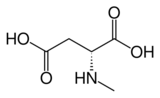
Chemistry:N-Methyl-D-aspartic acid
| |
| |
| |
| Names | |
|---|---|
| IUPAC name
N-Methyl-D-aspartic acid
| |
| Systematic IUPAC name
(2R)-2-(Methylamino)butanedioic acid[1] | |
| Other names
N-Methylaspartate; N-Methyl-D-aspartate; NMDA
| |
| Identifiers | |
3D model (JSmol)
|
|
| 1724431 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
| MeSH | N-Methylaspartate |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| C5H9NO4 | |
| Molar mass | 147.130 g·mol−1 |
| Appearance | White, opaque crystals |
| Odor | Odorless |
| Melting point | 189 to 190 °C (372 to 374 °F; 462 to 463 K) |
| log P | 1.39 |
| Acidity (pKa) | 2.206 |
| Basicity (pKb) | 11.791 |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
137 mg kg−1 (intraperitoneal, mouse) |
| Related compounds | |
Related amino acid derivatives
|
|
Related compounds
|
Dimethylacetamide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
N-methyl-D-aspartic acid or N-methyl-D-aspartate (NMDA) is an amino acid derivative that acts as a specific agonist at the NMDA receptor mimicking the action of glutamate, the neurotransmitter which normally acts at that receptor. Unlike glutamate, NMDA only binds to and regulates the NMDA receptor and has no effect on other glutamate receptors (such as those for AMPA and kainate). NMDA receptors are particularly important when they become overactive during, for example, withdrawal from alcohol as this causes symptoms such as agitation and, sometimes, epileptiform seizures.
Biological function
In 1962, J.C. Watkins reported synthesizing NMDA, an isomer of the previously known N-Methyl-DL-aspartic-acid (PubChem ID 4376).[2][3] NMDA is a water-soluble D-alpha-amino acid — an aspartic acid derivative with an N-methyl substituent and D-configuration — found across Animalia from lancelets to mammals.[4][5] At homeostatic levels NMDA plays an essential role as a neurotransmitter and neuroendocrine regulator.[6] At increased but sub–toxic levels NMDA becomes neuro-protective. [citation needed] In excessive amounts NMDA is an excitotoxin. Behavioral neuroscience research utilizes NMDA excitotoxicity to induce lesions in specific regions of an animal subject's brain or spinal cord to study behavioral changes.[7]
The mechanism of action for the NMDA receptor is a specific agonist binding to its NR2 subunits, and then a non-specific cation channel is opened, which can allow the passage of Ca2+ and Na+ into the cell and K+ out of the cell. Therefore, NMDA receptors will only open if glutamate is in the synapse and concurrently the postsynaptic membrane is already depolarized - acting as coincidence detectors at the neuronal level.[8] The excitatory postsynaptic potential (EPSP) produced by activation of an NMDA receptor also increases the concentration of Ca2+ in the cell. The Ca2+ can in turn function as a second messenger in various signaling pathways.[9][10][11][12] This process is modulated by a number of endogenous and exogenous compounds and plays a key role in a wide range of physiological (such as memory) and pathological processes (such as excitotoxicity).

Antagonists
Examples of antagonists, or more appropriately named receptor channel blockers, of the NMDA receptor are APV, amantadine, dextromethorphan (DXM), ketamine, magnesium,[13] tiletamine, phencyclidine (PCP), riluzole, memantine, methoxetamine (MXE), methoxphenidine (MXP) and kynurenic acid. While dizocilpine is generally considered to be the prototypical NMDA receptor blocker and is the most common agent used in research, animal studies have demonstrated some amount of neurotoxicity, which may or may not also occur in humans. These compounds are commonly referred to as NMDA receptor antagonists.
See also
- Anti-NMDA-receptor encephalitis
References
- ↑ "N-Methylaspartate - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 24 June 2005. Identification. https://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=22880&loc=ec_rcs.
- ↑ Watkins, J. C. (November 1962). "The synthesis of some acidic amino acids possessing neuropharmacological activity". Journal of Medicinal and Pharmaceutical Chemistry 5 (6): 1187–1199. doi:10.1021/jm01241a010. ISSN 1520-4804. PMID 14056452.
- ↑ Curtis, D. R.; Watkins, J. C. (September 1960). "The excitation and depression of spinal neurones by structurally related amino acids". Journal of Neurochemistry 6 (2): 117–141. doi:10.1111/j.1471-4159.1960.tb13458.x. ISSN 1471-4159. PMID 13718948.
- ↑ Todoroki, Natsumi; Shibata, Kimihiko; Yamada, Takahiro; Kera, Yoshio; Yamada, Ryo-hei (May 1999). "Determination of N-methyl-D-aspartic in tissues of bivalves by high-performance liquid chromatography". Journal of Chromatography B: Biomedical Sciences and Applications 728 (1): 41–47. doi:10.1016/S0378-4347(99)00089-4. ISSN 0378-4347. PMID 10379655.
- ↑ D'Aniello, Antimo; De Simone, Antonella; Spinelli, Patrizia; D'Aniello, Salvatore; Branno, Margherita; Aniello, Francesco; Rios, Jeannette; Tsesarskaja, Mara et al. (September 2002). "A specific enzymatic high-performance liquid chromatography method to determine N-methyl-D-aspartic acid in biological tissues". Analytical Biochemistry 308 (1): 42–51. doi:10.1016/S0003-2697(02)00326-3. ISSN 0003-2697. PMID 12234462.
- ↑ D'Aniello, Antimo; De Simone, Antonella; Spinelli, Patrizia; D'Aniello, Salvatore; Branno, Margherita; Aniello, Francesco; Rios, Jeannette; Tsesarskaja, Mara et al. (2002-09-01). "A specific enzymatic high-performance liquid chromatography method to determine N-methyl-D-aspartic acid in biological tissues". Analytical Biochemistry 308 (1): 42–51. doi:10.1016/S0003-2697(02)00326-3. ISSN 0003-2697. PMID 12234462. http://www.sciencedirect.com/science/article/pii/S0003269702003263. Retrieved 2020-05-02.
- ↑ Johnson, Patricia I.; Parente, Mary Ann; Stellar, James R. (May 1996). "NMDA-induced lesions of the nucleus accumbens or the ventral pallidum increase the rewarding efficacy of food to deprived rats". Brain Research 722 (1–2): 109–117. doi:10.1016/0006-8993(96)00202-8. ISSN 0006-8993. PMID 8813355.
- ↑ Buhusi, CV; Oprisan, SA; Buhusi, M (April 2016). "Clocks within Clocks: Timing by Coincidence Detection". Current Opinion in Behavioral Sciences 8: 207–213. doi:10.1016/j.cobeha.2016.02.024. PMID 27004236.
- ↑ Dingledine, R; Borges K (Mar 1999). "The glutamate receptor ion channels". Pharmacol. Rev. 51 (1): 7–61. PMID 10049997.
- ↑ Liu, Y; Zhang J (Oct 2000). "Recent development in NMDA receptors". Chin Med J (Engl) 113 (10): 948–956. PMID 11775847.
- ↑ Cull-Candy, S; Brickley S (Jun 2001). "NMDA receptor subunits: diversity, development and disease". Current Opinion in Neurobiology 11 (3): 327–335. doi:10.1016/S0959-4388(00)00215-4. PMID 11399431.
- ↑ Paoletti, P; Neyton J (Feb 2007). "NMDA receptor subunits: function and pharmacology". Current Opinion in Pharmacology 7 (1): 39–47. doi:10.1016/j.coph.2006.08.011. PMID 17088105. https://hal.archives-ouvertes.fr/hal-00115220/document.
- ↑ Murck, H. (2002-01-01). "Magnesium and Affective Disorders". Nutritional Neuroscience 5 (6): 375–389. doi:10.1080/1028415021000039194. ISSN 1028-415X. PMID 12509067.
Further reading
- Watkins, Jeffrey C.; Jane, David E. (2006), "The glutamate story", Br. J. Pharmacol. 147 (Suppl. 1): S100–S108, doi:10.1038/sj.bjp.0706444, PMID 16402093
- Blaise, Mathias-Costa; Sowdhamini, Ramanathan; Rao, Metpally Raghu Prasad; Pradhan, Nithyananda (2004), "Evolutionary trace analysis of ionotropic glutamate receptor sequences and modeling the interactions of agonists with different NMDA receptor subunits", J. Mol. Model. 10 (5–6): 305–316, doi:10.1007/s00894-004-0196-7, PMID 15597199
 |
