidity (pKa) | 1.883 |
| Basicity (pKb) | 12.114 |
| Related compounds | |
Related alkanoic acids
|
|
Related compounds
|
Dimethylacetamide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
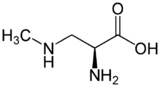
β-Methylamino-L-alanine, or BMAA, is a non-proteinogenic amino acid produced by cyanobacteria. BMAA is a neurotoxin. Its potential role in various neurodegenerative disorders is the subject of scientific research.
Structure and properties
BMAA is a derivative of the amino acid alanine with a methylamino group on the side chain. This non-proteinogenic amino acid is classified as a polar base.
Sources and detection
BMAA is produced by cyanobacteria in marine, freshwater, and terrestrial environments.[2][3] In cultured non-nitrogen-fixing cyanobacteria, BMAA production increases in a nitrogen-depleted medium.[4] The biosynthetic pathway in cyanobacteria is unknown, but involvement of BMAA and its structural analog 2,4-diaminobutanoic acid (2,4-DAB) in environmental iron scavenging has been hypothesized.[5][6] BMAA has been found in aquatic organisms and in plants with cyanobacterial symbionts such as certain lichens, the floating fern Azolla, the leaf petioles of the tropical flowering plant Gunnera, cycads as well as in animals that eat the fleshy covering of cycad seeds, including flying foxes.[7][8][9][10]
High concentrations of BMAA are present in shark fins.[11] Because BMAA is a neurotoxin, consumption of shark fin soup and cartilage pills therefore may pose a health risk.[12] The toxin can be detected via several laboratory methods, including liquid chromatography, high-performance liquid chromatography, mass spectrometry, amino acid analyzer, capillary electrophoresis, and NMR spectroscopy.[13]
Neurotoxicity
BMAA can cross the blood–brain barrier in rats. It takes longer to get into the brain than into other organs, but once there, it is trapped in proteins, forming a reservoir for slow release over time.[14][15]
Mechanisms
Although the mechanisms by which BMAA causes motor neuron dysfunction and death are not entirely understood, current research suggests that there are multiple mechanisms of action. Acutely, BMAA can act as an excitotoxin on glutamate receptors, such as NMDA, calcium-dependent AMPA, and kainate receptors.[16][17] The activation of the metabotropic glutamate receptor 5 is believed to induce oxidative stress in the neuron by depletion of glutathione.[18]
BMAA can be misincorporated into nascent proteins in place of L-serine, possibly causing protein misfolding and aggregation, both hallmarks of tangle diseases, including Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis (ALS), progressive supranuclear palsy (PSP), and Lewy body disease. In vitro research has shown that protein association of BMAA may be inhibited in the presence of excess L-serine.[19]
Effects
A study performed in 2015 with vervet monkeys (Chlorocebus sabaeus) in St. Kitts, which are homozygous for the apoE4 gene (a condition which in humans is a risk factor for Alzheimer's disease), found that vervets that were administered BMAA orally developed hallmark histopathology features of Alzheimer's disease, including amyloid beta plaques and neurofibrillary tangle accumulation. Vervets in the trial fed smaller doses of BMAA were found to have correlative decreases in these pathology features. Additionally, vervets that were co-administered BMAA with serine were found to have 70% less beta-amyloid plaques and neurofibrillary tangles than those administered BMAA alone, suggesting that serine may be protective against the neurotoxic effects of BMAA.
This experiment represents the first in-vivo model of Alzheimer's disease that features both beta-amyloid plaques and hyperphosphorylated tau protein. This study also demonstrates that BMAA, an environmental toxin, can trigger neurodegenerative disease as a result of a gene-environment interaction.[20]
Degenerative locomotor diseases have been described in animals grazing on cycad species, fueling interest in a possible link between the plant and the etiology of ALS/PDC. Subsequent laboratory investigations discovered the presence of BMAA. BMAA induced severe neurotoxicity in rhesus macaques, including:[21]
- limb muscle atrophy
- nonreactive degeneration of anterior horn cells
- degeneration and partial loss of pyramidal neurons of the motor cortex
- behavioral dysfunction
- conduction deficits in the central motor pathway
- neuropathological changes of motor cortex Betz cells
There are reports that low BMAA concentrations can selectively kill cultured motor neurons from mouse spinal cords and produce reactive oxygen species.[17][22]
Scientists have also found that newborn rats treated with BMAA show a progressive neurodegeneration in the hippocampus, including intracellular fibrillar inclusions, and impaired learning and memory as adults.[23][24][25] BMAA has been reported to be excreted into rodent breast milk, and subsequently transferred to the suckling offspring, suggesting mothers' and cows' milk might be other possible exposure routes.[26]
Human cases
This section is missing information about dosage exposed to -- to help contextualize risk. (September 2023) |
Chronic dietary exposure to BMAA is now considered to be a cause of the amyotrophic lateral sclerosis/parkinsonism–dementia complex (ALS/PDC) that had an extremely high rate of incidence among the Chamorro people of Guam.[27] The Chamorro call the condition lytico-bodig.[28] In the 1950s, ALS/PDC prevalence ratios and death rates for Chamorro residents of Guam and Rota were 50–100 times that of developed countries, including the United States.[28] No demonstrable heritable or viral factors were found for the disease, and a subsequent decline of ALS/PDC after 1963 on Guam led to the search for responsible environmental agents.[29] The use of flour made from cycad seed (Cycas micronesica[30]) in traditional food items decreased as that plant became rarer and the Chamorro population became more Americanized following World War II.[31] Cycads harbor symbiotic cyanobacteria of the genus Nostoc in specialized roots which push up through the leaf litter into the light; these cyanobacteria produce BMAA.[32]
In addition to eating traditional food items from cycad flour directly, BMAA may be ingested by humans through biomagnification. Flying foxes, a Chamorro delicacy, forage on the fleshy seed covering of cycad seeds and concentrate the toxin in their bodies. Twenty-four specimens of flying foxes from museum collections were tested for BMAA, which was found in large concentrations in the flying foxes from Guam.[33] As of 2021 studies continued examining BMAA biomagnification in marine and estuarine systems and its possible impact on human health outside of Guam.[34]
Studies on human brain tissue of ALS/PDC, ALS, Alzheimer's disease, Parkinson's disease, Huntington's disease, and neurological controls indicated that BMAA is present in non-genetic progressive neurodegenerative disease, but not in controls or genetic-based Huntington's disease.[35][36][37][38]
(As of 2021) research into the role of BMAA as an environmental factor in neurodegenerative disease continued.[39][40][41]
Clinical trials
Safe and effective ways of treating ALS patients with L-serine that has been found to protect non-human primates from BMAA-induced neurodegeneration, have been goals of clinical trials conducted by the Phoenix Neurological Associates and the Forbes/Norris ALS/MND clinic and sponsored by the Institute for Ethnomedicine.[42][43]
See also
- Oxalyldiaminopropionic acid, a related toxin
References
- ↑ "alpha-amino-beta-methylaminopropionate - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 19 August 2005. Identification. https://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=105089.
- ↑ "Diverse taxa of cyanobacteria produce b-N-methylamino-L-alanine, a neurotoxic amino acid". PNAS 102 (14): 5074–5078. 2005. doi:10.1073/pnas.0501526102. PMID 15809446. Bibcode: 2005PNAS..102.5074C.
- ↑ "β-N-methylamino-L-alanine (BMAA) in novel South African cyanobacterial isolates". Ecotoxicology and Environmental Safety 71 (2): 309–313. 2008. doi:10.1016/j.ecoenv.2008.04.010. PMID 18538391.
- ↑ "Nitrogen starvation of cyanobacteria results in the production of β-N-methylamino-L-alanine". Toxicon 58 (2): 187–194. 2011. doi:10.1016/j.toxicon.2011.05.017. PMID 21704054.
- ↑ "Genomic insights into the biosynthesis and physiology of the cyanobacterial neurotoxin 3-N-methyl-2,3-diaminopropanoic acid (BMAA)". Phytochemistry 200: 113198. 2022. doi:10.1016/j.phytochem.2022.113198. PMID 35447107. Bibcode: 2022PChem.200k3198M.
- ↑ "Genomic insights into the biosynthesis and physiology of the cyanobacterial neurotoxin 2,4-diaminobutanoic acid (2,4-DAB)". Phytochemistry 192: 112953. 2021. doi:10.1016/j.phytochem.2021.112953. PMID 34598041. Bibcode: 2021PChem.192k2953M. https://www.research.ed.ac.uk/en/publications/89c39093-6095-4158-b2b2-0eb895b79ee5.
- ↑ Vega, A; Bell, A. (1967). "a-amino-β-methylaminopropionic acid, a new amino acid from seeds of cycas circinalis". Phytochemistry 6 (5): 759–762. doi:10.1016/s0031-9422(00)86018-5.
- ↑ Banack, SA; Cox, PA (2003). "Biomagnification of cycad neurotoxins in flying foxes: implications for ALS-PDC in Guam". Neurology 61 (3): 387–9. doi:10.1212/01.wnl.0000078320.18564.9f. PMID 12913204.
- ↑ "Dietary BMAA exposure in an amyotrophic lateral sclerosis cluster from Southern France". PLOS ONE 8 (12): e83406. 2013. doi:10.1371/journal.pone.0083406. PMID 24349504. Bibcode: 2013PLoSO...883406M.
- ↑ "Linking β-methylamino-L-alanine exposure to sporadic amyotrophic lateral sclerosis in Annapolis, MD". Toxicon 70: 179–183. 2013. doi:10.1016/j.toxicon.2013.04.010. PMID 23660330.
- ↑ Kiyo Mondo; Neil Hammerschlag; Margaret Basile; John Pablo; Sandra A. Banack; Deborah C. Mash (2012). "Cyanobacterial Neurotoxin β-N-Methylamino-L-alanine (BMAA) in Shark Fins". Marine Drugs 10 (2): 509–520. doi:10.3390/md10020509. PMID 22412816.
- ↑ "Neurotoxins in shark fins: A human health concern". Science Daily. February 23, 2012. https://www.sciencedaily.com/releases/2012/02/120223182516.htm.
- ↑ Cohen, S.A. (2012). "Analytical techniques for the detection of α-amino-β-methylaminopropionic acid". Analyst 137 (9): 1991–2005. doi:10.1039/c2an16250d. PMID 22421821. Bibcode: 2012Ana...137.1991C.
- ↑ Mash D, et al. Neurotoxic non-protein amino acid BMAA in brain from patients dying with ALS and Alzheimer's disease[yes|permanent dead link|dead link}}] poster presented at: American Academy of Neurology Annual Meeting, Chicago, IL, 17 April 2008 Neurology 2008;70(suppl 1):A329.
- ↑ Xie X, et al. Tracking brain uptake and protein incorporation of cyanobacterial toxin BMAA abstract presented at: 22nd Annual Symposium on ALS/MND, Sydney, Australia, 1 December 2011.
- ↑ "Neurotoxicity of β -N-methylamino-L-alanine (BMAA) and β-N-oxalylamino-L-alanine (BOAA) on cultured cortical neurons". Brain Research 497 (1): 64–71. 1989. doi:10.1016/0006-8993(89)90970-0. PMID 2551452.
- ↑ 17.0 17.1 "β-N-methylamino-L-alanine enhances neurotoxicity through multiple mechanisms". Neurobiology of Disease 25 (2): 360–366. 2007. doi:10.1016/j.nbd.2006.10.002. PMID 17098435.
- ↑ "Synergistic toxicity of the environmental neurotoxins methylmercury and β-N-methylamino-L-alanine". NeuroReport 23 (4): 216–219. 2012. doi:10.1097/WNR.0b013e32834fe6d6. PMID 22314682.
- ↑ "The Non-Protein Amino Acid BMAA Is Misincorporated into Human Proteins in Place of l-Serine Causing Protein Misfolding and Aggregation". PLOS ONE 8 (9): e75376. 2013. doi:10.1371/journal.pone.0075376. PMID 24086518. Bibcode: 2013PLoSO...875376D.
- ↑ "Dietary exposure to an environmental toxin triggers neurofibrillary tangles and amyloid deposits in the brain". Proceedings of the Royal Society B 283 (1823): 20152397. 2015. doi:10.1098/rspb.2015.2397. PMID 26791617.
- ↑ Spencer, Peter S.; Hugon, J.; Ludolph, A.; Nunn, P. B.; Ross, S. M.; Roy, D. N.; Schaumburg, H. H. (28 September 2007). "14: Discovery and Partial Characterization of Primate Motor-System Toxins". Ciba Foundation Symposium 126 ‐ Selective Neuronal Death. Novartis Foundation Symposia. 126. Wiley Online Library. pp. 221–238. doi:10.1002/9780470513422.ch14. ISBN 978-0-470-51342-2.
- ↑ "BMAA selectively injures motor neurons via AMPA/kainate receptor activations". Experimental Neurology 201 (1): 244–52. 2006. doi:10.1016/j.expneurol.2006.04.017. PMID 16764863.
- ↑ Karlsson, Oskar; Berg, Anna-Lena; Hanrieder, Jörg; Arnerup, Gunnel; Lindström, Anna-Karin; Brittebo, Eva B. (2014). "Intracellular fibril formation, calcification, and enrichment of chaperones, cytoskeletal, and intermediate filament proteins in the adult hippocampus CA1 following neonatal exposure to the nonprotein amino acid BMAA". Archives of Toxicology 89 (3): 423–436. doi:10.1007/s00204-014-1262-2. ISSN 0340-5761. PMID 24798087.
- ↑ Karlsson, O.; Roman, E.; Brittebo, E. B. (2009). "Long-term Cognitive Impairments in Adult Rats Treated Neonatally with -N-Methylamino-L-Alanine". Toxicological Sciences 112 (1): 185–195. doi:10.1093/toxsci/kfp196. ISSN 1096-6080. PMID 19692667.
- ↑ Karlsson, O. (2011). Distribution and Long-term Effects of the Environmental Neurotoxin β-N-methylamino-L-alanine (BMAA): Brain changes and behavioral impairments following developmental exposure. http://urn.kb.se/resolve?urn=urn:nbn:se:uu:diva-140785
- ↑ Andersson, Marie; Karlsson, Oskar; Bergström, Ulrika; Brittebo, Eva B.; Brandt, Ingvar (2013). "Maternal Transfer of the Cyanobacterial Neurotoxin β-N-Methylamino-L-Alanine (BMAA) via Milk to Suckling Offspring". PLOS ONE 8 (10): e78133. doi:10.1371/journal.pone.0078133. ISSN 1932-6203. PMID 24194910.
- ↑ "Cycad neurotoxins, consumption of flying foxes, and ALS-PDC disease in Guam". Neurology 58 (6): 956–9. 2002. doi:10.1212/wnl.58.6.956. PMID 11914415.
- ↑ 28.0 28.1 Kurland, LK; Mulder, DW (1954). "Epidemiologic investigations of amyotrophic lateral sclerosis". Neurology 4 (5): 355–78. doi:10.1212/wnl.4.5.355. PMID 13185376.
- ↑ "Clinical features and changing patterns of neurodegenerative disorders on Guam, 1997-2000". Neurology 58 (1): 90–7. 2002. doi:10.1212/wnl.58.1.90. PMID 11781411.
- ↑ Hill, K.D. (1994). "The cycas rumphii complex (Cycadeceae) in New Guinea and the Western Pacific". Australian Systematic Botany 7 (6): 543–567. doi:10.1071/sb9940543.
- ↑ Whiting, M.G. (1963). "Toxicity of cycads". Economic Botany 17 (4): 270–302. doi:10.1007/bf02860136.
- ↑ Rai, A.N.; Soderback, E.; Bergman, B. (2000), "Tansley Review No. 116 - Cyanobacterium-Plant Symbioses", The New Phytologist 147 (3): 449–481, doi:10.1046/j.1469-8137.2000.00720.x, PMID 33862930
- ↑ "Neurotoxic flying foxes as dietary items for the Chamorro people, Mariana Islands". Ethnopharmacology 106 (1): 97–104. 2006. doi:10.1016/j.jep.2005.12.032. PMID 16457975.
- ↑ Wang, Chao; Yan, Chen; Qiu, Jiangbing; Liu, Chao; Yan, Yeju; Ji, Ying; Wang, Guixiang; Chen, Hongju et al. (2021). "Food web biomagnification of the neurotoxin β-N-methylamino-L-alanine in a diatom-dominated marine ecosystem in China". Journal of Hazardous Materials 404 (Pt B): 124217. doi:10.1016/j.jhazmat.2020.124217. ISSN 0304-3894. PMID 33129020.
- ↑ "A mechanism for slow release of biomagnified cyanobacterial neurotoxins and neurodegenerative disease in Guam". PNAS 101 (33): 12228–12231. 2004. doi:10.1073/pnas.0404926101. PMID 15295100. Bibcode: 2004PNAS..10112228M.
- ↑ "Occurrence of b-methylamino-L-alanine (BMAA) in ALS/PDC patients from Guam". Acta Neurologica Scandinavica 110 (4): 267–9. 2004. doi:10.1111/j.1600-0404.2004.00320.x. PMID 15355492.
- ↑ "Cyanobacterial neurotoxin BMAA in ALS and Alzheimer's disease". Acta Neurologica Scandinavica 120 (4): 215–225. 2009. doi:10.1111/j.1600-0404.2008.01150.x. PMID 19254284.
- ↑ Bradley, WG; Mash, DC (2009). "Beyond Guam: the cyanobacterial/BMAA hypothesis of the cause of ALS and other neurodegenerative diseases". Amyotrophic Lateral Sclerosis 10: 7–20. doi:10.3109/17482960903286009. PMID 19929726.
- ↑ "The cyanobacteria derived toxin beta-n-methylamino-L-alanine and Amyotrophic Lateral Sclerosis". Toxins 2 (12): 2837–2850. 2010. doi:10.3390/toxins2122837. PMID 22069578.
- ↑ Holtcamp, W. (2012). "The emerging science of BMAA: do cyanobacteria contribute to neurodegenerative disease?". Environmental Health Perspectives 120 (3): a110–a116. doi:10.1289/ehp.120-a110. PMID 22382274.
- ↑ RA, Dunlop; SA, Banack; SL, Bishop; JS, Metcalf; SJ, Murch; DA, Davis; EW, Stommel; O, Karlsson et al. (2021). "Is Exposure to BMAA a Risk Factor for Neurodegenerative Diseases? A Response to a Critical Review of the BMAA Hypothesis". Neurotoxicity Research 39 (1): 81–106. doi:10.1007/s12640-020-00302-0. ISSN 1029-8428. PMID 33547590.
- ↑ Determining the Safety of L-serine in ALS.
- ↑ Safety Study of High Doses of Zinc in ALS Patients (completed).
 |

