Medicine:Menopause
Menopause is the time when menstrual periods permanently stop, marking the end of the reproductive stage for the female human.[1][2][3] It typically occurs between the ages of 45 and 55, although the exact timing can vary.[4] Menopause is usually a natural change related to a decrease in circulating blood estrogen levels.[1] It can occur earlier in those who smoke tobacco.[1][5] Other causes include surgery that removes both ovaries, some types of chemotherapy, or anything that leads to a decrease in hormone levels.[6][1] At the physiological level, menopause happens because of a decrease in the ovaries' production of the hormones estrogen and progesterone.[1] While typically not needed, measuring hormone levels in the blood or urine can confirm a diagnosis.[7] Menopause is the opposite of menarche, the time when periods start.[8]
In the years before menopause, a woman's periods typically become irregular,[9][10] which means that periods may be longer or shorter in duration, or be lighter or heavier in the amount of flow.[9] During this time, women often experience hot flashes; these typically last from 30 seconds to ten minutes and may be associated with shivering, night sweats, and reddening of the skin.[9] Hot flashes can recur for four to five years.[2] Other symptoms may include vaginal dryness,[11] trouble sleeping, and mood changes.[9][12] The severity of symptoms varies between women.[2] Menopause is also known to cause thinning and drying of the skin as 30% of the skins collagen is lost during the first five years of menopause.[13] Menopause before the age of 45 years is considered to be "early menopause", and ovarian failure or surgical removal of the ovaries before the age of 40 years is termed "premature ovarian insufficiency".[14]
In addition to symptoms (hot flushes/flashes, night sweats, mood changes, arthralgia and vaginal dryness), the physical consequences of menopause include bone loss, increased central abdominal fat, and adverse changes in a woman's cholesterol profile and vascular function.[14] These changes predispose postmenopausal women to increased risks of osteoporosis and bone fracture, and of cardio-metabolic disease (diabetes and cardiovascular disease).[14]
Medical professionals often define menopause as having occurred when a woman has not had any menstrual bleeding for a year.[1] It may also be defined by a decrease in hormone production by the ovaries.[15] In those who have had surgery to remove their uterus but still have functioning ovaries, menopause is not considered to have yet occurred.[14] Following the removal of the uterus, symptoms of menopause typically occur earlier.[16] Iatrogenic menopause occurs when both ovaries are surgically removed (oophorectomy) along with the uterus for medical reasons.
Medical treatment of menopause is primarily to ameliorate symptoms and prevent bone loss.[17] Mild symptoms may be improved with treatment. With respect to hot flashes, avoiding nicotine, caffeine, and alcohol is often recommended; sleeping naked in a cool room and using a fan may help. The most effective treatment for menopausal symptoms is menopausal hormone therapy (MHT).[11][17] Non-hormonal therapies for hot flashes include cognitive-behavioral therapy, clinical hypnosis, gabapentin, and fezolinetant or selective serotonin reuptake inhibitors.[18][19] These will not improve symptoms such as neurocognitive impairments, joint pain or vaginal dryness, which affect over 55% of women.[17] Neurocognitive symptoms of menopause can be treated with lisdexamfetamine. Exercise may help with sleep disturbances. Many of the concerns about the use of MHT raised by older studies are no longer considered barriers to MHT in healthy women.[17] High-quality evidence for the effectiveness of alternative medicine has not been found.[2]
Signs and symptoms
File:Menopause - Project Wiki Women.mpg
During early menopause transition, the menstrual cycles remain regular but the interval between cycles begins to lengthen. Hormone levels begin to fluctuate. Ovulation may not occur with each cycle.[20]
The term menopause refers to a point in time that follows one year after the last menstruation.[20] During the menopausal transition and after menopause, women can experience a range of symptoms.[9] However, for women who enter the menopause transition without having regular menstrual cycles (due to prior surgery, other medical conditions or ongoing hormonal contraception) the menopause cannot be identified by bleeding patterns and is defined as the permanent loss of ovarian function.[17]
Vagina, uterus and bladder (urogenital tract)
During the transition to menopause, menstrual patterns can show shorter cycling (by 2–7 days);[20] longer cycles remain possible.[20] There may be irregular bleeding (lighter, heavier, spotting).[9][20] Dysfunctional uterine bleeding is often experienced by women approaching menopause due to the hormonal changes that accompany the menopause transition. Spotting or bleeding may simply be related to vaginal atrophy, a benign sore (polyp or lesion), or may be a functional endometrial response. The European Menopause and Andropause Society has released guidelines for assessment of the endometrium, which is usually the main source of spotting or bleeding.[21]
In post-menopausal women, however, any unscheduled vaginal bleeding is of concern and requires an appropriate investigation to rule out the possibility of malignant diseases. Urogenital symptoms may appear during menopause and continue through postmenopause and include painful intercourse, vaginal dryness and atrophic vaginitis (thinning of the membranes of the vulva, the vagina, the cervix and the outer urinary tract). There may also be considerable shrinking and loss in elasticity of all of the outer and inner genital areas. Urinary urgency may also occur and urinary incontinence in some women.[20][22]
Other physical effects
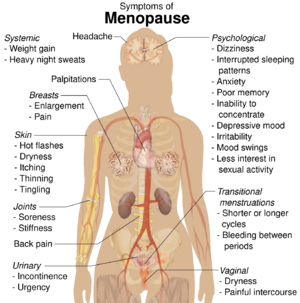
The most common physical symptoms of menopause are heavy night sweats, and hot flashes (also known as vasomotor symptoms).[23] Sleeping problems and insomnia are also common.[24] Other physical symptoms may be reported that are not specific to menopause but may be exacerbated by it, such as lack of energy, joint soreness, stiffness, back pain, breast enlargement, breast pain, heart palpitations, headache, dizziness, dry, itchy skin, thinning of the skin, tingling skin, rosacea, weight gain.[20][25]
Mood and memory effects
Psychological symptoms are often reported but they are not specific to menopause and can be caused by other factors.[26][27] They include anxiety, poor memory, inability to concentrate, depressive mood, irritability, mood swings, and less interest in sexual activity.[20][28][9]
Menopause-related cognitive impairment can be confused with the mild cognitive impairment that precedes dementia.[29] There is evidence of small decreases in verbal memory, on average, which may be caused by the effects of declining estrogen levels on the brain,[30] or perhaps by reduced blood flow to the brain during hot flashes.[31] However, these tend to resolve for most women during the postmenopause. Subjective reports of memory and concentration problems are associated with several factors, such as lack of sleep, and stress.[27][26]
Long-term effects
Cardiovascular health
Exposure to endogenous estrogen during reproductive years provides women with protection against cardiovascular disease, which is lost around 10 years after the onset of menopause. The menopausal transition is associated with an increase in fat mass (predominantly in visceral fat) and metabolic dysfunction–associated steatotic liver disease,[32] an increase in insulin resistance, dyslipidaemia, and endothelial dysfunction.[33] Women with vasomotor symptoms during menopause seem to have an especially unfavorable cardiometabolic profile,[34] as well as women with premature onset of menopause (before 45 years of age).[35] These risks can be reduced by managing risk factors, such as tobacco smoking, hypertension, increased blood lipids and body weight.[36][37]
Bone health
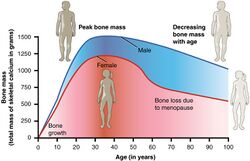
The annual rates of bone mineral density loss are highest starting one year before the final menstrual period and continuing through the two years after it.[38] Thus, post menopausal women are at increased risk of osteopenia, osteoporosis and fractures.
Causes
Menopause is a normal event in a woman's life and a natural part of aging.[22] Menopause can also be induced early.[39] Induced menopause occurs as a result of medical treatment such as chemotherapy, radiotherapy, oophorectomy, or complications of tubal ligation, hysterectomy, unilateral or bilateral salpingo-oophorectomy or leuprorelin usage.[40]
Age
Menopause typically occurs at some point between 47 and 54 years of age.[4] According to various data, more than 95% of women have their last period between the ages of 44–56 (median 49–50). 2% of women under the age of 40, 5% between the ages of 40–45 and the same number between the ages of 55–58 have their last bleeding.[41] The average age of the last period in the United States is 51 years, in Russia is 50 years, in Greece is 49 years, in Turkey is 47 years, in Egypt is 47 years and in India is 46 years.[42] Beyond the influence of genetics, these differences are also due to early-life environmental conditions[43] and associated with epigenetic effects.[44] The menopausal transition or perimenopause leading up to menopause usually lasts 3–4 years (sometimes as long as 5–14 years).[1][10]
Undiagnosed and untreated coeliac disease is a risk factor for early menopause. Coeliac disease can present with several non-gastrointestinal symptoms, in the absence of gastrointestinal symptoms, and most cases escape timely recognition and go undiagnosed, leading to a risk of long-term complications. A strict gluten-free diet reduces the risk. Women with early diagnosis and treatment of coeliac disease present a normal duration of fertile life span.[45][46] Women who have undergone hysterectomy with ovary conservation go through menopause on average 1.5 years earlier than the expected age.[17]
Premature ovarian insufficiency
In rare cases, a woman's ovaries stop working at a very early age, ranging anywhere from the age of puberty to age 40. This is known as premature ovarian failure or premature ovarian insufficiency (POI) and affects 1 to 2% of women by age 40.[47][48][49] It is diagnosed or confirmed by high blood levels of follicle stimulating hormone (FSH) and luteinizing hormone (LH) on at least three occasions at least four weeks apart.[50]
Premature ovarian insufficiency may be related to an auto immune disorder and therefore might co-occur with other autoimmune disorders such as thyroid disease, [adrenal insufficiency], and diabetes mellitus.[49] Other causes include chemotherapy, being a carrier of the fragile X syndrome gene, and radiotherapy.[49] However, in about 50–80% of cases of premature ovarian insufficiency, the cause is unknown, i.e., it is generally idiopathic.[48][50]
Early menopause can be related to cigarette smoking, higher body mass index, racial and ethnic factors, illnesses, and the removal of the uterus.[51]
Surgical menopause
Menopause can be surgically induced by bilateral oophorectomy (removal of ovaries),[39] which is often, but not always, done in conjunction with removal of the fallopian tubes (salpingo-oophorectomy) and uterus (hysterectomy).[52] Cessation of menses as a result of removal of the ovaries is called "surgical menopause". Surgical treatments, such as the removal of ovaries, might cause periods to stop altogether.[53] The sudden and complete drop in hormone levels may produce extreme withdrawal symptoms such as hot flashes, etc. The symptoms of early menopause may be more severe.[53]
Removal of the uterus without removal of the ovaries does not directly cause menopause, although pelvic surgery of this type can often precipitate a somewhat earlier menopause, perhaps because of a compromised blood supply to the ovaries. The time between surgery and possible early menopause is due to the fact that ovaries are still producing hormones.[53]
Mechanism
The transition has a variable degree of effects.[54] The stages of the menopause transition have been classified according to a woman's reported bleeding pattern, supported by changes in the pituitary follicle-stimulating hormone (FSH) levels.[55]
In younger women, during a normal menstrual cycle the ovaries produce estradiol, testosterone and progesterone in a cyclical pattern under the control of FSH and luteinizing hormone (LH), which are both produced by the pituitary gland. During perimenopause (approaching menopause), estradiol levels and patterns of production remain relatively unchanged or may increase compared to young women, but the cycles become frequently shorter or irregular.[56] The often observed increase in estrogen is presumed to be in response to elevated FSH levels that, in turn, is hypothesized to be caused by decreased feedback by inhibin.[57] Similarly, decreased inhibin feedback after hysterectomy is hypothesized to contribute to increased ovarian stimulation and earlier menopause.[58][59]
The menopausal transition is characterized by marked, and often dramatic, variations in FSH and estradiol levels. Because of this, measurements of these hormones are not considered to be reliable guides to a woman's exact menopausal status.[57] Menopause occurs because of the sharp decrease of estradiol and progesterone production by the ovaries. After menopause, estrogen continues to be produced mostly by aromatase in fat tissues and is produced in small amounts in many other tissues such as ovaries, bone, blood vessels, and the brain where it acts locally.[60] The substantial fall in circulating estradiol levels at menopause impacts many tissues, from brain to skin.
In contrast to the sudden fall in estradiol during menopause, the levels of total and free testosterone, as well as dehydroepiandrosterone sulfate (DHEAS) and androstenedione appear to decline more or less steadily with age. An effect of natural menopause on circulating androgen levels has not been observed.[61] Thus specific tissue effects of natural menopause cannot be attributed to loss of androgenic hormone production.[62]
Hot flashes and other vasomotor and body symptoms accompanying the menopausal transition are associated with estrogen insufficiency and changes that occur in the brain, primarily the hypothalamus and involve complex interplay between the neurotransmitters kisspeptin, neurokinin B, and dynorphin, which are found in KNDy neurons in the infundibular nucleus.[63]
Ovarian aging
Decreased inhibin feedback after hysterectomy is hypothesized to contribute to increased ovarian stimulation and earlier menopause. Hastened ovarian aging has been observed after endometrial ablation. While it is difficult to prove that these surgeries are causative, it has been hypothesized that the endometrium may be producing endocrine factors contributing to the endocrine feedback and regulation of the ovarian stimulation. Elimination of these factors contributes to faster depletion of the ovarian reserve. Reduced blood supply to the ovaries that may occur as a consequence of hysterectomy and uterine artery embolisation has been hypothesized to contribute to this effect.[58][59]
Impaired DNA repair mechanisms may contribute to earlier depletion of the ovarian reserve during aging.[64] As women age, double-strand breaks accumulate in the DNA of their primordial follicles. Primordial follicles are immature primary oocytes surrounded by a single layer of granulosa cells. An enzyme system is present in oocytes that ordinarily accurately repairs DNA double-strand breaks. This repair system is called "homologous recombinational repair", and it is especially effective during meiosis. Meiosis is the general process by which germ cells are formed in all sexual eukaryotes; it appears to be an adaptation for efficiently removing damages in germ line DNA.[65]
Human primary oocytes are present at an intermediate stage of meiosis, termed prophase I (see Oogenesis). Expression of four key DNA repair genes that are necessary for homologous recombinational repair during meiosis (BRCA1, MRE11, Rad51, and ATM) decline with age in oocytes.[64] This age-related decline in ability to repair DNA double-strand damages can account for the accumulation of these damages, that then likely contributes to the depletion of the ovarian reserve.
Diagnosis
Ways of assessing the impact on women of some of these menopause effects, include the Greene climacteric scale questionnaire,[66] the Cervantes scale[67] and the Menopause rating scale.[68]
Perimenopause
The term "perimenopause", which literally means "around the menopause", refers to the menopause transition years before the date of the final episode of flow.[1][10][69][70] According to the North American Menopause Society, this transition can last for four to eight years.[71] The Centre for Menstrual Cycle and Ovulation Research describes it as a six- to ten-year phase ending 12 months after the last menstrual period.[72]
During perimenopause, estrogen levels average about 20–30% higher than during premenopause, often with wide fluctuations.[72] These fluctuations cause many of the physical changes during perimenopause as well as menopause, especially during the last 1–2 years of perimenopause (before menopause).[69][73] Some of these changes are hot flashes, night sweats, difficulty sleeping, mood swings, vaginal dryness or atrophy, incontinence, osteoporosis, and heart disease.[72] Perimenopause is also associated with a higher likelihood of depression (affecting from 45 percent to 68 percent of perimenopausal women), which is twice as likely to affect those with a history of depression.[74][75]
During this period, fertility diminishes but is not considered to reach zero until the official date of menopause. The official date is determined retroactively, once 12 months have passed after the last appearance of menstrual blood. The menopause transition typically begins between 40 and 50 years of age (average 47.5).[76][77] The duration of perimenopause may be for up to eight years.[77] Women will often, but not always, start these transitions (perimenopause and menopause) about the same time as their mother did.[78] Some research appears to show that melatonin supplementation in perimenopausal women can improve thyroid function and gonadotropin levels, as well as restoring fertility and menstruation and preventing depression associated with menopause.[79]
Postmenopause
A period-like flow during postmenopause, even spotting, may be a sign of endometrial cancer.
Management
Perimenopause is a natural stage of life. It is not a disease or a disorder. Therefore, it does not automatically require any kind of medical treatment. However, in those cases where the physical, mental, and emotional effects of perimenopause are strong enough that they significantly disrupt the life of the woman experiencing them, palliative medical therapy may sometimes be appropriate.
Menopausal hormone therapy
In the context of the menopause, menopausal hormone therapy (MHT) is the use of estrogen in women without a uterus and estrogen plus progestogen in women who have an intact uterus.[80] MHT may be reasonable for the treatment of menopausal symptoms, such as hot flashes.[81] It is the most effective treatment option, especially when delivered as a skin patch.[82][83] Its use, however, appears to increase the risk of strokes and blood clots.[84] When used for menopausal symptoms the global recommendation is MHT should be prescribed for as long as there are defined treatment effects and goals for the individual woman.[17] MHT is also effective for preventing bone loss and osteoporotic fracture,[85] but it is generally recommended only for women at significant risk for whom other therapies are unsuitable.[86] MHT may be unsuitable for some women, including those at increased risk of cardiovascular disease, increased risk of thromboembolic disease (such as those with obesity or a history of venous thrombosis) or increased risk of some types of cancer.[86] There is some concern that this treatment increases the risk of breast cancer.[87][88] Women at increased risk of cardiometabolic disease and VTE may be able to use transdermal estradiol which does not appear to increase risks in low to moderate doses.[17] Adding testosterone to hormone therapy has a positive effect on sexual function in postmenopausal women, although it may be accompanied by hair growth or acne if used in excess. Transdermal testosterone therapy in appropriate dosing is generally safe.[89]
Selective estrogen receptor modulators
SERMs are a category of drugs, either synthetically produced or derived from a botanical source, that act selectively as agonists or antagonists on the estrogen receptors throughout the body. The most commonly prescribed SERMs are raloxifene and tamoxifen. Raloxifene exhibits oestrogen agonist activity on bone and lipids, and antagonist activity on breast and the endometrium.[90] Tamoxifen is in widespread use for treatment of hormone sensitive breast cancer. Raloxifene prevents vertebral fractures in postmenopausal, osteoporotic women and reduces the risk of invasive breast cancer.[91]
Other medications
Some of the SSRIs and SNRIs appear to provide some relief from vasomotor symptoms.[19][18] The most effective SSRIs and SNRIs are paroxetine, escitalopram, citalopram, venlafaxine, and desvenlafaxine.[18] They may, however, be associated with appetite and sleeping problems, constipation and nausea.[19][92]
Gabapentin or fezolinetant can also improve the frequency and severity of vasomotor symptoms.[19][18] Side effects of using gabapentin include drowsiness and headaches.[19][92]
Therapy
Cognitive behavioural therapy and clinical hypnosis can decrease the amount women are affected by hot flashes.[18] Mindfulness is not yet proven to be effective in easing vasomotor symptoms.[93][94][18]
Lifestyle and exercise
Exercise has been thought to reduce postmenopausal symptoms through the increase of endorphin levels, which decrease as estrogen production decreases.[95] However, there is insufficient evidence to suggest that exercise helps with the symptoms of menopause.[18] Similarly, yoga has not been shown to be useful as a treatment for vasomotor symptoms.[18]
However a high BMI is a risk factor for vasomotor symptoms in particular. Weight loss may help with symptom management.[96][18]
There is no strong evidence that cooling techniques such as using specific clothing or environment control tools (for example fans) help with symptoms.[18] Paced breathing and relaxation are not effective in easing symptoms.[18]
Research suggests that, as hormones and gut microbiome are intertwined in some ways, hormone fluctuations during menopause can alter the gut environment and microbial activity.[97] This can contribute to menopausal symptoms, including weight gain and mood swings.[98] Diet can support a healthy gut microbiome, and avoiding things like alcohol and added sugar, which can disrupt this environment, could mitigate symptoms.[99][100][101]
Dietary supplements
There is no evidence of consistent benefit of taking any dietary supplements or herbal products for menopausal symptoms.[18][102][103] These widely marketed but ineffective supplements include soy isoflavones, pollen extracts, black cohosh, omega-3 among many others.[18][104][105]
Alternative medicine
There is no evidence of consistent benefit of alternative therapies for menopausal symptoms despite their popularity.[103][18] As of 2023, there is no evidence to support the efficacy of acupuncture as a management for menopausal symptoms.[18][106][103] The Cochrane review found not enough evidence in 2016 to show a difference between Chinese herbal medicine and placebo for the vasomotor symptoms.[107]
Other efforts
- Lack of lubrication is a common problem during and after perimenopause. Vaginal moisturizers can help women with overall dryness, and lubricants can help with lubrication difficulties that may be present during intercourse. It is worth pointing out that moisturizers and lubricants are different products for different issues: some women complain that their genitalia are uncomfortably dry all the time, and they may do better with moisturizers. Those who need only lubricants do well using them only during intercourse.
- Low-dose prescription vaginal estrogen products such as estrogen creams are generally a safe way to use estrogen topically, to help vaginal thinning and dryness problems (see vaginal atrophy) while only minimally increasing the levels of estrogen in the bloodstream.
- Individual counseling or support groups can sometimes be helpful to handle sad, depressed, anxious or confused feelings women may be having as they pass through what can be for some a very challenging transition time.
- Osteoporosis can be minimized by smoking cessation, adequate vitamin D intake and regular weight-bearing exercise. The bisphosphonate drug alendronate may decrease the risk of a fracture, in women that have both bone loss and a previous fracture and less so for those with just osteoporosis.[108]
- A surgical procedure where a part of one of the ovaries is removed earlier in life and frozen and then over time thawed and returned to the body (ovarian tissue cryopreservation) has been tried. While at least 11 women have undergone the procedure and paid over £6,000, there is no evidence it is safe or effective.[109]
Society and culture
Attitudes and experiences
The menopause transition is a process, involving hormonal, menstrual, and typically vasomotor changes. However, the experience of the menopause as a whole is very much influenced by psychological and social factors, such as past experience, lifestyle, social and cultural meanings of menopause, and a woman's social and material circumstances. Menopause has been described as a biopsychosocial experience, with social and cultural factors playing a prominent role in the way menopause is experienced and perceived.[110]
The paradigm within which a woman considers menopause influences the way she views it: women who understand menopause as a medical condition rate it significantly more negatively than those who view it as a life transition or a symbol of aging.[111] There is some evidence that negative attitudes and expectations, held before the menopause, predict symptom experience during the menopause,[112] and beliefs and attitudes toward menopause tend to be more positive in postmenopausal than in premenopausal women.[113] Women with more negative attitudes towards the menopause report more symptoms during this transition.[112]
Though there has been a shift towards more public conversations around menopause, it is often still seen as a private process and is predominantly understood through the medical paradigm.[114] Despite these growing conversations, the embodied experience of menopause is often reduced to biological symptoms rather than understood as a complex social and cultural process.[115] Hot flashes are one of the most common symptoms of menopause.[116] They are also associated with psychological symptoms such as anxiety[116] and embarrassment.[117]
Heat and emotionality are frequently linked, which can socially associate hot flashes with a breakdown of emotional and physical regulation.[117] This can be partially understood as hot flashes breaking down barriers between private, internal bodily processes and the external world, physically manifesting menopause. Observers of menopause have sometimes interpreted it as a complete loss of the ability to regulate emotions, an interpretation frequently at odds with how it is actually experienced by the individual.[117]
Victorian medical understandings — which positioned women's health as primarily controlled by the reproductive system — could have influenced ideas around hot flashes as revealing the dominance of reproductive control over the individual. Feminist critiques of medical treatment of menopause, however, have framed it as part of a broader social and political system "that attempts to control the effects of the aging of the female body, which loses its social significance with the loss of reproductive capacity".[114] This tension between medicalisation and feminist perspectives highlights how menopause has been framed less as a natural life stage and more as a site of social regulation and control.
Menopause is a stage of life experienced in different ways. It can be characterized by personal challenges, changes in personal roles within the family and society. Women's approaches to changes during menopause are influenced by their personal, family and sociocultural background.[118] Women from different regions and countries also have different attitudes. Postmenopausal women had more positive attitudes toward menopause compared with peri- or premenopausal women. Other influencing factors of attitudes toward menopause include age, menopausal symptoms, psychological and socioeconomical status, and profession and ethnicity.[119]
Ethnicity and geography play roles in the experience of menopause. American women of different ethnicities report significantly different types of menopausal effects. One major study found Caucasian women most likely to report what are sometimes described as psychosomatic symptoms, while African-American women were more likely to report vasomotor symptoms.[120]
There may be variations in experiences of women from different ethnic backgrounds regarding menopause and care. Immigrant women reported more vasomotor symptoms and other physical symptoms and poorer mental health than non-immigrant women and were mostly dissatisfied with the care they had received. Self-management strategies for menopausal symptoms were also influenced by culture.[121]
Two multinational studies of Asian women, found that hot flushes were not the most commonly reported symptoms, instead body and joint aches, memory problems, sleeplessness, irritability and migraines were.[122] In another study comparing experiences of menopause amongst White Australian women and women in Laos, Australian women reported higher rates of depression, as well as fears of aging, weight gain and cancer – fears not reported by Laotian women, who positioned menopause as a positive event.[123] Japanese women experience menopause effects, or kōnenki (更年期), in a different way from American women.[124] Japanese women report lower rates of hot flashes and night sweats; this can be attributed to a variety of factors, both biological and social. Historically, kōnenki was associated with wealthy middle-class housewives in Japan, i.e., it was a "luxury disease" that women from traditional, inter-generational rural households did not report. Menopause in Japan was viewed as a symptom of the inevitable process of aging, rather than a "revolutionary transition", or a "deficiency disease" in need of management.[124]
As of 2005, in Japanese culture, reporting of vasomotor symptoms has been on the increase, with research finding that of 140 Japanese participants, hot flashes were prevalent in 22.1%.[125] This was almost double that of 20 years prior.[126] Whilst the exact cause for this is unknown, possible contributing factors include dietary changes, increased medicalisation of middle-aged women and increased media attention on the subject.[126] However, reporting of vasomotor symptoms is still "significantly" lower than in North America.[127]
Additionally, while most women in the United States apparently have a negative view of menopause as a time of deterioration or decline, some studies seem to indicate that women from some Asian cultures have an understanding of menopause that focuses on a sense of liberation and celebrates the freedom from the risk of pregnancy.[128] Diverging from these conclusions, one study appeared to show that many American women "experience this time as one of liberation and self-actualization".[129]
Recent biosocial and anthropological scholarship cautions against interpreting ethnic or national differences in menopausal timing and symptom burden as innate racial biology. A 2024 U.S. scoping review synthesizing fifteen studies reported that African American women, on average, reach natural menopause earlier than White women and report vasomotor symptoms more frequently. The review links these patterns to cumulative psychosocial stressors—including racial discrimination, lower income, and heavy caregiving demands—rather than to genetic difference. It also reports associations between intimate partner violence and childhood abuse with greater symptom severity, as well as higher hormone therapy use among women veterans, patterns interpreted as markers of heightened chronic stress exposure.[130] These findings highlight how experiences of inequality and trauma can become biologically embedded, shaping health outcomes across the life course. Anthropological syntheses likewise emphasize "local biologies," showing that symptom profiles vary across societies as labor rhythms, diet, clinical discourse, and life-course endocrinology interact. For example, Japanese women commonly report chilliness rather than hot flushes, and symptom labeling and expectations differ accordingly.[131] This underscores that menopause is not only a physiological transition but also a socially mediated experience that reflects broader cultural environments. Together, these sources suggest that apparent disparities are best understood as embodied effects of unequal social conditions and culturally specific meanings. Explanations focused only on attitudes or estrogen may overlook wider social determinants and limit clinical care.[131]
In some women, menopause may bring about a sense of loss related to the end of fertility. In addition, this change often aligns with other stressors, such as the responsibility of looking after elderly parents or dealing with the emotional challenges of "empty nest syndrome" when children move out of the family home. This situation can be accentuated in cultures where being older is negatively perceived.
LGBTQIA+ experiences
The experience of menopause for non-binary and transgender people can be especially difficult in relation to gender, as menopause is heavily defined as a feminine experience.[132] Menopause is typically defined as the discontinuation of menstrual periods, the conclusion of fertility, and the finalisation of ovarian function — all of which are deeply connected to traditional understandings of gender and femininity.[133] One study, published in the International Journal of Transgender Health, reported that non-binary individuals experienced menopause as having an impact on their gender identity, either positively or negatively, and that many also struggled to find supportive resources and spaces to share their experiences.[133]
Some participants further highlighted broader societal prejudices that undermined their sense of security — especially as menopause is still largely framed through traditional ideas of gender, hormones, and the female body.[133] They also emphasised that many medical professionals lacked adequate understanding of menopause in relation to non-binary and transgender people. This lack of awareness often created barriers to accessing essential health services that recognised and met their needs.[133] A 2021 UK GP Patient Survey found that while trans and non‑binary adults reported similar access to primary care as the general population, they were significantly less likely to feel involved in decisions about their treatment (85.0% versus 93.7%) and less likely to feel their needs were met overall (88.1% versus 95.2%).[134]
Menopause across cultures
Menopause is both viewed and experienced differently in cultures around the globe. From a Western perspective, focus on menopause tends to be more scientific, with an emphasis on the management of symptoms.
In post-war (modern) Japan, there has been a sharp increase in the medicalisation of menopause. Anthropologist Margaret Lock explores the cultural construction of the 'menopausal syndrome' present in Japanese rhetoric.[135] Whilst symptoms of 'headaches, shoulder stiffness, and dizziness' are viewed as usual menopause symptoms, 'changes in endocrine function and in the autonomic nervous system' are classified as menopausal.[135] The menopausal syndrome is perpetrated as being more common in housewives. This is because as they age, "their traditional role [as a housewife] is in jeopardy", creating larger susceptibility to stress.[135] Menopausal syndrome is highly stigmatised and associated with being selfish, pleasure driven, and deficient in willpower.[135] However, there is a significant difference between the rhetoric and statistical results. Lock reports that housewives do not experience higher menopausal symptoms. In her sample, 53% of women reported going through menopause, yet only 12% reported visiting the gynaecologist in the past two years, and only 2% had taken prescribed estrogen replacement therapy.[135] A curation of the societal role of women, increasing medicalisation, economic concern, and the 'usefulness' of middle-aged women has led to this increasing debate over menopause in Japan. However, while both symptoms and rhetoric grow, so does the 'political consciousness' of Japanese middle-aged women.[135]
Throughout Mayan history, the cultural perception of menopause is primarily understood as a biological transitioning, rather than bearing any medical or societal significance. An article written by Yewoubdar Beyen delves into the experiences of menopause by Mayan women in Yucatán.[136] Discoveries made by this study promote the cultural construction of menopause as "unmarked" by stigma, rituals or significance. The emphasis of menopause in Western societies differentiated from the Mayan perception, where it is beheld as a natural part of development, holding no substantiating symbolic importance. Beyen found that 68.2% of Mayan women reported no distressing health changes, which may suggest that the absence of medicalisation leads to a reduction of symptom reporting.[136] Similar results were evident in Donna Stewart's investigation on Highland Guatemalan Mayan women who welcomed their symptoms and perceived them positively.[137] Some Mayan women express a sense of liberation after menopause; free from menstruation and childbearing. Cross-cultural research affirms this, discovering that feelings around menopause varies through cultural interpretation.[138] The absence of negative cultural connotations enables Mayan women for a more normalised transition to an older stage of life. Collectively, these discoveries challenge the Western model of menopause by exploring how neutral cultural beliefs shape experiences of menopause.
A study conducted at Monash University saw 74% of post-menopausal Australian women under the age of 55 suffer from vasomotor symptoms, with 28% reporting them as "severely bothersome".[139] It is statistics like this which have promoted the use of Hormone Replacement Therapy (HRT), also known as Menopausal Hormone Therapy (MHT) in Australia. These ongoing studies have proven that for symptomatic women, non-hormonal remedies are significantly "less effective than MHT".[139] This in turn has opened the floor for conversations surrounding menopause and its various symptoms, leading to further education for health professionals and women alike, through public health campaigns promoting evidence-based education.[139]
Along with menopausal education in Australia, the importance of biocultural research in this area is being explored. An examination conducted by a team of female anthropologists at BMC Women's Health denotes that there is extensive research for Australian women as a collective while only two reports relating to the Indigenous population. Through this the anthropological team highlight the importance of understanding "culture, social disadvantage"[140] and suggest that this would aid the population and improve "management and support for Indigenous Australian women".[141]
Impact on work
Midlife is typically a life stage when men and women may be dealing with demanding life events and responsibilities, such as work, health problems, and caring roles. For example, in 2018 in the UK women aged 45–54 report more work-related stress than men or women of any other age group.[142] Hot flashes are often reported to be particularly distressing at work and lead to embarrassment and worry about potential stigmatisation.[143] A June 2023 study by the Mayo Clinic estimated an annual loss of $1.8 billion in the United States due to workdays missed as a result of menopause symptoms.[144] This was one of the largest studies to date examining the impact of menopause symptoms on work outcomes. The research concluded there was a strong need to improve medical treatment for menopausal women and make the workplace environment more supportive to avoid such productivity losses.
Etymology
Menopause literally means the "end of monthly cycles" (the end of monthly periods or menstruation), from the Greek word pausis ("pause") and mēn ("month"). This is a medical coinage; the Greek word for menses is actually different. In Ancient Greek, the menses were described in the plural, ta emmēnia ("the monthlies"), and its modern descendant has been clipped to ta emmēna. The Modern Greek medical term is emmenopausis in Katharevousa or emmenopausi in Demotic Greek. The Ancient Greeks did not produce medical concepts about any symptoms associated with end of menstruation and did not use a specific word to refer to this time of a woman's life. The word menopause was invented by French doctors at the beginning of the nineteenth century. Greek etymology was reconstructed at this time and it was the Parisian student doctor Charles-Pierre-Louis de Gardanne who invented a variation of the word in 1812, which was edited to its final French form in 1821.[145]
Some of them noted that peasant women had no complaints about the end of menses, while urban middle-class women had many troubling symptoms. Doctors at this time considered the symptoms to be the result of urban lifestyles of sedentary behaviour, alcohol consumption, too much time indoors, and over-eating, with a lack of fresh fruit and vegetables.[146]
The word "menopause" was coined specifically for female humans, where the end of fertility is traditionally indicated by the permanent stopping of monthly menstruations. However, menopause exists in some other animals, many of which do not have monthly menstruation;[147] in this case, the term means a natural end to fertility that occurs before the end of the natural lifespan.
In popular culture, law and politics
In the 21st century, celebrities have spoken out about their experiences of the menopause, which has led to it becoming less of a taboo as it has boosted awareness of the debilitating symptoms. Subsequently, TV shows have been running features on the menopause to help women experiencing symptoms. In the UK Lorraine Kelly has been an advocate for getting women to speak about their experiences including sharing her own. This has led to an increase in women seeking treatment such as HRT.[148] Davina McCall also led an awareness campaign based on a documentary on Channel 4.[149]
In the UK, Carolyn Harris sponsored the Menopause (Support and Services) Bill in June 2021. It was to exempt hormone replacement therapy from National Health Service prescription charges and to make provisions about menopause support and services, including public education and communication in supporting perimenopausal and post-menopausal women, and to raise awareness of menopause and its effects. The bill was withdrawn on 29 October 2021.[150]
In the US, David McKinley, Republican from West Virginia introduced the Menopause Research Act in September 2022 for $100 million in 2023 and 2024, but it stalled.[151]
Other animals
The majority of mammal species reach menopause when they cease the production of ovarian follicles, which contain eggs (oocytes), between one-third and two-thirds of their maximum possible lifespan.[152] However, few live long enough in the wild to reach this point. Humans are joined by a limited number of other species in which females live substantially longer than their ability to reproduce. Examples of others include cetaceans: beluga whales,[153] narwhals,[153] orcas,[154] false killer whales[155] and short-finned pilot whales.[156]
Menopause has been reported in a variety of other vertebrate species, but these examples tend to be from captive individuals, and thus are not necessarily representative of what happens in natural populations in the wild. Menopause in captivity has been observed in several species of nonhuman primates,[147] including rhesus macaques[157] and chimpanzees.[158] Some research suggests that wild chimpanzees do not experience menopause, as their fertility declines are associated with declines in overall health.[159] Menopause has been reported in elephants in captivity[160] and guppies.[161] Dogs do not experience menopause; the canine estrus cycle simply becomes irregular and infrequent. Although older female dogs are not considered good candidates for breeding, offspring have been produced by older animals, see Canine reproduction. Similar observations have been made in cats.[162]
Life histories show a varying degree of senescence; rapid senescing organisms (e.g., Pacific salmon and annual plants) do not have a post-reproductive life-stage. Gradual senescence is exhibited by all placental mammalian life histories.[original research?]
Evolution
There are various theories on the origin and process of the evolution of the menopause. These attempt to suggest evolutionary benefits to the human species stemming from the cessation of women's reproductive capability before the end of their natural lifespan. It is conjectured that in highly social groups natural selection favors females that stop reproducing and devote that post-reproductive life span to continuing to care for existing offspring, both their own and those of others to whom they are related, especially their granddaughters and grandsons.[163]
See also
- European Menopause and Andropause Society
- Menopause in the workplace
- Menopause in incarceration
- Pregnancy over age 50
- Biological clock
- Evolution of menopause
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Cite error: Invalid
<ref>tag; no text was provided for refs namedNIH2013-Basics - ↑ 2.0 2.1 2.2 2.3 Menopause: Overview. Institute for Quality & Efficiency in Health Care. July 2, 2020. https://www.ncbi.nlm.nih.gov/books/NBK279311/. Retrieved October 20, 2022.
- ↑ Angelou, Kyveli; Grigoriadis, Themos; Diakosavvas, Michail; Zacharakis, Dimitris; Athanasiou, Stavros (2020-04-08). "The Genitourinary Syndrome of Menopause: An Overview of the Recent Data" (in en). Cureus 12 (4). doi:10.7759/cureus.7586. ISSN 2168-8184. PMID 32399320.
- ↑ 4.0 4.1 "Menopause". The Medical Clinics of North America 99 (3): 521–534. May 2015. doi:10.1016/j.mcna.2015.01.006. PMID 25841598.
- ↑ The menopausal transition: interface between gynecology and psychiatry ([Online-Ausg.] ed.). Basel: Karger. 2009. p. 73. ISBN 978-3-8055-9101-0.
- ↑ "Menopause & Chemotherapy - Managing Side Effects - Chemocare". https://chemocare.com/chemotherapy/side-effects/menopause-chemotherapy.aspx.
- ↑ "How do health care providers diagnose menopause?". Eunice Kennedy Shriver National Institute of Child Health and Human Development. 6 May 2013. https://www.nichd.nih.gov/health/topics/menopause/conditioninfo/diagnosed.
- ↑ "9". Dynamics of Human Reproduction: Biology, Biometry, Demography. Transaction Publishers. p. 401. ISBN 978-0-202-36570-1. https://books.google.com/books?id=I0_SrDBNy24C&pg=PA401.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 "Menopause Symptoms and Relief". February 22, 2021. https://www.womenshealth.gov/menopause/menopause-symptoms-and-relief.
- ↑ 10.0 10.1 10.2 "What Is Menopause?". September 30, 2021. https://www.nia.nih.gov/health/what-menopause.
- ↑ 11.0 11.1 Mark, J. K. K.; Samsudin, S.; Looi, I.; Yuen, K. H. (2024-05-03). "Vaginal dryness: a review of current understanding and management strategies" (in en). Climacteric 27 (3): 236–244. doi:10.1080/13697137.2024.2306892. ISSN 1369-7137. PMID 38318859. https://www.tandfonline.com/doi/full/10.1080/13697137.2024.2306892.
- ↑ Marino, Jean M. (November 2021). "Genitourinary Syndrome of Menopause" (in en). Journal of Midwifery & Women's Health 66 (6): 729–739. doi:10.1111/jmwh.13277. ISSN 1526-9523. PMID 34464022. https://onlinelibrary.wiley.com/doi/10.1111/jmwh.13277.
- ↑ "Caring for your skin in menopause". https://www.aad.org/public/everyday-care/skin-care-secrets/anti-aging/skin-care-during-menopause.
- ↑ 14.0 14.1 14.2 14.3 "Menopause". Nature Reviews. Disease Primers 1 (1). April 2015. doi:10.1038/nrdp.2015.4. PMID 27188659.
- ↑ Menopause: a biocultural perspective ([Online-Ausg.] ed.). New Brunswick, N.J.: Rutgers University Press. 2006. p. 81. ISBN 978-0-8135-3856-3. https://books.google.com/books?id=fWH606kOGYQC&pg=PA81.
- ↑ International position paper on women's health and menopause: a comprehensive approach. DIANE Publishing. 2002. p. 36. ISBN 978-1-4289-0521-4. https://books.google.com/books?id=NCzYsz6Uv0IC&pg=PA36.
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 17.6 17.7 "Treating menopause - MHT and beyond". Nature Reviews. Endocrinology 18 (8): 490–502. August 2022. doi:10.1038/s41574-022-00685-4. PMID 35624141.
- ↑ 18.00 18.01 18.02 18.03 18.04 18.05 18.06 18.07 18.08 18.09 18.10 18.11 18.12 18.13 18.14 "The 2023 nonhormone therapy position statement of The North American Menopause Society" (in en). Menopause 30 (6): 573–590. 2023-06-21. doi:10.1097/GME.0000000000002200. ISSN 1072-3714. PMID 37252752. https://journals.lww.com/10.1097/GME.0000000000002200.
- ↑ 19.0 19.1 19.2 19.3 19.4 "Hormonal and nonhormonal treatment of vasomotor symptoms". Obstetrics and Gynecology Clinics of North America 42 (1): 163–179. March 2015. doi:10.1016/j.ogc.2014.09.008. PMID 25681847.
- ↑ 20.0 20.1 20.2 20.3 20.4 20.5 20.6 20.7 Williams Gynecology. New York: McGraw-Hill Medical. 2012. pp. 555–56. ISBN 978-0-07-171672-7.
- ↑ "EMAS clinical guide: assessment of the endometrium in peri and postmenopausal women". Maturitas 75 (2): 181–90. June 2013. doi:10.1016/j.maturitas.2013.03.011. PMID 23619009.
- ↑ 22.0 22.1 Allafi, Ahmad Hasan; Al-Johani, Amal Saleem; Babukur, Raed M.; Fikri, Jehad; Alanazi, Raneem Rashed; Ali, Sara Dafaalla Mohamed Hag; Alkathiry, Abdulrahman; Alfozan, Abdalmelk Mohmed et al. (2024). "The Link Between Menopause and Urinary Incontinence: A Systematic Review". Cureus 16 (10). doi:10.7759/cureus.71260. ISSN 2168-8184. PMID 39525118.
- ↑ "Management of Menopausal Symptoms: A Review". JAMA 329 (5): 405–420. February 2023. doi:10.1001/jama.2022.24140. PMID 36749328.
- ↑ "Menopausal Symptoms and Their Management". Endocrinology and Metabolism Clinics of North America 44 (3): 497–515. September 2015. doi:10.1016/j.ecl.2015.05.001. PMID 26316239.
- ↑ "Red in the Face" (in en). 27 June 2017. https://newsinhealth.nih.gov/2012/08/red-face.
- ↑ 26.0 26.1 Hogervorst, Eef; Craig, Jen; O'Donnell, Emma (2022-05-01). "Cognition and mental health in menopause: A review" (in en). Best Practice & Research Clinical Obstetrics & Gynaecology 81: 69–84. doi:10.1016/j.bpobgyn.2021.10.009. ISSN 1521-6934. PMID 34969617. https://www.sciencedirect.com/science/article/pii/S1521693421001644.
- ↑ 27.0 27.1 Kilpi, Fanny; Soares, Ana Luiza G.; Fraser, Abigail; Nelson, Scott M.; Sattar, Naveed; Fallon, Sean James; Tilling, Kate; Lawlor, Deborah A. (14 August 2020). "Changes in six domains of cognitive function with reproductive and chronological ageing and sex hormones: a longitudinal study in 2411 UK mid-life women" (in en). BMC Women's Health 20 (1): 177. doi:10.1186/s12905-020-01040-3. ISSN 1472-6874. PMID 32795281.
- ↑ "Depressive disorders and the menopause transition". Maturitas 71 (2): 120–30. February 2012. doi:10.1016/j.maturitas.2011.11.017. PMID 22196311.
- ↑ "Memory and Mood in the Menopause". Managing the Menopause: 21st Century Solutions. Cambridge University Press. 20 August 2015. ISBN 978-1-316-35271-7. https://books.google.com/books?id=UZ9rCgAAQBAJ&pg=PA91.
- ↑ Pre-Menopause, Menopause and Beyond: Volume 5: Frontiers in Gynecological Endocrinology. Springer. 30 January 2018. pp. 38–39. ISBN 978-3-319-63540-8. https://books.google.com/books?id=JtBJDwAAQBAJ&pg=PA38.
- ↑ Current Medical Diagnosis and Treatment 2018, 57th Edition. McGraw Hill Professional. 11 September 2017. p. 1212. ISBN 978-1-259-86149-9. https://books.google.com/books?id=xTExDwAAQBAJ.
- ↑ Stokar, Joshua; Dresner-Pollak, Rivka (2026). "Earlier Menopause and Risk of Metabolic Dysfunction-Associated Steatotic Liver Disease: A Global Cohort Study" (in en). Diabetes/Metabolism Research and Reviews 42 (1). doi:10.1002/dmrr.70121. ISSN 1520-7560. https://onlinelibrary.wiley.com/doi/abs/10.1002/dmrr.70121.
- ↑ "Menopause: a cardiometabolic transition" (in English). The Lancet. Diabetes & Endocrinology 10 (6): 442–456. June 2022. doi:10.1016/S2213-8587(22)00076-6. PMID 35525259.
- ↑ "Vasomotor symptoms: natural history, physiology, and links with cardiovascular health". Climacteric 21 (2): 96–100. April 2018. doi:10.1080/13697137.2018.1430131. PMID 29390899.
- ↑ "Cardiometabolic health in premature ovarian insufficiency". Climacteric 24 (5): 474–480. October 2021. doi:10.1080/13697137.2021.1910232. PMID 34169795.
- ↑ "Autonomic Cardiovascular Damage during Post-menopause: the Role of Physical Training". Aging and Disease 4 (6): 320–328. September 2013. doi:10.14336/AD.2013.0400320. PMID 24307965.
- ↑ ESHRE Capri Workshop Group (2011). "Perimenopausal risk factors and future health". Human Reproduction Update 17 (5): 706–717. doi:10.1093/humupd/dmr020. PMID 21565809.
- ↑ "Changes in bone mineral density with age in men and women: a longitudinal study". Osteoporosis International 13 (2): 105–112. 2002-02-01. doi:10.1007/s001980200001. PMID 11905520.
- ↑ 39.0 39.1 "Early or premature menopause | Office on Women's Health". https://www.womenshealth.gov/menopause/early-or-premature-menopause.
- ↑ "Gynaecologic Problems: Menopausal Problems". Health on the Net Foundation. http://www.hon.ch/Dossier/MotherChild/gynae_problems/menopausal.html.
- ↑ "International variability in ages at menarche, first livebirth, and menopause. World Health Organization Collaborative Study of Neoplasia and Steroid Contraceptives". American Journal of Epidemiology 148 (12): 1195–205. December 1998. doi:10.1093/oxfordjournals.aje.a009609. PMID 9867266.
- ↑ "Menopause and treatments". Quality of Life Research 9 (6): 695–707. 2000. doi:10.1023/A:1008913605129.
- ↑ Begum, Khurshida; Muttukrishna, Shanthi; Sievert, Lynnette Leidy; Sharmeen, Taniya; Murphy, Lorna; Chowdhury, Osul; Kasim, Adetayo; Gunu, Richard et al. (March 2016). "Ethnicity or environment: effects of migration on ovarian reserve among Bangladeshi women in the United Kingdom" (in en). Fertility and Sterility 105 (3): 744–754.e1. doi:10.1016/j.fertnstert.2015.11.024. PMID 26706133. https://linkinghub.elsevier.com/retrieve/pii/S0015028215021081.
- ↑ Bar-Sadeh, Ben; Rudnizky, Sergei; Pnueli, Lilach; Bentley, Gillian R.; Stöger, Reinhard; Kaplan, Ariel; Melamed, Philippa (2020-09-15). "Unravelling the role of epigenetics in reproductive adaptations to early-life environment" (in en). Nature Reviews Endocrinology 16 (9): 519–533. doi:10.1038/s41574-020-0370-8. ISSN 1759-5029. PMID 32620937. https://www.nature.com/articles/s41574-020-0370-8.
- ↑ "Celiac disease and reproductive disorders: meta-analysis of epidemiologic associations and potential pathogenic mechanisms". Human Reproduction Update 20 (4): 582–93. 2014. doi:10.1093/humupd/dmu007. PMID 24619876.
- ↑ "Risk of infertility in patients with celiac disease: a meta-analysis of observational studies". Arquivos de Gastroenterologia 51 (2): 144–50. 2014. doi:10.1590/S0004-28032014000200014. PMID 25003268.
- ↑ "Premature ovarian insufficiency: the context of long-term effects". Journal of Endocrinological Investigation 39 (9): 983–90. September 2016. doi:10.1007/s40618-016-0467-z. PMID 27091671.
- ↑ 48.0 48.1 "Aetiological coding sequence variants in non-syndromic premature ovarian failure: From genetic linkage analysis to next generation sequencing". Molecular and Cellular Endocrinology 411: 243–57. August 2015. doi:10.1016/j.mce.2015.05.005. PMID 25960166.
- ↑ 49.0 49.1 49.2 "Premature ovarian insufficiency: Pathogenesis and management". Journal of Mid-Life Health 6 (4): 147–53. 2015. doi:10.4103/0976-7800.172292. PMID 26903753.
- ↑ 50.0 50.1 "Premature ovarian failure". Endocrinology and Metabolism Clinics of North America 27 (4): 989–1006. December 1998. doi:10.1016/s0889-8529(05)70051-7. PMID 9922918.
- ↑ Bucher, et al. 1930
- ↑ "Executive summary of the Stages of Reproductive Aging Workshop + 10: addressing the unfinished agenda of staging reproductive aging". Fertility and Sterility 97 (4): 843–51. April 2012. doi:10.1016/j.fertnstert.2012.01.128. PMID 22341880.
- ↑ 53.0 53.1 53.2 "Early or premature menopause". Womenshealth.gov. 12 July 2017. https://www.womenshealth.gov/menopause/early-or-premature-menopause.
- ↑ "Risk for new onset of depression during the menopausal transition: the Harvard study of moods and cycles". Archives of General Psychiatry 63 (4): 385–90. April 2006. doi:10.1001/archpsyc.63.4.385. PMID 16585467.
- ↑ "Executive summary: Stages of Reproductive Aging Workshop (STRAW)". Climacteric 4 (4): 267–72. December 2001. doi:10.1080/cmt.4.4.267.272. PMID 11770182.
- ↑ "Perimenopause: the complex endocrinology of the menopausal transition". Endocrine Reviews 19 (4): 397–428. August 1998. doi:10.1210/edrv.19.4.0341. PMID 9715373.
- ↑ 57.0 57.1 "Diagnostic role of follicle-stimulating hormone (FSH) measurements during the menopausal transition—an analysis of FSH, oestradiol and inhibin". European Journal of Endocrinology 130 (1): 38–42. January 1994. doi:10.1530/eje.0.1300038. PMID 8124478.
- ↑ 58.0 58.1 "Inhibin B and ovarian function after total abdominal hysterectomy in women of reproductive age". Gynecological Endocrinology 17 (2): 125–31. April 2003. doi:10.1080/713603218. PMID 12737673.
- ↑ 59.0 59.1 "Effect of total abdominal hysterectomy on ovarian blood supply in women of reproductive age". Journal of Ultrasound in Medicine 24 (2): 169–74. February 2005. doi:10.7863/jum.2005.24.2.169. PMID 15661947.
- ↑ "Minireview: aromatase and the regulation of estrogen biosynthesis—some new perspectives". Endocrinology 142 (11): 4589–94. November 2001. doi:10.1210/endo.142.11.8547. PMID 11606422.
- ↑ "Androgen levels in adult females: changes with age, menopause, and oophorectomy". The Journal of Clinical Endocrinology and Metabolism 90 (7): 3847–53. July 2005. doi:10.1210/jc.2005-0212. PMID 15827095.
- ↑ "Ovarian androgen production in postmenopausal women". The Journal of Clinical Endocrinology and Metabolism 92 (8): 3040–3. August 2007. doi:10.1210/jc.2007-0581. PMID 17519304.
- ↑ "The kisspeptin-GnRH pathway in human reproductive health and disease". Human Reproduction Update 20 (4): 485–500. 2014. doi:10.1093/humupd/dmu009. PMID 24615662.
- ↑ 64.0 64.1 "Impairment of BRCA1-related DNA double-strand break repair leads to ovarian aging in mice and humans". Science Translational Medicine 5 (172): 172ra21. February 2013. doi:10.1126/scitranslmed.3004925. PMID 23408054.
- ↑ "The evolution of meiotic sex and its alternatives". Proceedings of the Royal Society B: Biological Sciences (Proc Biol Sci) 283 (1838). September 2016. doi:10.1098/rspb.2016.1221. PMID 27605505.
- ↑ "Constructing a standard climacteric scale". Maturitas 29 (1): 25–31. May 1998. doi:10.1016/s0378-5122(98)00025-5. PMID 9643514.
- ↑ "Quality of life in a large cohort of mid-aged Colombian women assessed using the Cervantes Scale". Menopause 19 (8): 924–30. August 2012. doi:10.1097/gme.0b013e318247908d. PMID 22549166.
- ↑ "Factors related to increased daytime sleepiness during the menopausal transition as evaluated by the Epworth sleepiness scale". Maturitas 65 (1): 75–80. January 2010. doi:10.1016/j.maturitas.2009.11.003. PMID 19945237.
- ↑ 69.0 69.1 "What Is Perimenopause?". WebMD. https://www.webmd.com/menopause/guide/guide-perimenopause#1.
- ↑ "Perimenopause – Symptoms and causes". Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/perimenopause/symptoms-causes/syc-20354666.
- ↑ "Menopause 101". A primer for the perimenopausal. The North American Menopause Society. http://www.menopause.org/for-women/menopauseflashes/menopause-101-a-primer-for-the-perimenopausal.
- ↑ 72.0 72.1 72.2 "Perimenopause". Centre for Menstrual Cycle and Ovulation Research (CeMCOR). http://www.cemcor.ca/help_yourself/perimenopause.
- ↑ "Approaching menopause (but not there yet!): caring for women in midlife". Nursing for Women's Health 15 (4): 320–4. August–September 2011. doi:10.1111/j.1751-486X.2011.01652.x. PMID 21884497.
- ↑ Silver, Dr. Nazanin E. (2023). "Mood Changes During Perimenopause Are Real. Here's What to Know." (in en). https://www.acog.org/womens-health/experts-and-stories/the-latest/mood-changes-during-perimenopause-are-real-heres-what-to-know.
- ↑ "When depression sneaks up on menopause". Knowable Magazine. 27 January 2022. doi:10.1146/knowable-012722-1. https://knowablemagazine.org/article/mind/2022/when-depression-sneaks-menopause. Retrieved 17 May 2022.
- ↑ Disorders of menstruation. Chichester, West Sussex: Wiley-Blackwell. 2011. ISBN 978-1-4443-9181-7. https://books.google.com/books?id=wVkKVsCdNIkC&pg=PT247.
- ↑ 77.0 77.1 "In the clinic. Perimenopause". Annals of Internal Medicine 162 (3): ITC1–15. February 2015. doi:10.7326/AITC201502030. PMID 25643316.
- ↑ "Inevitable Menopause". http://ce.nurse.com/course/CE232-60/inevitable-menopause/.
- ↑ "Effects of melatonin in perimenopausal and menopausal women: our personal experience". Annals of the New York Academy of Sciences 1057 (1): 393–402. December 2005. doi:10.1196/annals.1356.030. PMID 16399909. Bibcode: 2005NYASA1057..393B.
- ↑ "Oestrogen and Progestin as Hormone Therapy". http://med.monash.edu.au/sphpm/womenshealth/docs/postmenopausal-hormone-therapy.pdf.
- ↑ North American Menopause Society (March 2010). "Estrogen and progestogen use in postmenopausal women: 2010 position statement of The North American Menopause Society". Menopause 17 (2): 242–255. doi:10.1097/gme.0b013e3181d0f6b9. PMID 20154637.
- ↑ North American Menopause Society (March 2012). "The 2012 hormone therapy position statement of: The North American Menopause Society". Menopause 19 (3): 257–71. doi:10.1097/GME.0000000000000921. PMID 22367731.
- ↑ "Vasomotor symptoms resulting from natural menopause: a systematic review and network meta-analysis of treatment effects from the National Institute for Health and Care Excellence guideline on menopause". BJOG 124 (10): 1514–1523. September 2017. doi:10.1111/1471-0528.14619. PMID 28276200. http://eprints.gla.ac.uk/138525/7/138525.pdf.
- ↑ "Hormone therapy for preventing cardiovascular disease in post-menopausal women". The Cochrane Database of Systematic Reviews 2015 (3). March 2015. doi:10.1002/14651858.CD002229.pub4. PMID 25754617.
- ↑ "The WHI: the effect of hormone replacement therapy on fracture prevention". Climacteric 15 (3): 263–6. June 2012. doi:10.3109/13697137.2012.659975. PMID 22612613.
- ↑ 86.0 86.1 "Long-term hormone therapy for perimenopausal and postmenopausal women". The Cochrane Database of Systematic Reviews 1 (1). January 2017. doi:10.1002/14651858.CD004143.pub5. PMID 28093732.
- ↑ "Menopausal hormone therapy and breast cancer mortality: clinical implications". Therapeutic Advances in Drug Safety 6 (2): 45–56. April 2015. doi:10.1177/2042098614568300. PMID 25922653.
- ↑ "hiện tượng bốc hỏa tiền mãn kinh". https://driphydration.vn/hien-tuong-boc-hoa-tien-man-kinh/.
- ↑ "Global Consensus Position Statement on the Use of Testosterone Therapy for Women". Climacteric 22 (5): 429–434. October 2019. doi:10.1080/13697137.2019.1637079. PMID 31474158.
- ↑ "Postmenopausal hormone therapy: from monkey glands to transdermal patches". The Journal of Endocrinology 185 (2): 207–22. May 2005. doi:10.1677/joe.1.05847. PMID 15845914.
- ↑ "The STAR trial: evidence for raloxifene as a breast cancer risk reduction agent for postmenopausal women". Journal of the National Comprehensive Cancer Network 5 (8): 719–24. September 2007. doi:10.6004/jnccn.2007.0073. PMID 17927929.
- ↑ 92.0 92.1 "Menopause". Primary Care. Women's Health 45 (4): 625–641. December 2018. doi:10.1016/j.pop.2018.08.001. PMID 30401346.
- ↑ "Use of mindfulness, meditation and relaxation to treat vasomotor symptoms". Climacteric 20 (2): 178–182. April 2017. doi:10.1080/13697137.2017.1283685. PMID 28286985.
- ↑ "Mindfulness, cognitive behavioural and behaviour-based therapy for natural and treatment-induced menopausal symptoms: a systematic review and meta-analysis". BJOG 126 (3): 330–339. February 2019. doi:10.1111/1471-0528.15153. PMID 29542222.
- ↑ "Non-hormonal treatments for menopausal symptoms". BMJ 359. November 2017. doi:10.1136/bmj.j5101. PMID 29170264.
- ↑ "Review of Efficacy of Complementary and Alternative Medicine Treatments for Menopausal Symptoms". Journal of Midwifery & Women's Health 62 (3): 286–297. May 2017. doi:10.1111/jmwh.12628. PMID 28561959.
- ↑ Peters, Brandilyn A (August 10, 2022). "Spotlight on the Gut Microbiome in Menopause: Current Insights". International Journal of Women's Health 14: 1059–1072. doi:10.2147/IJWH.S340491. PMID 35983178.
- ↑ Yu, S (2025). "Deciphering the influence of gut and oral microbiomes on menopause". Journal of Genetics and Genomics 52 (5): 601–614. doi:10.1016/j.jgg.2024.11.010. PMID 39577767. https://www.sciencedirect.com/science/article/pii/S1673852724003114. Retrieved October 15, 2025.
- ↑ Zhang, P (August 24, 2022). "Influence of Foods and Nutrition on the Gut Microbiome and Implications for Intestinal Health". International Journal of Molecular Sciences 23 (17): 9588. doi:10.3390/ijms23179588. PMID 36076980.
- ↑ British Dietetic Association (April 2025). "Eating well for the perimenopause and menopause". https://www.bda.uk.com/resource/menopause-diet.html.
- ↑ Barton, Fionnuala (21 July 2025). "Your Gut Microbiome in Menopause". https://www.fielddoctor.co.uk/health-hub/your-gut-microbiome-in-menopause.
- ↑ "Effects of herbal and dietary supplements on cognition in menopause: a systematic review". Maturitas 68 (3): 256–63. March 2011. doi:10.1016/j.maturitas.2010.12.005. PMID 21237589.
- ↑ 103.0 103.1 103.2 "Complementary and alternative therapies for the management of menopause-related symptoms: a systematic evidence review". Archives of Internal Medicine 166 (14): 1453–65. July 2006. doi:10.1001/archinte.166.14.1453. PMID 16864755.
- ↑ "Use of Plant-Based Therapies and Menopausal Symptoms: A Systematic Review and Meta-analysis". JAMA 315 (23): 2554–2563. June 2016. doi:10.1001/jama.2016.8012. PMID 27327802.
- ↑ "Black cohosh (Cimicifuga spp.) for menopausal symptoms". The Cochrane Database of Systematic Reviews 9 (9). September 2012. doi:10.1002/14651858.CD007244.pub2. PMID 22972105.
- ↑ "Acupuncture for menopausal hot flushes". The Cochrane Database of Systematic Reviews 7 (7). July 2013. doi:10.1002/14651858.CD007410.pub2. PMID 23897589.
- ↑ "Chinese herbal medicine for menopausal symptoms". The Cochrane Database of Systematic Reviews 3 (5). March 2016. doi:10.1002/14651858.CD009023.pub2. PMID 26976671.
- ↑ "Alendronate for the primary and secondary prevention of osteoporotic fractures in postmenopausal women". The Cochrane Database of Systematic Reviews (1). January 2008. doi:10.1002/14651858.CD001155.pub2. PMID 18253985.
- ↑ "Concerns over new 'menopause delay' procedure" (in en-GB). 2020-01-28. https://www.bbc.com/news/health-51269237.
- ↑ Evans, Subhadra (2024). "The need for biopsychosocial menopause care: a narrative review.". Menopause (New York, N.Y.) 31 (12): 1090–1096. doi:10.1097/GME.0000000000002441. PMID 39470610.
- ↑ "Attitudes toward menopause: The influence of sociocultural paradigms". Psychology of Women Quarterly 17 (3): 275–88. 1993. doi:10.1111/j.1471-6402.1993.tb00487.x.
- ↑ 112.0 112.1 "The impact of attitudes towards the menopause on women's symptom experience: a systematic review". Maturitas 65 (1): 28–36. January 2010. doi:10.1016/j.maturitas.2009.10.016. PMID 19954900.
- ↑ Brown, Lydia; Brown, Valerie; Judd, Fiona; Bryant, Christina (2018-10-02). "It's not as bad as you think: menopausal representations are more positive in postmenopausal women" (in en). Journal of Psychosomatic Obstetrics & Gynecology 39 (4): 281–288. doi:10.1080/0167482X.2017.1368486. ISSN 0167-482X. PMID 28937311. https://www.tandfonline.com/doi/full/10.1080/0167482X.2017.1368486.
- ↑ 114.0 114.1 Voicu, Ilona. "The social construction of menopause as disease: A literature review." Journal of Comparative Research in Anthropology and Sociology, vol. 9, no. 2 (Winter 2018): 71–82. ISSN 2068-0317. http://compaso.eu/wpd/wp-content/uploads/2019/02/Voicu_Journal-of-Comparative-Research-in-Anthropology-and-Sociology_vol9_issue2.pdf
- ↑ Williams, Makeba (2024). "Culturally responsive care for menopausal women". Maturitas 185. doi:10.1016/j.maturitas.2024.107995. PMID 38663163.
- ↑ 116.0 116.1 Bansal, Ramandeep, and Neelam Aggarwal. "Menopausal Hot Flashes: A Concise Review." Journal of Mid-Life Health 10, no. 1 (2019): 6–13. Available via EBSCO.
- ↑ 117.0 117.1 117.2 Martin, Emily. The Woman in the Body: A Cultural Analysis of Reproduction. Boston, MA: Beacon Press, 1987. Available via Alexander Street
- ↑ "Women's experience of menopause: a systematic review of qualitative evidence". JBI Database of Systematic Reviews and Implementation Reports 13 (8): 250–337. September 2015. doi:10.11124/jbisrir-2015-1948. PMID 26455946.
- ↑ "Influencing factors on women's attitudes toward menopause: a systematic review". Menopause 28 (10): 1192–1200. September 2021. doi:10.1097/GME.0000000000001833. PMID 34520416.
- ↑ "Is there a menopausal syndrome? Menopausal status and symptoms across racial/ethnic group". Social Science & Medicine 52 (3): 345–56. 2001. doi:10.1016/S0277-9536(00)00147-7. PMID 11330770.
- ↑ "Experiences of menopause, self-management strategies for menopausal symptoms and perceptions of health care among immigrant women: a systematic review". Climacteric 21 (2): 101–110. April 2018. doi:10.1080/13697137.2017.1421922. PMID 29345497.
- ↑ "Prevalence of menopausal symptoms in different ethnic groups of Asian women and responsiveness to therapy with three doses of conjugated estrogens/medroxyprogesterone acetate: the Pan-Asia Menopause (PAM) study". Maturitas 52 (3–4): 264–276. November 2005. doi:10.1016/j.maturitas.2005.03.012. PMID 15921865.
- ↑ "Cross-cultural study: experience, understanding of menopause, and related therapies in Australian and Laotian women". Menopause 19 (12): 1300–1308. December 2012. doi:10.1097/gme.0b013e31825fd14e. PMID 22929035.
- ↑ 124.0 124.1 "Menopause: lessons from anthropology". Psychosomatic Medicine 60 (4): 410–9. 1998. doi:10.1097/00006842-199807000-00005. PMID 9710286.
- ↑ "Factor analysis of climacteric symptoms in Japan". Maturitas 52 (3–4): 205–22. 2005. doi:10.1016/j.maturitas.2005.02.002. PMID 16154301.
- ↑ 126.0 126.1 "Chapter 4: Local Biologies and Human Difference". An Anthropology of Biomedicine. West Sussex: Wiley-Blackwell. 2010. pp. 84–89.
- ↑ "Lifestyle and demographic factors in relation to vasomotor symptoms: baseline results from the Study of Women's Health Across the Nation". American Journal of Epidemiology 159 (12): 1189–99. June 2004. doi:10.1093/aje/kwh168. PMID 15191936.
- ↑ "Female attitudes to menopause". Social Psychiatry 5: 35–40. 1970. doi:10.1007/BF01539794.
- ↑ "Menopause: social expectations, women's realities". Archives of Women's Mental Health 5 (1): 5–8. August 2002. doi:10.1007/s007370200016. PMID 12503068.
- ↑ Blackson, Emma A.; McCarthy, Christina; Bell, Caryn; Ramirez, Shokufeh; Bazzano, Alessandra N. (2024). "Experiences of menopausal transition among populations exposed to chronic psychosocial stress in the United States: A scoping review". BMC Women's Health 24 (1). doi:10.1186/s12905-024-03329-z. PMID 39232712.
- ↑ 131.0 131.1 Melby, Melissa K.; Lampl, Michelle (2011). "Menopause, A Biocultural Perspective". Annual Review of Anthropology 40: 53–70. doi:10.1146/annurev-anthro-081309-145641.
- ↑ Mohamed, Sophie; Hunter, Myra S. (2019). "Transgender women's experiences and beliefs about hormone therapy through and beyond mid-age: An exploratory UK study". International Journal of Transgenderism 20 (1): 98–107. doi:10.1080/15532739.2018.1493626. PMID 32999597.
- ↑ 133.0 133.1 133.2 133.3 Toze, Michael; Westwood, Sue (2025). "Experiences of menopause among non-binary and trans people". International Journal of Transgender Health 26 (2): 447–458. doi:10.1080/26895269.2024.2389924. PMID 40276004.
- ↑ "GP survey reveals health and healthcare inequalities of trans and non-binary adults | University of Cambridge" (in en). 2023-02-07. https://www.cam.ac.uk/research/news/gp-survey-reveals-health-and-healthcare-inequalities-of-trans-and-non-binary-adults.
- ↑ 135.0 135.1 135.2 135.3 135.4 135.5 Lock, Margaret (1988). "new Japanese mythologies: faltering discipline and the ailing housewife" (in en). American Ethnologist 15 (1): 43–61. doi:10.1525/ae.1988.15.1.02a00040. ISSN 1548-1425. https://onlinelibrary.wiley.com/doi/abs/10.1525/ae.1988.15.1.02a00040.
- ↑ 136.0 136.1 Beyene, Yewoubdar (1986). "Cultural significance and physiological manifestations of menopause a biocultural analysis". Culture, Medicine and Psychiatry 10 (1): 47–71. doi:10.1007/BF00053262. PMID 3698649. https://link.springer.com/article/10.1007/BF00053262. Retrieved 8 August 2025.
- ↑ Stewart, Donna E (2003). "Menopause in highland Guatemala Mayan women". Maturitas 44 (4): 293–297. doi:10.1016/s0378-5122(03)00036-7. PMID 12697370. https://www.maturitas.org/article/S0378-5122(03)00036-7/abstract. Retrieved 8 August 2025.
- ↑ Melby, Melissa K (2005). "Culture and Symptom Reporting At Menopause". Human Reproduction Update 11 (5): 495–512. doi:10.1093/humupd/dmi018. PMID 15919681. https://academic.oup.com/humupd/article-abstract/11/5/495/605995?redirectedFrom=fulltext&login=false#google_vignette. Retrieved 8 August 2025.
- ↑ 139.0 139.1 139.2 Davis, Susan R.; Magraith, Karen (2023-06-19). "Advancing menopause care in Australia: barriers and opportunities". Medical Journal of Australia 218 (11): 500–502. doi:10.5694/mja2.51981. ISSN 0025-729X. PMID 37330995.
- ↑ Jones, Emma K.; Jurgenson, Janelle R.; Katzenellenbogen, Judith M.; Thompson, Sandra C. (2012-12-12). "Menopause and the influence of culture: another gap for Indigenous Australian women?". BMC Women's Health 12. doi:10.1186/1472-6874-12-43. ISSN 1472-6874. PMID 23234340.
- ↑ Jones, Emma K.; Jurgenson, Janelle R.; Katzenellenbogen, Judith M.; Thompson, Sandra C. (2012-12-12). "Menopause and the influence of culture: another gap for Indigenous Australian women?". BMC women's health 12: 43. doi:10.1186/1472-6874-12-43. ISSN 1472-6874. PMID 23234340. PMC 3554544. https://pmc.ncbi.nlm.nih.gov/articles/PMC3554544/.
- ↑ "Work related stress, depression or anxiety". Health and Safety Executive (HSE). 31 October 2018. http://greeningconsultants.co.uk/wp-content/uploads/2019/03/HSE-Stats-2018.pdf.
- ↑ Griffiths, Amanda; S Hunter, Myra (2015). "Psychosocial factors and menopause: The impact of menopause on personal and working life". in C Davies, Sally. Annual Report of the Chief Medical Officer, 2014, The Health of the 51%: Women. Department of Health. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/595439/CMO_annual_report_2014.pdf.
- ↑ Faubion, Stephanie S.; Enders, Felicity; Hedges, Mary S.; Chaudhry, Rajeev; Kling, Juliana M.; Shufelt, Chrisandra L.; Saadedine, Mariam; Mara, Kristin et al. (2023-06-01). "Impact of Menopause Symptoms on Women in the Workplace". Mayo Clinic Proceedings 98 (6): 833–845. doi:10.1016/j.mayocp.2023.02.025. ISSN 0025-6196. PMID 37115119. https://www.sciencedirect.com/science/article/pii/S002561962300112X.
- ↑ Moore ADM |2022|The French Invention of Menopause and the Medicalisation of Women's Ageing, A History|Oxford University Press |page 85
- ↑ "Conceptual Layers in the Invention of Menopause in Nineteenth-Century France". French History 32 (2): 226–248. 2018. doi:10.1093/fh/cry006.
- ↑ 147.0 147.1 "Menopause in nonhuman primates?". Biology of Reproduction 79 (3): 398–406. September 2008. doi:10.1095/biolreprod.108.068536. PMID 18495681.
- ↑ "'Breakthrough moment': how Lorraine Kelly helped shift the menopause debate" (in en). 29 April 2022. https://www.theguardian.com/society/2022/apr/29/breakthrough-moment-how-celebrities-helped-shift-the-menopause-debate.
- ↑ "Davina McCall: Sex, Myths and the Menopause" (in en). https://www.channel4.com/programmes/davina-mccall-sex-myths-and-the-menopause.
- ↑ House of Commons (27 October 2021). "Menopause (Support and Services) Bill, House of Commons, Session 2021-22". https://bills.parliament.uk/bills/2897.
- ↑ McKinley, David B. (September 6, 2022). "H.R.8774 - Menopause Research Act of 2022". https://www.congress.gov/bill/117th-congress/house-bill/8774?s=1&r=13.
- ↑ Winkler, I.; Goncalves, A. (October 2023). "Do mammals have menopause?". Cell 186 (22): 4729–4733. doi:10.1016/j.cell.2023.09.026. PMID 37890455.
- ↑ 153.0 153.1 "Analyses of ovarian activity reveal repeated evolution of post-reproductive lifespans in toothed whales". Scientific Reports 8 (1). August 2018. doi:10.1038/s41598-018-31047-8. PMID 30150784. Bibcode: 2018NatSR...812833E.
- ↑ "Ecological knowledge, leadership, and the evolution of menopause in killer whales". Current Biology 25 (6): 746–750. March 2015. doi:10.1016/j.cub.2015.01.037. PMID 25754636. Bibcode: 2015CBio...25..746B.
- ↑ "Why did menopause evolve? New study of whales gives some clues". https://www.reuters.com/science/why-did-menopause-evolve-new-study-whales-gives-some-clues-2024-03-13/.
- ↑ "Evidence for reproductive senescence in female cetaceans.". Reports of the International Whaling Commission.. 1986. pp. 57–74. http://www.helenemarsh.com/publications/JournalPapers/1986/MarshKasuyaSenescence1986.pdf.
- ↑ "Menopause in female rhesus monkeys". American Journal of Primatology 35 (1): 59–71. 1995. doi:10.1002/ajp.1350350106. PMID 31924061.
- ↑ "Aging". Research on Nonhuman Primates. Advances in Veterinary Science and Comparative Medicine. 28. 1984. pp. 305–341. doi:10.1016/b978-0-12-039228-5.50015-2. ISBN 978-0-12-039228-5.
- ↑ "Aging and fertility patterns in wild chimpanzees provide insights into the evolution of menopause". Current Biology 17 (24): 2150–6. December 2007. doi:10.1016/j.cub.2007.11.033. PMID 18083515. Bibcode: 2007CBio...17.2150E.
- ↑ The Asian Elephant. Cambridge University Press. 1992. ISBN 978-0-521-43758-5. https://books.google.com/books?id=95MoRwdQlcYC&q=elephant+menopause&pg=PA179.
- ↑ "The evolution of senescence and post-reproductive lifespan in guppies (Poecilia reticulata)". PLOS Biology 4 (1). January 2006. doi:10.1371/journal.pbio.0040007. PMID 16363919.
- ↑ "How long is a cat in heat?". Animal Planet. 15 May 2012. http://www.animalplanet.com/pets/how-long-is-a-cat-in-heat/.
- ↑ Almansa, Juan Carranza (2022-11-16). "The evolutionary origins of menopause explained" (in en). http://theconversation.com/the-evolutionary-origins-of-menopause-explained-192543.
Further reading
- Rebecca Mead, "If You Can't Stand the Heat: Why menopause never gets old", The New Yorker, 10 March 2025, pp. 28–31. "If you've got ovaries, you'll go through it. Why do so many feel so blindsided?" (p. 28.)
External links
| Wikimedia Commons has media related to Menopause. |
- Menopause: MedlinePlus
- What Is Menopause?, National Institute on Aging
- Menopause & Me, The North American Menopause Society
| Classification | |
|---|---|
| External resources |
 |
