Chemistry:Vitamin D
| Vitamin D | |
|---|---|
| Drug class | |
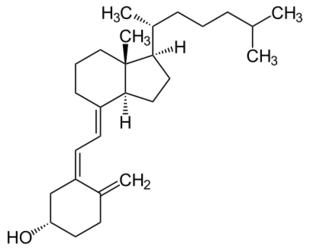 Cholecalciferol (D3) | |
| Class identifiers | |
| Synonyms | Calciferols |
| Use | Rickets, osteoporosis, osteomalacia, vitamin D deficiency |
| ATC code | A11CC |
| Biological target | vitamin D receptor |
| Clinical data | |
| Drugs.com | MedFacts Natural Products |
| External links | |
| MeSH | D014807 |
Vitamin D is a group of structurally related, fat-soluble compounds responsible for increasing intestinal absorption of calcium and phosphate, along with numerous other biological functions.[1][2] In humans, the most important compounds within this group are vitamin D3 (cholecalciferol) and vitamin D2 (ergocalciferol).[2][3]
Unlike the other twelve vitamins, vitamin D is only conditionally essential in the diet, as with adequate skin exposure to the ultraviolet B (UVB) radiation component of sunlight there is synthesis of cholecalciferol in the deeper layers of the skin's epidermis. Vitamin D can also be obtained through diet, food fortification and dietary supplements.[2] For most people, skin synthesis contributes more than dietary sources.[4] In the United States, milk and plant-based milk substitutes are fortified with vitamin D3, as are many breakfast cereals. US dietary guides generally assume that all of a person's vitamin D is taken orally, given the potential for insufficient sunlight exposure due to urban living, cultural choices for the amount of clothing worn when outdoors, and use of sunscreen because of concerns about safe sunlight exposure.
Cholecalciferol is converted in the liver to calcifediol (also known as calcidiol or 25-hydroxycholecalciferol), while ergocalciferol is converted to ercalcidiol (25-hydroxyergocalciferol). These two vitamin D metabolites, collectively referred to as 25-hydroxyvitamin D or 25(OH)D, are measured in serum to assess a person's vitamin D status. Calcifediol is further hydroxylated by the kidneys and certain immune cells to form calcitriol (1,25-dihydroxycholecalciferol; 1,25(OH)2D), the biologically active form of vitamin D.[3] Calcitriol attaches to vitamin D receptors, which are nuclear receptors found in various tissues throughout the body.
The discovery of the vitamin in 1922 was due to an effort to identify the dietary deficiency in children with rickets.[5][6] Adolf Windaus received the Nobel Prize in Chemistry in 1928 for his work on the constitution of sterols and their connection with vitamins.[7] Present day, government food fortification programs in some countries and recommendations to consume vitamin D supplements are intended to prevent or treat vitamin D deficiency rickets and osteomalacia. There are many other health conditions linked to vitamin D deficiency. However, the evidence for the health benefits of vitamin D supplementation in individuals who are already vitamin D sufficient is unproven.[2][8][9][10]
History


In northern European countries, cod liver oil had a long history of folklore medical uses, including applied to the skin and taken orally as a treatment for rheumatism and gout.[11] There were several extraction processes. Fresh livers cut to pieces and suspended on screens over pans of boiling water would drip oil that could be skimmed off the water, yielding a pale oil with a mild fish odor and flavor. For industrial purposes such as a lubricant, cod livers were placed in barrels to rot, with the oil skimmed off over months. The resulting oil was light to dark brown, and exceedingly foul smelling and tasting. In the 1800s, cod liver oil became popular as a bottled medicinal product for oral consumption – a teaspoon a day – with both pale and brown oils being used. The trigger for the surge in oral use was the observation made in several European countries—starting with Germany[12] in the 1820s and spreading to other countries into the 1860s—that young children fed cod liver oil did not develop rickets.[11] In northern Europe and the United States, the practice of giving children cod liver oil to prevent rickets persisted well in the 1950s. This overlapped with the fortification of cow's milk with vitamin D, which began in the early 1930s.[11]
Knowledge of cod liver oil being rickets-preventive in humans carried over to treating animals. In 1899, London surgeon John Bland-Sutton was asked to investigate why litters of lion cubs at the London Zoo were dying with a presentation that included rickets. He recommended that the diets of the pregnant and nursing females and the weaned cubs be switched from lean horse meat to goat – including calcium- and phosphorus-containing bones – and cod liver oil, solving the problem. Subsequently, researchers realized that animal models such as dogs and rats could be used for rickets research,[13] leading to the identification and naming of the responsible vitamin in 1922.[14]
In 1914, American researchers Elmer McCollum and Marguerite Davis had discovered a substance in cod liver oil which later was named "vitamin A".[5] Edward Mellanby, a British researcher, observed that dogs that were fed cod liver oil did not develop rickets, and (wrongly) concluded that vitamin A could prevent the disease. In 1922, McCollum tested modified cod liver oil in which the vitamin A had been destroyed. The modified oil cured the sick dogs, so McCollum concluded the factor in cod liver oil which cured rickets was distinct from vitamin A. He called it vitamin D because it was the fourth vitamin to be named.[5][15][16]
In 1925, it was established that when 7-dehydrocholesterol is irradiated with light, a form of a fat-soluble substance is produced, now known as vitamin D3.[5][6] Adolf Windaus, at the University of Göttingen in Germany, received the Nobel Prize in Chemistry in 1928 "for the services rendered through his research into the constitution of the sterols and their connection with the vitamins".[7] Alfred Fabian Hess, his research associate, stated: "Light equals vitamin D."[17] In 1932, Otto Rosenheim and Harold King published a paper putting forward structures for sterols and bile acids,[18] and soon thereafter collaborated with Kenneth Callow and others on the isolation and characterization of vitamin D.[19] Windaus further clarified the chemical structure of vitamin D.[20]
In 1969, a specific binding protein for vitamin D called the vitamin D receptor was identified.[21] Shortly thereafter, the conversion of vitamin D to calcifediol and then to calcitriol, the biologically active form, was confirmed.[22] The photosynthesis of vitamin D3 in skin via previtamin D3 and its subsequent metabolism was described in 1980.[23]
The discovery of vitamin D helped to increase the viability and prevalence of intensive animal farming. Prior to its discovery, mortality rates were higher whenever farm animals were moved indoors during winter. Being able to place vitamin D in the feed removed that issue and enabled placing a high number of animals in year-round indoor farming.[24]
Types
| Name | Chemical composition | Structure |
|---|---|---|
| Vitamin D1 | Mixture of molecular compounds of ergocalciferol with lumisterol, 1:1 | |
| Vitamin D2 | ergocalciferol (made from ergosterol) | 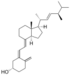
|
| Vitamin D3 | cholecalciferol
(made from 7-dehydrocholesterol in the skin). |
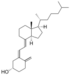
|
| Vitamin D4 | 22-dihydroergocalciferol | 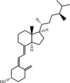
|
| Vitamin D5 | sitocalciferol
(made from 7-dehydrositosterol) |
|
Several forms (vitamers) of vitamin D exist, with the two major forms being vitamin D2 or ergocalciferol, and vitamin D3 or cholecalciferol.[1] The common-use term "vitamin D" refers to both D2 and D3, which were chemically characterized, respectively, in 1931 and 1935. Vitamin D3 was shown to result from the ultraviolet irradiation of 7-dehydrocholesterol. Although a chemical nomenclature for vitamin D forms was recommended in 1981,[25] alternative names remain commonly used.[3]
Chemically, the various forms of vitamin D are secosteroids, meaning that one of the bonds in the steroid rings is broken.[26] The structural difference between vitamin D2 and vitamin D3 lies in the side chain: vitamin D2 has a double bond between carbons 22 and 23, and a methyl group on carbon 24. Vitamin D analogues have also been synthesized.[3]
US dietary guides generally assume that all of a person's vitamin D is taken orally, given the potential for insufficient sunlight exposure due to urban living, cultural choices for the amount of clothing worn when outdoors, and use of sunscreen because of concerns about safe levels of sunlight exposure, including the risk of skin cancer.[2][27]: 362–394
Biology
The active vitamin D metabolite, calcitriol, exerts its biological effects by binding to the vitamin D receptor (VDR), which is primarily located in the nuclei of target cells.[1][26] When calcitriol binds to the VDR, it enables the receptor to act as a transcription factor, modulating the gene expression of transport proteins involved in calcium absorption in the intestine, such as TRPV6 and calbindin.[28] The VDR is part of the nuclear receptor superfamily of steroid hormone receptors, which are hormone-dependent regulators of gene expression. These receptors are expressed in cells across most organs. VDR expression decreases as age increases.[1][4]
Activation of VDR in the intestine, bone, kidney, and parathyroid gland cells plays a crucial role in maintaining calcium and phosphorus levels in the blood, a process that is assisted by parathyroid hormone and calcitonin, thereby supporting bone health.[1][29][4] VDR also regulates cell proliferation and differentiation. Additionally, vitamin D influences the immune system, with VDRs being expressed in several types of white blood cells, including monocytes and activated T and B cells.[30]
Deficiency
Worldwide, more than one billion people[31] – infants, children, adults and elderly[32] – can be considered vitamin D deficient, with reported percentages dependent on what measurement is used to define "deficient".[33] A 2023 systematic review in The Lancet Regional Health estimated that ~15% of the global population is vitamin D deficient when defined as serum 25(OH)D <30 nmol/L, with higher prevalence in regions with limited sunlight exposure.[34] Deficiency is common in the Middle-East,[32] Asia,[35] Africa[36] and South America,[37] but also exists in North America and Europe.[38][32][39][40] Dark-skinned populations in North America, Europe and Australia have a higher percentage of deficiency compared to light-skinned populations that had their origins in Europe.[41][42][43]
Serum 25(OH)D concentration is used as a biomarker for vitamin D deficiency. Units of measurement are either ng/mL or nmol/L, with one ng/mL equal to 2.5 nmol/L. There is no consensus on defining vitamin D deficiency, insufficiency, sufficiency, or optimal for all aspects of health.[33] According to the US Institute of Medicine Dietary Reference Intake Committee, below 30 nmol/L significantly increases the risk of vitamin D deficiency caused rickets in infants and young children and reduces absorption of dietary calcium from the normal range of 60–80% to as low as 15%, whereas above 40 nmol/L is needed to prevent osteomalacia bone loss in the elderly, and above 50 nmol/L to be sufficient for all health needs.[27]: 75–111 Other sources have defined deficiency as less than 25 nmol/L, insufficiency as 30–50 nmol/L[44] and optimal as greater than 75 nmol/L.[45][46] Part of the controversy is because studies have reported differences in serum levels of 25(OH)D between ethnic groups, with studies pointing to genetic as well as environmental reasons behind these variations. African-American populations have lower serum 25(OH)D than their age-matched white population, but at all ages have superior calcium absorption efficiency, a higher bone mineral density, and as elderly, a lower risk of osteoporosis and fractures.[27]: 439–440 Supplementation in this population to achieve proposed 'standard' concentrations could, in theory, cause harmful vascular calcification.[47]
Using the 25(OH)D assay as a screening tool of the generally healthy population to identify and treat individuals is considered not as cost-effective as a government-mandated fortification program. Instead, there is a recommendation that testing should be limited to those showing symptoms of vitamin D deficiency or who have health conditions known to cause vitamin deficiency.[4][40]
Causes
Causes of insufficient vitamin D synthesis in the skin include insufficient exposure to UVB light from sunlight due to living in high latitudes (farther distance from the equator with resultant shorter daylight hours in winter). Serum concentration by the end of winter can be lower by one-third to half that at the end of summer.[27]: 100–101, 371–379 [4][48] The prevalence of vitamin D deficiency increases with age due to a decrease in 7-dehydrocholesterol synthesis in the skin and a decline in kidney capacity to convert calcidiol to calcitriol,[49] the latter seen to a greater degree in people with chronic kidney disease.[50] Despite these age effects, elderly people can still synthesize sufficient calcitriol if enough skin is exposed to UVB light. Absent that, a dietary supplement is recommended.[49] Other causes of insufficient synthesis are sunlight being blocked by air pollution,[51] urban/indoor living, long-term hospitalizations and stays in extended care facilities, cultural or religious lifestyle choices that favor sun-blocking clothing, recommendations to use sun-blocking clothing or sunscreen to reduce risk of skin cancer, and lastly, the UV-B blocking nature of dark skin.[39]
Consumption of foods that naturally contain vitamin D is rarely sufficient to maintain a recommended serum concentration of 25(OH)D in the absence of the contribution of skin synthesis. Fractional contributions are roughly 20% diet and 80% sunlight.[4] Vegans had a lower dietary intake of vitamin D and lower serum 25(OH)D when compared to omnivores, with lacto-ovo-vegetarians falling in between due to the vitamin content of egg yolks and fortified dairy products.[52] Governments have mandated or voluntary food fortification programs to bridge the difference in, respectively, 15 and 10 countries.[53] The US is one of the few mandated countries. The original fortification practices, circa the early 1930s, were limited to cow's milk, which had a large effect on reducing infant and child rickets. In July 2016 the US Food and Drug Administration approved the addition of vitamin D to plant milk beverages intended as milk alternatives, such as beverages made from soy, almond, coconut, and oats.[54] At an individual level, people may choose to consume a multivitamin/mineral product or else a vitamin-D-only product.[55]
There are many disease states, medical treatments, and medications that put people at risk for vitamin D deficiency. Chronic diseases that increase risk include kidney[50] and liver failure, Crohn's disease, inflammatory bowel disease, and malabsorption syndromes such as cystic fibrosis, and hyper- or hypo-parathyroidism.[39] Obesity sequesters vitamin D in fat tissues, thereby lowering serum levels,[56] but bariatric surgery to treat obesity interferes with dietary vitamin D absorption, also causing deficiency.[57] Medications interacting with vitamin D metabolism include antiretrovirals, anti-seizure drugs, glucocorticoids, systemic antifungals such as ketoconazole, cholestyramine, and rifampicin.[4][39] Organ transplant recipients receive immunosuppressive therapy that is associated with an increased risk to develop skin cancer, so they are advised to avoid sunlight exposure, and to take a vitamin D supplement.[58]
Treatment
Daily dose regimens are preferred to admission of large doses at weekly or monthly schedules, and D3 may be preferred over D2, but there is a lack of consensus as to optimal type, dose, duration or what to measure to deem success. Daily regimens on the order of 4,000 IU/day (for other than infants) have a greater effect on 25(OH)D recovery from deficiency and a lower risk of side effects compared to weekly or monthly bolus doses, with the latter as high as 100,000 IU. The only advantage of bolus dosing could be better compliance, as bolus dosing is usually administered by a healthcare professional rather than self-administered.[4] While some studies have found that vitamin D3 raises 25(OH)D blood levels faster and remains active in the body longer,[59][60] others contend that vitamin D2 sources are equally bioavailable and effective for raising and sustaining 25(OH)D.[61][62] If digestive disorders compromise absorption, then intramuscular injection of up to 100,000 IU of vitamin D3 is therapeutic.[4]
Dark skin as deficiency risk
Melanin, specifically the sub-type eumelanin, is a biomolecule consisting of linked molecules of oxidized amino acid tyrosine. It is produced by cells called melanocytes in a process called melanogenesis. In the skin, melanin is located in the bottom layer (the stratum basale) of the skin's epidermis. Melanin can be permanently incorporated into the skin, resulting in dark skin, or else have its synthesis initiated by exposure to UV radiation, causing the skin to darken as a temporary sun tan. Eumelanin is an effective absorbent of light; the pigment can dissipate over 99.9% of absorbed UV radiation.[63] Because of this property, eumelanin is thought to protect skin cells from sunlight's ultraviolet A (UVA) and ultraviolet B (UVB) radiation damage, reducing the risk of skin tissue folate depletion, preventing premature skin aging and reducing the risks of sunburn and skin cancer.[64] Melanin inhibits UVB-powered vitamin D synthesis in the skin. In areas of the world not distant from the equator, abundant, year-round exposure to sunlight means that even dark-skinned populations have adequate skin synthesis.[65] However, when dark-skinned people cover much of their bodies with clothing for cultural or climate reasons, or are living a primarily indoor life in urban conditions, or live at higher latitudes which provide less sunlight in winter, they are at risk for vitamin D deficiency.[39][66] The last cause has been described as a "latitude-skin color mismatch".[65]
In the United States, vitamin D deficiency is particularly common among non-white Hispanic and African-American populations.[44][65][67] However, despite having on-average 25(OH)D serum concentrations below the 50 nmol/L amount considered sufficient, African Americans have higher bone mineral density and lower fracture risk when compared to European-origin people. Possible mechanisms may include higher calcium retention, lower calcium excretion, and greater bone resistance to parathyroid hormone,[65][67][68] also genetically lower serum vitamin D-binding protein which would result in adequate bioavailable 25(OH)D despite total serum 25(OH)D being lower.[69] The bone density and fracture risk paradox does not necessarily carry over to non-skeletal health conditions such as arterial calcification, cancer, diabetes or all-cause mortality. There is conflicting evidence that in the African American population, 'deficiency' as currently defined increases the risk of non-skeletal health conditions, and some evidence that supplementation increases risk,[65][67] including for harmful vascular calcification.[47] African Americans, and by extension other dark-skinned populations, may need different definitions for vitamin D deficiency, insufficiency, and adequate.[47]
Infant deficiency
Comparative studies carried out in lactating mothers indicate a mean value of vitamin D content in the breast milk of 45 IU/liter.[70] This vitamin D content is too low to meet the vitamin D requirement of 400 IU/day recommended by several government organizations ("...as breast milk is not a meaningful source of vitamin D."[27]: 385 ). The same government organizations recommend that lactating women consume 600 IU/day,[2][71][72][73] but this is insufficient to raise breast milk content to deliver recommended intake.[70] There is evidence that breast milk content can be increased, but because the transfer of the vitamin from the lactating mother's serum to milk is inefficient, this requires that she consume a dietary supplement above the government-set safe upper limit of 4,000 IU/day.[70] Given the shortfall, there are recommendations that breast-fed infants be fed a vitamin D dietary supplement of 400 IU/day during the first year of life.[70] If not breastfeeding, infant formulas are designed to deliver 400 IU/day for an infant consuming a liter of formula per day[74] – a normal volume for a full-term infant after the first month.[75]
Excess
Vitamin D toxicity, or hypervitaminosis D, is the toxic state of an excess of vitamin D. It is rare, having occurred historically during a time of unregulated fortification of foods, especially those provided to infants,[27]: 431–432 or more recently, with consumption of high-dose vitamin D dietary supplements following inappropriate prescribing, non-prescribed consumption of high-dose, over-the-counter preparations, or manufacturing errors resulting in content far in excess of what is on the label.[55][76][77] Ultraviolet light alone – sunlight or tanning beds – can raise serum 25(OH)D concentration to a bit higher than 100 nmol/L, but not to a level that causes hypervitaminosis D, the reasons being that there is a limiting amount of the precursor 7-dehydrocholesterol synthesized in the skin and a negative feedback in the kidney wherein the presence of calcitriol induces diversion to metabolically inactive 24,25-hydroxyvitamin D rather than metabolically active calcitriol (1,25-hydroxyvitamin D).[78] Further metabolism yields calcitroic acid, an inactive water-soluble compound that is excreted in bile.[79]
There is no general agreement about the intake levels at which vitamin D may cause harm. According to the IOM review, "Doses below 10,000 IU/day are not usually associated with toxicity, whereas doses equal to or above 50,000 IU/day for several weeks or months are frequently associated with toxic side effects including documented hypercalcemia."[27]: 427 The normal range for blood concentration of 25-hydroxyvitamin D in adults is 20 to 50 nanograms per milliliter (ng/mL; equivalent to 50 to 125 nmol/L). Blood levels necessary to cause adverse effects in adults are thought to be greater than about 150 ng/mL.[27]: 424–446
An excess of vitamin D causes hypercalcaemia (high blood concentrations of calcium), which can cause overcalcification of the bones and soft tissues including arteries, heart, and kidneys. Untreated, this can lead to irreversible kidney failure. Symptoms of vitamin D toxicity may include the following: increased thirst, increased urination, nausea, vomiting, diarrhea, decreased appetite, irritability, constipation, fatigue, muscle weakness, and insomnia.[80][81][82]
In 2011, the US National Academy of Medicine revised tolerable upper intake levels (UL) to protect against vitamin D toxicity. Before the revision the UL for ages 9+ years was 50 μg/d (2000 IU/d).[27]: 424–445 Per the revision: "UL is defined as "the highest average daily intake of a nutrient that is likely to pose no risk of adverse health effects for nearly all persons in the general population".[83] The US ULs in micrograms (mcg or μg) and international units (IU) for both males and females, by age, are:
- 0–6 months: 25 μg/d (1000 IU/d)
- 7–12 months: 38 μg/d (1500 IU/d)
- 1–3 years: 63 μg/d (2500 IU/d)
- 4–8 years: 75 μg/d (3000 IU/d)
- 9+ years: 100 μg/d (4000 IU/d)
- Pregnant and lactating: 100 μg/d (4000 IU/d)
Although in the US the adult UL is set at 4,000 IU/day, over-the-counter products are available at 5,000, 10,000 and even 50,000 IU (the last with directions to take once a week). The percentage of the US population taking over 4,000 IU/day has increased since 1999.[55]
Treatment
In almost every case, stopping the vitamin D supplementation combined with a low-calcium diet and corticosteroid drugs will allow for a full recovery within a month.[80][81][82]
Special cases
Idiopathic infantile hypercalcemia is caused by a mutation of the CYP24A1 gene, leading to a reduction in the degradation of vitamin D. Infants who have such a mutation have an increased sensitivity to vitamin D and in case of additional intake a risk of hypercalcaemia.[84] The disorder can continue into adulthood.[85]
Health effects
Supplementation with vitamin D is a reliable method for preventing or treating rickets. On the other hand, the effects of vitamin D supplementation on non-skeletal health are uncertain.[86][87] A review did not find any effect from supplementation on the rates of non-skeletal disease, other than a tentative decrease in mortality in the elderly.[88] Vitamin D supplements do not alter the outcomes for myocardial infarction, stroke or cerebrovascular disease, cancer, bone fractures or knee osteoarthritis.[9][89]
A US Institute of Medicine (IOM) report states: "Outcomes related to cancer, cardiovascular disease and hypertension, and diabetes and metabolic syndrome, falls and physical performance, immune functioning and autoimmune disorders, infections, neuropsychological functioning, and preeclampsia could not be linked reliably with intake of either calcium or vitamin D, and were often conflicting."[27]: 5 Evidence for and against each disease state is provided in detail.[27]: 124–299 Some researchers claim the IOM was too definitive in its recommendations and made a mathematical mistake when calculating the blood level of vitamin D associated with bone health.[90] Members of the IOM panel maintain that they used a "standard procedure for dietary recommendations" and that the report is solidly based on the data.[90]
Mortality, all-causes
Vitamin D3 supplementation has been tentatively found to lead to a reduced risk of death in the elderly,[91][88] but the effect has not been deemed pronounced, or certain enough, to make taking supplements recommendable.[9] Other forms (vitamin D2, alfacalcidol, and calcitriol) do not appear to have any beneficial effects concerning the risk of death.[91] High blood levels appear to be associated with a lower risk of death, but it is unclear if supplementation can result in this benefit.[92] Both an excess and a deficiency in vitamin D appear to cause abnormal functioning and premature aging.[93][94] The relationship between serum calcifediol concentrations and all-cause mortality is "U-shaped": mortality is elevated at high and low calcifediol levels, relative to moderate levels. Harm from elevated calcifediol appears to occur at a lower level in dark-skinned Canadian and American populations than in light-skinned populations.[27]: 424–435
Bone health
Rickets

Rickets, a childhood disease, is characterized by impeded growth and soft, weak, deformed long bones that bend and bow under their weight as children start to walk. Maternal vitamin D deficiency can cause fetal bone defects from before birth and impairment of bone quality after birth.[95][96] Rickets typically appears between 3 and 18 months of age.[97] This condition can be caused by vitamin D, calcium or phosphorus deficiency.[98] Vitamin D deficiency remains the main cause of rickets among young infants in most countries because breast milk is low in vitamin D, and darker skin, social customs, and climatic conditions can contribute to inadequate sun exposure.[citation needed] A post-weaning Western omnivore diet characterized by high intakes of meat, fish, eggs and vitamin-D–fortified milk is protective, whereas low intakes of those foods and high cereal/grain intake contribute to risk.[99][100][101] For young children with rickets, supplementation with vitamin D plus calcium was superior to the vitamin alone for bone healing.[102][103]
Osteomalacia and osteoporosis

Characteristics of osteomalacia are softening of the bones, leading to bending of the spine, bone fragility, and increased risk for fractures.[1] Osteomalacia is usually present when 25-hydroxyvitamin D levels are less than about 10 ng/mL.[105] Osteomalacia progress to osteoporosis, a condition of reduced bone mineral density with increased bone fragility and risk of bone fractures. Osteoporosis can be a long-term effect of calcium and/or vitamin D insufficiency, the latter contributing by reducing calcium absorption.[2] In the absence of confirmed vitamin D deficiency there is no evidence that vitamin D supplementation without concomitant calcium slows or stops the progression of osteomalacia to osteoporosis.[8] For older people with osteoporosis, taking vitamin D with calcium may help prevent hip fractures, but it also slightly increases the risk of stomach and kidney problems.[106][107] The reduced risk for fractures is not seen in healthier, community-dwelling elderly.[9][108][109] Low serum vitamin D levels have been associated with falls,[110] but taking extra vitamin D does not appear to reduce that risk.[111]
Athletes who are vitamin D deficient are at an increased risk of stress fractures and/or major breaks, particularly those engaging in contact sports. Incremental decreases in risk are observed with rising serum 25(OH)D concentrations plateauing at 50 ng/mL with no additional benefits seen in levels beyond this point.[112]
Cancer
While serum low 25-hydroxyvitamin D status has been associated with a higher risk of cancer in observational studies,[113][114][115] the general conclusion is that there is insufficient evidence for an effect of vitamin D supplementation on the risk of cancer,[2][116][117] although there is some evidence for reduction in cancer mortality.[113][118]
Cardiovascular disease
Vitamin D supplementation is not associated with a reduced risk of stroke, cerebrovascular disease, myocardial infarction, or ischemic heart disease.[9][119][120] Supplementation does not lower blood pressure in the general population.[121][122][123] One meta-analysis found a small increase in risk of stroke when calcium and vitamin D supplements were taken together.[124]
Immune system
Vitamin D receptors are found in cell types involved in immunity. Functions are not understood. Some autoimmune and infectious diseases are associated with vitamin D deficiency, but either there is no evidence that supplementation has a benefit or not, or for some, evidence indicating there are no benefits.[125][126][127][128]
Autoimmune diseases
Low plasma vitamin D concentrations have been reported for autoimmune thyroid diseases,[129] lupus,[130] myasthenia gravis,[131] rheumatoid arthritis,[132] and multiple sclerosis.[133] For multiple sclerosis and rheumatoid arthritis, intervention trials using vitamin D supplementation did not demonstrate therapeutic effects.[126][134]
Infectious diseases
A 2021 meta-analysis found that "vitamin D supplementation was safe and overall reduced the risk of ARI […] although the risk reduction was small".[135] In general, vitamin D functions to activate the innate and dampen the adaptive immune systems with antibacterial, antiviral and anti-inflammatory effects.[136][137] Low serum levels of vitamin D appear to be a risk factor for tuberculosis.[138] However, supplementation trials showed no benefit.[127][128]
Inflammatory bowel disease
Vitamin D deficiency has been linked to the severity of inflammatory bowel disease (IBD).[139] However, whether vitamin D deficiency causes IBD or is a consequence of the disease is not clear.[140] Supplementation leads to improvements in scores for clinical inflammatory bowel disease activity and biochemical markers,[140][141] and less frequent relapse of symptoms in IBD.[140]
Asthma
Vitamin D supplementation does not help prevent asthma attacks or alleviate symptoms.[142]
COVID-19
In July 2020, the US National Institutes of Health stated "There is insufficient evidence to recommend for or against using vitamin D supplementation for the prevention or treatment of COVID-19."[143] Same year, the UK National Institute for Health and Care Excellence (NICE) position was to not recommend to offer a vitamin D supplement to people solely to prevent or treat COVID-19.[144] NICE updated its position in 2022 to "Do not use vitamin D to treat COVID-19 except as part of a clinical trial."[145] Both organizations included recommendations to continue the previously established recommendations on vitamin D supplementation for other reasons, such as bone and muscle health, as applicable. Both organizations noted that more people may require supplementation due to lower amounts of sun exposure during the pandemic.[143][144]
Vitamin D deficiency and insufficiency have been associated with adverse outcomes in COVID-19.[146][147][148][149][150] Supplementation trials, mostly large, single, oral dose upon hospital admission, reported lower subsequent transfers to intensive care and to all-cause mortality.[151][152][153]
Other diseases and conditions
Chronic obstructive pulmonary disease
Meta Analysis has shown vitamin D supplementation did not influence the overall number of moderate or severe exacerbations of chronic obstructive pulmonary disease (COPD), but did reveal a protective effect in a subgroup with low vitamin D (baseline 25-hydroxyvitamin D levels <25 nmol/L).[154]
Diabetes
A meta-analysis reported that vitamin D supplementation significantly reduced the risk of type 2 diabetes for non-obese people with prediabetes.[155] Another meta-analysis reported that vitamin D supplementation significantly improved glycemic control [homeostatic model assessment-insulin resistance (HOMA-IR)], hemoglobin A1C (HbA1C), and fasting blood glucose (FBG) in individuals with type 2 diabetes.[156] In prospective studies, high versus low levels of vitamin D were respectively associated with a significant decrease in risk of type 2 diabetes, combined type 2 diabetes and prediabetes, and prediabetes.[157] A systematic review included one clinical trial that showed vitamin D supplementation together with insulin maintained levels of fasting C-peptide after 12 months better than insulin alone.[158]
Attention deficit hyperactivity disorder (ADHD)
A meta-analysis of observational studies showed that children with ADHD have lower vitamin D levels and that there was a small association between low vitamin D levels at the time of birth and later development of ADHD.[159] Several small, randomized controlled trials of vitamin D supplementation indicated improved ADHD symptoms such as impulsivity and hyperactivity.[160]
Depression
A 2014 systematic review concluded that vitamin D supplementation does not reduce depressive symptoms overall but may have a moderate benefit for patients with clinically significant depression, though more high-quality studies were determined to be needed.[161]
Cognition and dementia
A systematic review of clinical studies found an association between low vitamin D levels with cognitive impairment and a higher risk of developing Alzheimer's disease. However, lower vitamin D concentrations are also associated with poor nutrition and spending less time outdoors. Therefore, alternative explanations for the increase in cognitive impairment exist and hence a direct causal relationship between vitamin D levels and cognition could not be established.[162]
Schizophrenia
People diagnosed with schizophrenia tend to have lower serum vitamin D concentrations compared to those without the condition. This may be a consequence of the disease rather than a cause, due, for example, to low dietary vitamin D and less time spent exposed to sunlight.[163][164] Results from supplementation trials have been inconclusive.[163]
Sexual dysfunction
Erectile dysfunction can be a consequence of vitamin D deficiency. Mechanisms may include the regulation of vascular stiffness, the production of vasodilating nitric oxide, and the regulation of vessel permeability. However, the clinical trial literature does not yet contain sufficient evidence that supplementation treats the problem. Part of the complexity is that vitamin D deficiency is also linked to morbidities that are associated with erectile dysfunction, such as obesity, hypertension, diabetes mellitus, hypercholesterolemia, chronic kidney disease and hypogonadism.[165][166]
In women, vitamin D receptors are expressed in the superficial layers of the urogenital organs. There is an association between vitamin D deficiency and a decline in sexual functions, including sexual desire, orgasm, and satisfaction in women, with symptom severity correlated with vitamin D serum concentration. The clinical trial literature does not yet contain sufficient evidence that supplementation reverses these dysfunctions or improves other aspects of vaginal or urogenital health.[167]
Pregnancy
Pregnant women often do not take the recommended amount of vitamin D.[168] Low levels of vitamin D in pregnancy are associated with gestational diabetes, pre-eclampsia, and small for gestational age infants.[169] Although taking vitamin D supplements during pregnancy raises blood levels of vitamin D in the mother at term, the full extent of benefits for the mother or baby is unclear.[169][170][171]
Obesity
Obesity increases the risk of having low serum vitamin D. Supplementation does not lead to weight loss, but weight loss increases serum vitamin D. The theory is that fatty tissue sequesters vitamin D.[56] Bariatric surgery as a treatment for obesity can lead to vitamin deficiencies. Long-term follow-up reported deficiencies for vitamins D, E, A, K and B12, with D the most common at 36%.[57]
Uterine fibroids
There is evidence that the pathogenesis of uterine fibroids is associated with low serum vitamin D and that supplementation reduces the size of fibroids.[172][173]
Tooth decay
In one review, deficiency of vitamin D in children increased the risk of tooth decay by about 22%.[174]
Allowed health claims
Governmental regulatory agencies stipulate for the food and dietary supplement industries certain health claims as allowable as statements on packaging.
Europe: European Food Safety Authority (EFSA)
- normal function of the immune system[175]
- normal inflammatory response[175]
- normal muscle function[175]
- reduced risk of falling in people over age 60[176]
US: Food and Drug Administration (FDA)
- "Adequate calcium and vitamin D, as part of a well-balanced diet, along with physical activity, may reduce the risk of osteoporosis."[104]
Canada: Health Canada
- "Adequate calcium and regular exercise may help achieve strong bones in children and adolescents and reduce the risk of osteoporosis in older adults. An adequate intake of vitamin D is also necessary."[177]
Japan: Foods with Nutrient Function Claims (FNFC)
- "Vitamin D is a nutrient which promotes the absorption of calcium in the gut intestine and aids in the development of bone."[178]
Dietary intake
| United Kingdom | ||
| Age group | Intake (μg/day) | Maximum intake (μg/day)[71] |
|---|---|---|
| Breast-fed infants 0–12 months | 8.5–10 | 25 |
| Formula-fed infants (<500 mL/d) | 10 | 25 |
| Children 1 – 10 years | 10 | 50 |
| Children >10 and adults | 10 | 100 |
| United States | ||
| Age group | RDA (IU/day)[27] | (μg/day) |
| Infants 0–6 months | 400* | 10 |
| Infants 6–12 months | 400* | 10 |
| 1–70 years | 600 | 15 |
| Adults > 70 years | 800 | 20 |
| Pregnant/Lactating | 600 | 15 |
| Age group | Tolerable upper intake level (IU/day)[27] | (μg/day) |
| Infants 0–6 months | 1,000 | 25 |
| Infants 6–12 months | 1,500 | 37.5 |
| 1–3 years | 2,500 | 62.5 |
| 4–8 years | 3,000 | 75 |
| 9+ years | 4,000 | 100 |
| Pregnant/lactating | 4,000 | 100 |
| Canada | ||
| Age group | RDA (IU)[72] | Tolerable upper intake (IU)[72] |
| Infants 0–6 months | 400* | 1,000 |
| Infants 7–12 months | 400* | 1,500 |
| Children 1–3 years | 600 | 2,500 |
| Children 4–8 years | 600 | 3,000 |
| Children and adults 9–70 years | 600 | 4,000 |
| Adults > 70 years | 800 | 4,000 |
| Pregnancy & lactation | 600 | 4,000 |
| Australia and New Zealand | ||
| Age group | Adequate Intake (μg)[179] | Upper Level of Intake (μg)[179] |
| Infants 0–12 months | 5* | 25 |
| Children 1–18 years | 5* | 80 |
| Adults 19–50 years | 5* | 80 |
| Adults 51–70 years | 10* | 80 |
| Adults > 70 years | 15* | 80 |
| European Food Safety Authority | ||
| Age group | Adequate Intake (μg)[73] | Tolerable upper limit (μg)[180] |
| Infants 0–12 months | 10 | 25 |
| Children 1–10 years | 15 | 50 |
| Children 11–17 years | 15 | 100 |
| Adults | 15 | 100 |
| Pregnancy & Lactation | 15 | 100 |
| * Adequate intake, no RDA/RDI yet established | ||
Recommended levels
Various government institutions have proposed different recommendations for the amount of daily intake of vitamin D. These vary according to age, pregnancy, or lactation, and the extent assumptions are made regarding skin synthesis.[2][71][72][73][179] Older recommendations were lower. For example, the US Adequate Intake recommendations from 1997 were 200 IU/day for infants, children, adults to age 50, and women during pregnancy or lactation, 400 IU/day for ages 51–70, and 600 IU/day for 71 and older.[181]
Conversion: 1 μg (microgram) = 40 IU (international unit).[71] For dietary recommendation and food labeling purposes government agencies consider vitamin D3 and D2 bioequivalent.[27][71][72][73][179]
United Kingdom
The UK National Health Service (NHS) recommends that people at risk of vitamin D deficiency, breast-fed babies, formula-fed babies taking no more than 500 ml of infant formula a day, and children aged 6 months to 4 years, should take daily vitamin D supplements throughout the year to ensure sufficient intake.[71] This includes people with limited skin synthesis of vitamin D, who are not often outdoors, are frail, housebound, living in a care home, or usually wearing clothes that cover up most of the skin, or with dark skin, such as having an African, African-Caribbean or south Asian background. Other people may be able to make adequate vitamin D from sunlight exposure from April to September. The NHS and Public Health England recommend that everyone, including those who are pregnant and breastfeeding, consider taking a daily supplement containing 10 μg (400 IU) of vitamin D during autumn and winter because of inadequate sunlight for vitamin D synthesis.[182]
United States
The dietary reference intake for vitamin D issued in 2011 by the Institute of Medicine (IoM) (renamed National Academy of Medicine in 2015), superseded previous recommendations which were expressed in terms of adequate intake. The recommendations were formed assuming the individual has no skin synthesis of vitamin D because of inadequate sun exposure. The reference intake for vitamin D refers to total intake from food, beverages, and supplements, and assumes that calcium requirements are being met.[27]: 362–394 The tolerable upper intake level (UL) is defined as "the highest average daily intake of a nutrient that is likely to pose no risk of adverse health effects for nearly all persons in the general population".[27]: 424–446 Although ULs are believed to be safe, information on the long-term effects is incomplete and these levels of intake are not recommended for long-term consumption.[27]: 404 : 439–440
For US food and dietary supplement labeling purposes, the amount in a serving is expressed as a percent of Daily Value (%DV). For vitamin D labeling purposes, 100% of the daily value was 400 IU (10 μg), but in May 2016, it was revised to 800 IU (20 μg) to bring it into agreement with the recommended dietary allowance (RDA).[183][184] A table of the old and new adult daily values is provided at Reference Daily Intake.
Canada
Health Canada published recommended dietary intakes (DRIs) and tolerable upper intake levels (ULs) for vitamin D.[72]
Australia and New Zealand
Australia and New Zealand published nutrient reference values including guidelines for dietary vitamin D intake in 2006.[179] About a third of Australians have vitamin D deficiency.[185][186]
European Union
The European Food Safety Authority (EFSA) in 2016[73] reviewed the current evidence, finding the relationship between serum 25(OH)D concentration and musculoskeletal health outcomes is widely variable. They considered that average requirements and population reference intake values for vitamin D cannot be derived and that a serum 25(OH)D concentration of 50 nmol/L was a suitable target value. For all people over the age of 1, including women who are pregnant or lactating, they set an adequate intake of 15 μg/day (600 IU).[73]
On the other hand, the EU Commission defined nutrition labelling for foodstuffs as regards recommended daily allowances (RDA) for vitamin D to 5 μg/day (200 IU) as 100%.[187]
The EFSA reviewed safe levels of intake in 2012,[180] setting the tolerable upper limit for adults at 100 μg/day (4000 IU), a similar conclusion as the IOM.
The Swedish National Food Agency recommends a daily intake of 10 μg (400 IU) of vitamin D3 for children and adults up to 75 years, and 20 μg (800 IU) for adults 75 and older.[188]
Non-government organisations in Europe have made their own recommendations. The German Society for Nutrition recommends 20 μg.[189] The European Menopause and Andropause Society recommends postmenopausal women consume 15 μg (600 IU) until age 70, and 20 μg (800 IU) from age 71. This dose should be increased to 100 μg (4,000 IU) in some patients with very low vitamin D status or in case of co-morbid conditions.[190]
Food sources
Few foods naturally contain vitamin D. Cod liver oil as a dietary supplement contains 450 IU/teaspoon. Fatty fish (but not lean fish such as tuna) are the best natural food sources of vitamin D3. Beef liver, eggs, and cheese have modest amounts. Mushrooms provide variable amounts of vitamin D2, as mushrooms can be treated with UV light to greatly increase their content.[61][191] In certain countries, breakfast cereals, dairy milk and plant milk products are fortified. Infant formulas are fortified with 400 to 1000 IU per liter,[2][192] a normal volume for a full-term infant after the first month.[75] Cooking only minimally decreases vitamin content.[192]
| Food source[2] | Amount (IU / serving) |
|---|---|
| Trout (rainbow), farmed, cooked, 3 ounces | 645 |
| Salmon (sockeye), cooked, 3 ounces | 570 |
| Mushrooms, exposed to UV light, 1⁄2 cup (120 mL) | 366[61] |
| Mushrooms, not exposed to UV light, 1⁄2 cup (120 mL) | 7[61] |
| Milk, 2% milkfat, fortified, 1 cup | 120 |
| Plant milks, fortified, 1 cup | 100–144 |
| Ready-to-eat cereal, fortified, 1 serving | 80 |
| Egg, 1 large, scrambled | 44 |
| Liver, beef, cooked, 3 ounces | 42 |
| Cheese, cheddar, 1.5 ounce | 17 |
Fortification
In the early 1930s, the United States and countries in northern Europe began to fortify milk with vitamin D in an effort to eradicate rickets. This, plus medical advice to expose infants to sunlight, effectively ended the high prevalence of rickets. The proven health benefit of vitamin D led to fortification to many foods, even foods as inappropriate as hot dogs and beer. In the 1950s, due to some highly publicized cases of hypercalcemia and birth defects, vitamin D fortification became regulated, and in some countries discontinued.[48] As of 2024, governments have established mandated or voluntary food fortification programs to combat deficiency in, respectively, 15 and 10 countries.[53] Depending on the country,[53] manufactured foods fortified with either D2 or D3 may include dairy milk and other dairy foods, fruit juices and fruit juice drinks, meal replacement food bars, soy-protein–based beverages, wheat flour or corn meal products, infant formulas, breakfast cereals and 'plant milks',[54][193][38] the last described as beverages made from soy, almond, rice, oats and other plant sources intended as alternatives to dairy milk.[194]
Biosynthesis

Synthesis of vitamin D in nature is dependent on the presence of UV radiation and subsequent activation in the liver and the kidneys. Many animals synthesize vitamin D3 from 7-dehydrocholesterol, and many fungi synthesize vitamin D2 from ergosterol.[61]
Vitamin D3 is produced photochemically from 7-dehydrocholesterol in the skin of most vertebrate animals, including humans.[195] The skin consists of two primary layers: the inner layer called the dermis, and the outer, thinner epidermis. Vitamin D is produced in the keratinocytes of two innermost strata of the epidermis, the stratum basale and stratum spinosum, which also can produce calcitriol and express the vitamin D receptor.[196] The 7-dehydrocholesterol reacts with UVB light at wavelengths of 290–315 nm. These wavelengths are present in sunlight, as well as in the light emitted by the UV lamps in tanning beds (which produce ultraviolet primarily in the UVA spectrum, but typically produce 4% to 10% of the total UV emissions as UVB). Exposure to light through windows is insufficient because glass almost completely blocks UVB light.[197] In skin, either permanently in dark skin or temporarily due to tanning, melanin is located in the stratum basale, where it blocks UVB light and thus inhibits vitamin D synthesis.[63]
The transformation in the skin that converts 7-dehydrocholesterol to vitamin D3 occurs in two steps. First, 7-dehydrocholesterol is photolyzed by ultraviolet light in a 6-electron conrotatory ring-opening electrocyclic reaction; the product is previtamin D3. Second, previtamin D3 spontaneously isomerizes to vitamin D3 (cholecalciferol) via a [1,7]-sigmatropic hydrogen shift. In fungi, the conversion from ergosterol to vitamin D2 follows a similar procedure, forming previtamin D2 by UVB photolysis, which isomerizes to vitamin D2 (ergocalciferol).[4]
Interactive pathway
Click on View at the bottom to open. Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ↑ The interactive pathway map can be edited at WikiPathways: "VitaminDSynthesis_WP1531". http://www.wikipathways.org/index.php/Pathway:WP1531.
Evolution
For at least 1.2 billion years, eukaryotes – a classification of life forms that includes single-cell species, fungi, plants, and animals, but not bacteria – have been able to synthesize 7-dehydrocholesterol. When this molecule is exposed to UVB light from the sun it absorbs the energy in the process of being converted to vitamin D. The function was to prevent DNA damage, the converted molecule being an end product without vitamin function. Present day, phytoplankton in the ocean photosynthesize vitamin D without any calcium management function, as do some species of algae, lichen, fungi, and plants.[198][199][200] Only circa 500 million years ago, when animals began to leave the oceans for land, did the UV-converted molecule take on a hormone function as a promoter of calcium regulation. This function required the development of a nuclear vitamin D receptor (VDR) that binds the biologically active vitamin D metabolite 1α,25-dihydroxyvitamin (D3), plasma transport proteins, and vitamin D metabolizing CYP450 enzymes regulated by calciotropic hormones. The triumvirate of receptor protein, transport and metabolizing enzymes are found only in vertebrates.[201][202][203]
The initial vitamin function evolved for control of metabolic genes supporting innate and adaptive immunity. Only later did the VDR system start to function as an important regulator of calcium supply for a calcified skeleton in land-based vertebrates. From amphibians onward, bone management is biodynamic, with bone functioning as internal calcium reservoir under the control of osteoclasts via the combined action of parathyroid hormone and 1α,25-dihydroxyvitamin D3.[201][202][203]
Animal biosynthesis and food sources
Most land-based vertebrates – mammals, reptiles, birds, and amphibians – produce vitamin D in response to ultraviolet light. Carnivores and omnivores also get the vitamins from their diets, and herbivores can get some vitamins from fungi that are consumed along with plant foods.[204] In the wild, reptiles require either exposure to sunlight or consumption of prey, or both. In captivity, artificial lighting that provides UVB light is preferred to fortified food.[205] The same holds true for birds[206] and amphibians.[207] There are some exceptions. Feline species and dogs are practically incapable of vitamin D synthesis due to the high activity of 7-dehydrocholesterol reductase, which converts any 7-dehydrocholesterol in the skin to cholesterol before it can be UVB light-modified, but instead get vitamin D from diet.[208][209]
Fish do not synthesize vitamin D from exposure to ultraviolet light. Wild-caught fish obtain vitamin D via a diet of phytoplankton, zooplankton, and the aquatic food chain. Commercially raised fish are fed D3 fortified diets. As with land-based vertebrates, the vitamin is transported by vitamin D binding protein to cellular receptors. Aquaculture research shows that the vitamin is needed for bone health, optimizing growth, reducing fatty liver problems and supporting immune health. Unlike land-based vertebrates, large amounts of vitamin D3 are stored in the liver and fatty tissues, making fish a good dietary source for human consumption.[210]
Human evolution
During the long period between one and three million years ago, hominids, including ancestors of homo sapiens, underwent several evolutionary changes. A long-term climate shift toward drier conditions promoted life changes from sedentary forest-dwelling with a primarily plant-based diet toward upright walking/running on open terrain and more meat consumption.[211] One consequence of the shift to a culture that included more physically active hunting was a need for evaporative cooling from sweat, which to be functional, meant an evolutionary shift toward less body hair, as evaporation from sweat-wet hair would have cooled the hair but not the skin underneath.[212] A second consequence was darker skin.[211] The early humans who evolved in the regions of the globe near the equator had permanent large quantities of the skin pigment melanin in their skins, resulting in brown/black skin tones. For people with light skin tone, exposure to UV radiation induces the synthesis of melanin causing the skin to darken, i.e., sun tanning. Either way, the pigment can protect by dissipating up to 99.9% of absorbed UV radiation.[63] In this way, melanin protects skin cells from UVA and UVB radiation damage that causes photoaging and the risk of melanoma, a cancer of melanin cells.[213] Melanin also protects against photodegradation of the vitamin folate in skin tissue, and in the eyes, preserves eye health.[211]
The dark-skinned humans who had evolved in Africa populated the rest of the world through migration some 50,000 to 80,000 years ago.[214] Following settlement in northward regions of Asia and Europe which seasonally get less sunlight, the selective pressure for radiation-protective skin tone decreased while a need for efficient vitamin D synthesis in skin increased, resulting in low-melanin, lighter skin tones in the rest of the prehistoric world.[203][202][211] For people with low skin melanin, moderate sun exposure to the face, arms and lower legs several times a week is sufficient.[215] However, for recent cultural changes such as indoor living and working, UV-blocking skin products to reduce the risk of sunburn and emigration of dark-skinned people to countries far from the equator have all contributed to an increased incidence of vitamin D insufficiency and deficiency that need to be addressed by food fortification and vitamin D dietary supplements.[211]
Industrial synthesis
Vitamin D3 (cholecalciferol) is produced industrially by exposing 7-dehydrocholesterol to UVB and UVC light, followed by purification. The 7-dehydrocholesterol is sourced as an extraction from lanolin, a waxy skin secretion in sheep's wool.[216] Vitamin D2 (ergocalciferol) is produced in a similar way using ergosterol from yeast as a starting material.[216][217]
Metabolism
Activation

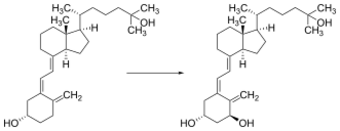
Whether synthesized in the skin or ingested, vitamin D is hydroxylated in the liver at position 25 (upper right of the molecule) to form the prohormone calcifediol, also referred to as 25(OH)D).[3] This reaction is catalyzed by the microsomal enzyme vitamin D 25-hydroxylase, the product of the CYP2R1 human gene.[218] Once made, the product is released into the blood where it is bound to vitamin D-binding protein.[219]
Calcifediol is transported to the proximal tubules of the kidneys, where it is hydroxylated at the 1-α position (lower right of the molecule) to form calcitriol (1,25-dihydroxycholecalciferol, also referred to as 1,25(OH)2D).[1] The conversion of calcifediol to calcitriol is catalyzed by the enzyme 25-hydroxyvitamin D3 1-alpha-hydroxylase, which is the product of the CYP27B1 human gene. The activity of CYP27B1 is increased by parathyroid hormone and also by low plasma calcium or phosphate.[1] Following the final converting step in the kidney, calcitriol is released into the circulation. By binding to vitamin D-binding protein, calcitriol is transported throughout the body.[26] In addition to the kidneys, calcitriol is also synthesized by certain other cells, including monocyte-macrophages in the immune system. When synthesized by monocyte-macrophages, calcitriol acts locally as a cytokine, modulating body defenses against microbial invaders by stimulating the innate immune system.[220]
Deactivation
The bioactivity of calcitriol is terminated by hydroxylation at position 24 by vitamin D3 24-hydroxylase, coded for by gene CYP24A1, forming calcitetrol.[3] Further metabolism yields calcitroic acid, an inactive water-soluble compound that is excreted in bile.[79]
Vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol) share a similar but not identical mechanism of action.[3] Metabolites produced by vitamin D2 are named with an er- or ergo- prefix to differentiate them from the D3-based counterparts (sometimes with a chole- prefix).[25]
- Metabolites produced from vitamin D2 tend to bind less well to the vitamin D-binding protein.[3]
- Vitamin D3 can alternatively be hydroxylated to calcifediol by sterol 27-hydroxylase, an enzyme coded for by gene CYP27A1, but vitamin D2 cannot.[3]
- Ergocalciferol can be directly hydroxylated at position 24 by the enzyme coded for by CYP27A1.[3] This hydroxylation also leads to a greater degree of inactivation: the activity of calcitriol decreases to 60% of original after 24-hydroxylation, whereas ercalcitriol undergoes a 10-fold decrease in activity on conversion to ercalcitetrol.
Mechanism of action
Calcitriol exerts its effects primarily by binding to the vitamin D receptor (VDR), which leads to the upregulation of gene transcription.[222] In the absence of calcitriol, the VDR is mainly located in the cytoplasm of cells. Calcitriol enters cells and binds to the VDR which forms a complex with its coreceptor RXR and the activated VDR/RXR complex is translocated into the nucleus.[220] The VDR/RXR complex subsequently binds to vitamin D response elements (VDRE) which are specific DNA sequences adjacent to genes, numbers estimated as being in the thousands. The VDR/RXR/DNA complex recruits other proteins that transcribe the downstream gene into mRNA which in turn is translated into protein causing a change in cell function.[3][65]
In addition to calcitriol, other vitamin D metabolites may contribute to vitamin D's biological effects. For example, CYP11A1, an enzyme chiefly known for its role in steroidogenesis, has been found to hydroxylate vitamin D3 at several positions, including C-20, C-22, and C-23, without cleaving the side chain. The resulting metabolites, such as 20-hydroxyvitamin D3 and 20,23-dihydroxyvitamin D3, act as inverse agonists for RORα and RORγ2. This interaction leads to effects such as the downregulation of IL-17 signaling, which influences the immune system.[223] Finally, some effects of vitamin D occur too rapidly to be explained by its influence on gene transcription. For example, calcitriol triggers rapid calcium uptake (within 1-10 minutes) in a variety of cells. These non-genomic actions may involve membrane-bound receptors like PDIA3.[224][225][226]
Genes regulated by the vitamin D receptor influence a wide range of physiological processes beyond calcium homeostasis and bone metabolism. They play a significant role in immune function, cellular signaling, and even blood coagulation, demonstrating the broad impact of vitamin D-regulated genes on human physiology.[227] Examples of these genes are outlined below.
Vitamin D receptor-regulated genes involved in vitamin D metabolism are CYP27B1, which encodes the enzyme that produces active vitamin D.[228][229] and CYP24A1, which encodes the enzyme responsible for degrading active vitamin D,[228][229] In the area of calcium homeostasis and bone metabolism, several genes are regulated by vitamin D. These include TNFSF11 (RANKL), crucial for bone metabolism;[228][230] SPP1 (Osteopontin), which is important for bone metabolism;[228][230] and BGLAP (Osteocalcin), which is involved in bone mineralization.[228][230] Additional genes include TRPV6, a calcium channel critical for intestinal calcium absorption;[227] S100G (Calbindin-D9k), a calcium-binding protein that facilitates calcium translocation in enterocytes;[227] ATP2B1 (PMCA1b), a plasma membrane calcium ATPase involved in calcium extrusion from the cell;[227] and the S100A family of genes, which encode calcium-binding proteins involved in various cellular processes.[228]
Vitamin D also plays a role in immune function, influencing genes such as CAMP (Cathelicidin Antimicrobial Peptide), which is involved in innate immune responses;[228][227] CD14, which participates in innate immune responses;[228] and HLA class II genes, which are important for adaptive immune function.[228][227] Cytokines such as IL2 and IL12, crucial for T cell responses, are also regulated by vitamin D.[231] In the domain of blood coagulation, vitamin D regulates the expression of THBD (Thrombomodulin), a key gene involved in the coagulation process.[228] Vitamin D also affects genes involved in cell differentiation and proliferation, including p21 and p27, which regulate the cell cycle,[232] as well as transcription factors such as c-fos and c-myc, which are involved in cell proliferation.[232]
Regulation

Negative feedback
Calcitriol plays a key role in regulating vitamin D levels through a negative feedback mechanism.[221] It strongly upregulates the expression of the enzyme CYP24A1, which inactivates vitamin D. This activation happens through binding of the activated vitamin D receptor (VDR) to two vitamin D response elements (VDREs) in the CYP24A1 gene. VDR also recruits proteins like histone acetyltransferases and RNA polymerase II to enhance this process. At the same time, calcitriol suppresses the production of CYP27B1, another enzyme involved in vitamin D metabolism, by modifying its gene's promoter region through an epigenetic mechanism. Together, these actions help tightly control vitamin D levels in the kidney.[221]
Hormonal
Vitamin D metabolism is regulated not only by the negative feedback mechanism of calcitriol but also by two hormones: parathyroid hormone (PTH) and fibroblast growth factor-23 (FGF-23). These hormones are essential for maintaining the body's calcium and phosphate balance.[221]
Parathyroid hormone (PTH) regulates serum calcium through its effects on bone, kidneys, and the small intestine.Bone remodeling, a constant process throughout life, involves bone mineral content being released by osteoclasts (bone resorption) and deposited by Added internal link to Osteoblast. PTH enhances the release of calcium from the large reservoir contained in the bones. It accomplishes this by binding to osteoblasts, in this way inhibiting the cells responsible for adding mineral content to bones, thus favoring the actions of osteoclasts.[234] In the kidneys, around 250 mmol of calcium ions are filtered into the glomerular filtrate per day, with the great majority reabsorbed and the remainder excreted in the urine.[235] PTH inhibits reabsorption of phosphate (HPO42−) by the kidneys, resulting in a decrease in plasma phosphate concentration. Given that phosphate ions form water-insoluble salts with calcium, a decrease in the phosphate concentration in plasma (for a given total calcium concentration) increases the amount of ionized (free) calcium.[234] A third important effect of PTH on the kidneys is stimulation of the conversion of 25-hydroxy vitamin D into 1,25-dihydroxy vitamin D (calcitriol).[234] This form of vitamin D is the active hormone which promotes calcium uptake from the intestine via the action of calbindin.[236] Calcitriol also reduces calcium loss to urine.[233]
Per the diagram, calcitriol suppresses the parathyroid hormone gene, thus creating a negative feedback loop that combines to tightly maintain plasma calcium in a normal range of 2.1-2.6 mmol/L for total calcium and 1.1-1.3 mmol/L for ionized calcium.[227] However, there are also vitamin D receptors in bone cells, so that with serum vitamin D in great excess, osteoclastic bone resorption is promoted regardless of PTH, resulting in hypercalcemia and its symptomology.[237]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 "Vitamin D". Micronutrient Information Center, Linus Pauling Institute, Oregon State University, Corvallis. 11 February 2021. https://lpi.oregonstate.edu/mic/vitamins/vitamin-D.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 "Vitamin D: Fact Sheet for Health Professionals". Office of Dietary Supplements, US National Institutes of Health. 26 July 2024. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 "Vitamin D metabolism, mechanism of action, and clinical applications". Chemistry & Biology 21 (3): 319–329. March 2014. doi:10.1016/j.chembiol.2013.12.016. PMID 24529992.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 "Consensus Statement on Vitamin D Status Assessment and Supplementation: Whys, Whens, and Hows". Endocrine Reviews 45 (5): 625–654. September 2024. doi:10.1210/endrev/bnae009. PMID 38676447.
- ↑ 5.0 5.1 5.2 5.3 "The discovery of vitamin D: the contribution of Adolf Windaus". The Journal of Nutrition 134 (6): 1299–302. June 2004. doi:10.1093/jn/134.6.1299. PMID 15173387.
- ↑ 6.0 6.1 "History of the discovery of vitamin D and its active metabolites". BoneKEy Reports 3: 479. January 2014. doi:10.1038/bonekey.2013.213. PMID 24466410.
- ↑ 7.0 7.1 "Adolf Windaus – Biography". Nobelprize.org. 25 March 2010. https://nobelprize.org/nobel_prizes/chemistry/laureates/1928/windaus-bio.html.
- ↑ 8.0 8.1 "Effects of vitamin D supplements on bone mineral density: a systematic review and meta-analysis". Lancet 383 (9912): 146–155. January 2014. doi:10.1016/s0140-6736(13)61647-5. PMID 24119980.
- ↑ 9.0 9.1 9.2 9.3 9.4 "The effect of vitamin D supplementation on skeletal, vascular, or cancer outcomes: a trial sequential meta-analysis". The Lancet. Diabetes & Endocrinology 2 (4): 307–320. April 2014. doi:10.1016/S2213-8587(13)70212-2. PMID 24703049.
- ↑ "The Lancet Diabetes & Endocrinology: Vitamin D supplementation in adults does not prevent fractures, falls or improve bone mineral density". https://www.eurekalert.org/news-releases/808633. "The authors conclude that there is therefore little reason to use vitamin D supplements to maintain or improve musculoskeletal health, except for the prevention of rare conditions such as rickets and osteomalacia in high-risk groups, which can be caused by vitamin D deficiency after long lack of exposure to sunshine."
- ↑ 11.0 11.1 11.2 "Vitamin D: part II; cod liver oil, ultraviolet radiation, and eradication of rickets". International Orthopaedics 43 (3): 735–749. March 2019. doi:10.1007/s00264-019-04288-z. PMID 30627846.
- ↑ Ihde, Aaron J. (1975). "Studies on the History of Rickets. II : The Roles of Cod Liver Oil and Light". Pharmacy in History 17 (1): 13–20. PMID 11609872. https://www.jstor.org/stable/41108885.
- ↑ "Metabolic bone disease in lion cubs at the London Zoo in 1889: the original animal model of rickets". J Biomed Sci 17 Suppl 1 (Suppl 1): S36. August 2010. doi:10.1186/1423-0127-17-S1-S36. PMID 20804612.
- ↑ "100 YEARS OF VITAMIN D: Historical aspects of vitamin D". Endocrine Connections 11 (4). April 2022. doi:10.1530/EC-21-0594. PMID 35245207.
- ↑ "Age-old children's disease back in force". Toronto Star. 25 July 2007. https://www.thestar.com/printarticle/239341.
- ↑ "Vitamin A, Vitamin D, Cartilage, Bones, and Teeth". Vitamins and Hormones. 21. Academic Press. January 1964. pp. 51–52. https://books.google.com/books?id=gGb7vm2SapcC. Retrieved 19 March 2023.
- ↑ "History of Vitamin D". University of California at Riverside. 2011. http://vitamind.ucr.edu/about/.
- ↑ "The Ring-system of sterols and bile acids. Part II". J. Chem. Technol. Biotechnol. 51 (47): 954–7. 1932. doi:10.1002/jctb.5000514702.
- ↑ "Crystalline Vitamin D". Proceedings of the Royal Society of London. Series B, Containing Papers of a Biological Character 109 (764): 488–506. 1932. doi:10.1098/rspb.1932.0008.
- ↑ "Industrial aspects of vitamin D". Vitamin D. Academic Press. 2011. p. 73. ISBN 978-0-12-387035-3. https://books.google.com/books?id=w7hMAFmsM84C. Retrieved 19 March 2023.
- ↑ "Chromosomal receptor for a vitamin D metabolite". Proceedings of the National Academy of Sciences of the United States of America 62 (1): 155–162. January 1969. doi:10.1073/pnas.62.1.155. PMID 5253652. Bibcode: 1969PNAS...62..155H.
- ↑ "1,25-dihydroxycholecalciferol: identification of the proposed active form of vitamin D3 in the intestine". Science 173 (3991): 51–54. July 1971. doi:10.1126/science.173.3991.51. PMID 4325863. Bibcode: 1971Sci...173...51N.
- ↑ "Photosynthesis of previtamin D3 in human skin and the physiologic consequences". Science 210 (4466): 203–205. October 1980. doi:10.1126/science.6251551. PMID 6251551. Bibcode: 1980Sci...210..203H.
- ↑ Torrella, Kenny (2023-02-10). "How a shipping error more than a century ago launched the $30 billion chicken industry" (in en-US). https://www.vox.com/future-perfect/2023/2/10/23589333/cecile-steele-chicken-meat-poultry-eggs-delaware.
- ↑ 25.0 25.1 "IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN): Nomenclature of vitamin D. Recommendations 1981". European Journal of Biochemistry 124 (2): 223–227. May 1982. doi:10.1111/j.1432-1033.1982.tb06581.x. PMID 7094913.
- ↑ 26.0 26.1 26.2 "Vitamin D". Present Knowledge in Nutrition (Eleventh ed.). London, United Kingdom: Academic Press (Elsevier). 2020. pp. 93–114. ISBN 978-0-323-66162-1.
- ↑ 27.00 27.01 27.02 27.03 27.04 27.05 27.06 27.07 27.08 27.09 27.10 27.11 27.12 27.13 27.14 27.15 27.16 27.17 Institute of Medicine (2011). Dietary Reference Intakes for Calcium and Vitamin D. The National Academies Collection: Reports funded by National Institutes of Health. National Academies Press. doi:10.17226/13050. ISBN 978-0-309-16394-1. https://www.ncbi.nlm.nih.gov/books/NBK56070/. Retrieved 17 September 2017.
- ↑ "Intestinal calcium absorption: Molecular vitamin D mediated mechanisms". Journal of Cellular Biochemistry 88 (2): 332–339. February 2003. doi:10.1002/jcb.10360. PMID 12520535.
- ↑ "Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease". The American Journal of Clinical Nutrition 80 (6 Suppl): 1678S–1688S. December 2004. doi:10.1093/ajcn/80.6.1678S. PMID 15585788.
- ↑ "An update on the association of vitamin D deficiency with common infectious diseases". Canadian Journal of Physiology and Pharmacology 93 (5): 363–368. May 2015. doi:10.1139/cjpp-2014-0352. PMID 25741906.
- ↑ "Vitamin D deficiency: a worldwide problem with health consequences". The American Journal of Clinical Nutrition 87 (4): 1080S–1086S. April 2008. doi:10.1093/ajcn/87.4.1080S. PMID 18400738.
- ↑ 32.0 32.1 32.2 "Is vitamin D deficiency a major global public health problem?". The Journal of Steroid Biochemistry and Molecular Biology 144 (Pt A): 138–145. October 2014. doi:10.1016/j.jsbmb.2013.11.003. PMID 24239505.
- ↑ 33.0 33.1 "Vitamin D: What's the "right" level?". 16 April 2020. https://www.health.harvard.edu/blog/vitamin-d-whats-right-level-2016121910893.
- ↑ Cashman, Kevin D. (2023). "Global prevalence of vitamin D deficiency: a systematic review and meta-analysis". The Lancet Regional Health – Europe 28 (6). doi:10.1016/j.lanepe.2022.100576. PMID 36573029.
- ↑ "High prevalence of vitamin D deficiency in Asia: A systematic review and meta-analysis". Critical Reviews in Food Science and Nutrition 63 (19): 3602–3611. 2023. doi:10.1080/10408398.2021.1990850. PMID 34783278.
- ↑ "Prevalence of vitamin D deficiency in Africa: a systematic review and meta-analysis". The Lancet. Global Health 8 (1): e134–e142. January 2020. doi:10.1016/S2214-109X(19)30457-7. PMID 31786117.
- ↑ "Prevalence of vitamin D deficiency in South America: a systematic review and meta-analysis". Nutrition Reviews 81 (10): 1290–1309. September 2023. doi:10.1093/nutrit/nuad010. PMID 36882047.
- ↑ 38.0 38.1 "Vitamin D: An overview of vitamin D status and intake in Europe". Nutrition Bulletin 39 (4): 322–350. December 2014. doi:10.1111/nbu.12108. PMID 25635171.
- ↑ 39.0 39.1 39.2 39.3 39.4 "Vitamin D deficiency 2.0: an update on the current status worldwide". European Journal of Clinical Nutrition 74 (11): 1498–1513. November 2020. doi:10.1038/s41430-020-0558-y. PMID 31959942.
- ↑ 40.0 40.1 "Optimisation of vitamin D status in global populations". Osteoporosis International 35 (8): 1313–1322. August 2024. doi:10.1007/s00198-024-07127-z. PMID 38836946.
- ↑ "Vitamin D deficiency in Europe: pandemic?". The American Journal of Clinical Nutrition 103 (4): 1033–1044. April 2016. doi:10.3945/ajcn.115.120873. PMID 26864360.
- ↑ "The prevalence of vitamin D deficiency among dark-skinned populations according to their stage of migration and region of birth: A meta-analysis". Nutrition 32 (1): 21–32. January 2016. doi:10.1016/j.nut.2015.07.007. PMID 26643747.
- ↑ "Special considerations for vitamin D in the south Asian population in the UK". Therapeutic Advances in Musculoskeletal Disease 9 (6): 137–144. June 2017. doi:10.1177/1759720X17704430. PMID 28620422.
- ↑ 44.0 44.1 "Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline". The Journal of Clinical Endocrinology and Metabolism 96 (7): 1911–1930. July 2011. doi:10.1210/jc.2011-0385. PMID 21646368.
- ↑ "Optimal Serum 25-Hydroxyvitamin D Levels for Multiple Health Outcomes". Sunlight, Vitamin D and Skin Cancer (Review). Advances in Experimental Medicine and Biology. 810. Springer. 2008. pp. 500–25. doi:10.1007/978-0-387-77574-6_5. ISBN 978-0-387-77573-9.
- ↑ "Plausible ergogenic effects of vitamin D on athletic performance and recovery". Journal of the International Society of Sports Nutrition 12. 2015. doi:10.1186/s12970-015-0093-8. PMID 26288575.
- ↑ 47.0 47.1 47.2 "Effect of race and genetics on vitamin D metabolism, bone and vascular health". Nature Reviews. Nephrology 8 (8): 459–466. June 2012. doi:10.1038/nrneph.2012.112. PMID 22688752.
- ↑ 48.0 48.1 "Sunlight and Vitamin D: A global perspective for health". Dermato-Endocrinology 5 (1): 51–108. January 2013. doi:10.4161/derm.24494. PMID 24494042.
- ↑ 49.0 49.1 "Vitamin D in the older population: a consensus statement". Endocrine 79 (1): 31–44. January 2023. doi:10.1007/s12020-022-03208-3. PMID 36287374.
- ↑ 50.0 50.1 "Vitamin D in Chronic Kidney Disease and Dialysis Patients". Nutrients 9 (4): 328. March 2017. doi:10.3390/nu9040328. PMID 28346348.
- ↑ "The impact of air pollutants, UV exposure and geographic location on vitamin D deficiency". Food and Chemical Toxicology 113: 241–254. March 2018. doi:10.1016/j.fct.2018.01.052. PMID 29409825.
- ↑ "Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review". Nutrients 14 (1): 29. December 2021. doi:10.3390/nu14010029. PMID 35010904.
- ↑ 53.0 53.1 53.2 "Map: Count of Nutrients In Fortification Standards". 2024. https://fortificationdata.org/map-number-of-nutrients/.
- ↑ 54.0 54.1 "Vitamin D for Milk and Milk Alternatives". Food and Drug Administration. 15 July 2016. https://www.fda.gov/food/food-additives-petitions/vitamin-d-milk-and-milk-alternatives.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 55.0 55.1 55.2 "Trends in Use of High-Dose Vitamin D Supplements Exceeding 1000 or 4000 International Units Daily, 1999-2014". JAMA 317 (23): 2448–2450. June 2017. doi:10.1001/jama.2017.4392. PMID 28632857.
- ↑ 56.0 56.1 "Vitamin D status and weight loss: a systematic review and meta-analysis of randomized and nonrandomized controlled weight-loss trials". The American Journal of Clinical Nutrition 104 (4): 1151–1159. October 2016. doi:10.3945/ajcn.116.136879. PMID 27604772.
- ↑ 57.0 57.1 "Long-term prevalence of vitamin deficiencies after bariatric surgery: a meta-analysis". Langenbeck's Archives of Surgery 409 (1). July 2024. doi:10.1007/s00423-024-03422-9. PMID 39030449.
- ↑ "Update: Solar UV Radiation, Vitamin D, and Skin Cancer Surveillance in Organ Transplant Recipients (OTRs)". Sunlight, Vitamin D and Skin Cancer. Adv Exp Med Biol. 1268. 2020. pp. 335–53. doi:10.1007/978-3-030-46227-7_17. ISBN 978-3-030-46226-0.
- ↑ "Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: a systematic review and meta-analysis". The American Journal of Clinical Nutrition 95 (6): 1357–1364. June 2012. doi:10.3945/ajcn.111.031070. PMID 22552031.
- ↑ "Vitamin D: deficiency, sufficiency and toxicity". Nutrients 5 (9): 3605–3616. September 2013. doi:10.3390/nu5093605. PMID 24067388.
- ↑ 61.0 61.1 61.2 61.3 61.4 "Photobiology of vitamin D in mushrooms and its bioavailability in humans". Dermato-Endocrinology 5 (1): 165–176. January 2013. doi:10.4161/derm.23321. PMID 24494050.
- ↑ "Vitamin D bioavailability: state of the art". Critical Reviews in Food Science and Nutrition 55 (9): 1193–1205. 2015. doi:10.1080/10408398.2012.688897. PMID 24915331. https://www.hal.inserm.fr/inserm-01478467/file/Vitamin%20D%20bioavailability%20HAL.pdf. Retrieved 27 April 2021.
- ↑ 63.0 63.1 63.2 "Radiative relaxation quantum yields for synthetic eumelanin". Photochemistry and Photobiology 79 (2): 211–216. February 2004. doi:10.1111/j.1751-1097.2004.tb00012.x. PMID 15068035.
- ↑ Understanding UVA and UVB, http://www.skincancer.org/prevention/uva-and-uvb/understanding-uva-and-uvb, retrieved 2012-04-30
- ↑ 65.0 65.1 65.2 65.3 65.4 65.5 "Does the High Prevalence of Vitamin D Deficiency in African Americans Contribute to Health Disparities?". Nutrients 13 (2): 499. February 2021. doi:10.3390/nu13020499. PMID 33546262.
- ↑ "Utility of sun-reactive skin typing and melanin index for discerning vitamin D deficiency". Pediatric Research 82 (3): 444–451. September 2017. doi:10.1038/pr.2017.114. PMID 28467404.
- ↑ 67.0 67.1 67.2 "The uncertain significance of low vitamin D levels in African descent populations: a review of the bone and cardiometabolic literature". Progress in Cardiovascular Diseases 56 (3): 261–269. 2013. doi:10.1016/j.pcad.2013.10.015. PMID 24267433.
- ↑ "Assessing Vitamin D Status in African Americans and the Influence of Vitamin D on Skeletal Health Parameters". Endocrinology and Metabolism Clinics of North America 46 (1): 135–152. March 2017. doi:10.1016/j.ecl.2016.09.006. PMID 28131129.
- ↑ "Vitamin D-binding protein and vitamin D status of black Americans and white Americans". The New England Journal of Medicine 369 (21): 1991–2000. November 2013. doi:10.1056/NEJMoa1306357. PMID 24256378.
- ↑ 70.0 70.1 70.2 70.3 "Pregnancy, Breastfeeding, and Vitamin D". International Journal of Molecular Sciences 24 (15). July 2023. doi:10.3390/ijms241511881. PMID 37569256.
- ↑ 71.0 71.1 71.2 71.3 71.4 71.5 "Vitamins and minerals – Vitamin D". 3 August 2020. https://www.nhs.uk/conditions/vitamins-and-minerals/vitamin-d/.
- ↑ 72.0 72.1 72.2 72.3 72.4 72.5 "Vitamin D and Calcium: Updated Dietary Reference Intakes". Nutrition and Healthy Eating. Health Canada. 5 December 2008. https://www.canada.ca/en/health-canada/services/food-nutrition/healthy-eating/vitamins-minerals/vitamin-calcium-updated-dietary-reference-intakes-nutrition.html.
- ↑ 73.0 73.1 73.2 73.3 73.4 73.5 EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA) (29 June 2016). "Dietary reference values for vitamin D". EFSA Journal 14 (10): e04547. doi:10.2903/j.efsa.2016.4547.
- ↑ "Comparison of the serum vitamin D level between breastfed and formula-fed infants: several factors which can affect serum vitamin D concentration". Korean Journal of Pediatrics 56 (5): 202–204. May 2013. doi:10.3345/kjp.2013.56.5.202. PMID 23741233.
- ↑ 75.0 75.1 "Feeding Your Baby: The First Year". 13 September 2023. https://my.clevelandclinic.org/health/articles/9693-feeding-your-baby-the-first-year.
- ↑ "Changing Incidence of Serum 25-Hydroxyvitamin D Values Above 50 ng/mL: A 10-Year Population-Based Study". Mayo Clinic Proceedings 90 (5): 577–586. May 2015. doi:10.1016/j.mayocp.2015.02.012. PMID 25939935.
- ↑ "A review of the growing risk of vitamin D toxicity from inappropriate practice". British Journal of Clinical Pharmacology 84 (6): 1121–1127. June 2018. doi:10.1111/bcp.13573. PMID 29498758.
- ↑ "Contributions of sunlight and diet to vitamin D status". Calcified Tissue International 92 (2): 163–176. February 2013. doi:10.1007/s00223-012-9634-1. PMID 23001438.
- ↑ 79.0 79.1 "Calcitroic Acid-A Review". ACS Chemical Biology 11 (10): 2665–72. October 2016. doi:10.1021/acschembio.6b00569. PMID 27574921.
- ↑ 80.0 80.1 Vitamin D at Merck Manual of Diagnosis and Therapy Professional Edition
- ↑ 81.0 81.1 Nutrition Through the Life Cycle. Cengage Learning. 28 June 2013. ISBN 978-1-285-82025-5. https://books.google.com/books?id=TeQZBQAAQBAJ. Retrieved 9 April 2017.
- ↑ 82.0 82.1 Discovering Nutrition. Jones & Bartlett Publishers. 18 March 2015. ISBN 978-1-284-06465-0. https://books.google.com/books?id=6IWHCgAAQBAJ. Retrieved 9 April 2017.
- ↑ "The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know". The Journal of Clinical Endocrinology and Metabolism 96 (1): 53–58. January 2011. doi:10.1210/jc.2010-2704. PMID 21118827.
- ↑ "CYP24A1 and SLC34A1 genetic defects associated with idiopathic infantile hypercalcemia: from genotype to phenotype". Clinical Chemistry and Laboratory Medicine 57 (11): 1650–1667. October 2019. doi:10.1515/cclm-2018-1208. PMID 31188746.
- ↑ "Vitamin D-Mediated Hypercalcemia: Mechanisms, Diagnosis, and Treatment". Endocrine Reviews 37 (5): 521–547. October 2016. doi:10.1210/er.2016-1070. PMID 27588937.
- ↑ "Vitamin D and calcium: a systematic review of health outcomes". Evidence Report/Technology Assessment (183): 1–420. August 2009. PMID 20629479.
- ↑ "Vitamin D and multiple health outcomes: umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials". BMJ 348. April 2014. doi:10.1136/bmj.g2035. PMID 24690624.
- ↑ 88.0 88.1 "Vitamin D status and ill health: a systematic review". The Lancet. Diabetes & Endocrinology 2 (1): 76–89. January 2014. doi:10.1016/S2213-8587(13)70165-7. PMID 24622671.
- ↑ "Vitamin D supplementation for the management of knee osteoarthritis: a systematic review of randomized controlled trials". Rheumatology International 37 (9): 1489–1498. September 2017. doi:10.1007/s00296-017-3719-0. PMID 28421358.
- ↑ 90.0 90.1 "Nutrition advice: the vitamin D-lemma". Nature 475 (7354): 23–25. July 2011. doi:10.1038/475023a. PMID 21734684. https://www.nature.com/news/2011/110706/pdf/475023a.pdf. Retrieved 17 November 2011.
- ↑ 91.0 91.1 "Vitamin D supplementation for prevention of mortality in adults". The Cochrane Database of Systematic Reviews 2014 (1). January 2014. doi:10.1002/14651858.CD007470.pub3. PMID 24414552.
- ↑ "Vitamin D and mortality: meta-analysis of individual participant data from a large consortium of cohort studies from Europe and the United States". BMJ 348 (jun17 16). June 2014. doi:10.1136/bmj.g3656. PMID 24938302.
- ↑ "Vitamin D and aging". The Journal of Steroid Biochemistry and Molecular Biology 114 (1–2): 78–84. March 2009. doi:10.1016/j.jsbmb.2008.12.020. PMID 19444937.
- ↑ "Vitamin D, nervous system and aging". Psychoneuroendocrinology 34 (Suppl 1): S278–S286. December 2009. doi:10.1016/j.psyneuen.2009.07.003. PMID 19660871.
- ↑ "The Return of Congenital Rickets, Are We Missing Occult Cases?". Calcified Tissue International 99 (3): 227–236. September 2016. doi:10.1007/s00223-016-0146-2. PMID 27245342.
- ↑ "Congenital rickets due to vitamin D deficiency in the mothers". Clinical Nutrition 34 (5): 793–798. October 2015. doi:10.1016/j.clnu.2014.12.006. PMID 25552383.
- ↑ "Prevention of rickets and vitamin D deficiency in infants, children, and adolescents". Pediatrics 122 (5): 1142–1152. November 2008. doi:10.1542/peds.2008-1862. PMID 18977996.
- ↑ "Interventions for the prevention of nutritional rickets in term born children". The Cochrane Database of Systematic Reviews 2007 (4). October 2007. doi:10.1002/14651858.CD006164.pub2. PMID 17943890.
- ↑ "The problem of rickets in UK Asians". Journal of Human Nutrition and Dietetics 2 (2): 105–16. 1989. doi:10.1111/j.1365-277X.1989.tb00015.x.
- ↑ "Nutritional rickets: deficiency of vitamin D, calcium, or both?". The American Journal of Clinical Nutrition 80 (6 Suppl): 1725S–1729S. December 2004. doi:10.1093/ajcn/80.6.1725S. PMID 15585795.
- ↑ Nature's Perfect Food: How Milk Became America's Drink. NYU Press. 1 February 2002. ISBN 978-0-8147-1938-1. https://books.google.com/books?id=Nr1_u2DvDckC. Retrieved 9 April 2017.
- ↑ "Vitamin D, calcium or a combination of vitamin D and calcium for the treatment of nutritional rickets in children". The Cochrane Database of Systematic Reviews 2020 (4). April 2020. doi:10.1002/14651858.CD012581.pub2. PMID 32303107.
- ↑ "Global Consensus Recommendations on Prevention and Management of Nutritional Rickets". The Journal of Clinical Endocrinology and Metabolism 101 (2): 394–415. February 2016. doi:10.1210/jc.2015-2175. PMID 26745253.
- ↑ 104.0 104.1 "Guidance for Industry: Food Labeling Guide". Food and Drug Administration (FDA). January 2013. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-food-labeling-guide.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "High prevalence of vitamin D inadequacy and implications for health". Mayo Clinic Proceedings 81 (3): 353–373. March 2006. doi:10.4065/81.3.353. PMID 16529140.
- ↑ "Vitamin D and vitamin D analogues for preventing fractures in post-menopausal women and older men". The Cochrane Database of Systematic Reviews 2014 (4). April 2014. doi:10.1002/14651858.CD000227.pub4. PMID 24729336.
- ↑ "A pooled analysis of vitamin D dose requirements for fracture prevention". The New England Journal of Medicine 367 (1): 40–49. July 2012. doi:10.1056/NEJMoa1109617. PMID 22762317. https://research.vu.nl/ws/files/709966/300335.pdf. Retrieved 17 July 2019.
- ↑ "Vitamin D with or without calcium supplementation for prevention of cancer and fractures: an updated meta-analysis for the U.S. Preventive Services Task Force". Annals of Internal Medicine 155 (12): 827–838. December 2011. doi:10.7326/0003-4819-155-12-201112200-00005. PMID 22184690.
- ↑ "Association Between Calcium or Vitamin D Supplementation and Fracture Incidence in Community-Dwelling Older Adults: A Systematic Review and Meta-analysis". JAMA 318 (24): 2466–2482. December 2017. doi:10.1001/jama.2017.19344. PMID 29279934.
- ↑ "Effectiveness and safety of vitamin D in relation to bone health". Evidence Report/Technology Assessment (158): 1–235. August 2007. PMID 18088161.
- ↑ "Vitamin D supplementation and falls: a trial sequential meta-analysis". The Lancet. Diabetes & Endocrinology 2 (7): 573–580. July 2014. doi:10.1016/S2213-8587(14)70068-3. PMID 24768505.
- ↑ "Sports health benefits of vitamin d". Sports Health 4 (6): 496–501. November 2012. doi:10.1177/1941738112461621. PMID 24179588.
- ↑ 113.0 113.1 "Vitamin D and Clinical Cancer Outcomes: A Review of Meta-Analyses". JBMR Plus 5 (1). January 2021. doi:10.1002/jbm4.10420. PMID 33553987.
- ↑ "Comparative efficacy of vitamin D status in reducing the risk of bladder cancer: A systematic review and network meta-analysis". Nutrition 32 (5): 515–523. May 2016. doi:10.1016/j.nut.2015.10.023. PMID 26822497.
- ↑ "Circulating vitamin D levels and colorectal cancer risk: A meta-analysis and systematic review of case-control and prospective cohort studies". Critical Reviews in Food Science and Nutrition 63 (1): 1–17. July 2021. doi:10.1080/10408398.2021.1939649. PMID 34224246.
- ↑ "Vitamin D and cancer prevention". National Cancer Institute, US National Institutes of Health. 21 October 2013. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/vitamin-d-fact-sheet#q4.
- ↑ "Cancer and vitamin D supplementation: a systematic review and meta-analysis". The American Journal of Clinical Nutrition 107 (4): 652–663. April 2018. doi:10.1093/ajcn/nqx047. PMID 29635490.
- ↑ "Vitamin D supplementation and total cancer incidence and mortality: a meta-analysis of randomized controlled trials". Annals of Oncology 30 (5): 733–743. May 2019. doi:10.1093/annonc/mdz059. PMID 30796437.
- ↑ "Vitamin D Supplementation and Cardiovascular Disease Risks in More Than 83 000 Individuals in 21 Randomized Clinical Trials: A Meta-analysis". JAMA Cardiology 4 (8): 765–776. August 2019. doi:10.1001/jamacardio.2019.1870. PMID 31215980.
- ↑ "Vitamin D supplementation, cardiac events and stroke: A systematic review and meta-regression analysis". International Journal of Cardiology. Heart & Vasculature 28. June 2020. doi:10.1016/j.ijcha.2020.100537. PMID 32462077.
- ↑ "Effect of Vitamin D Supplementation on Blood Pressure: A Systematic Review and Meta-analysis Incorporating Individual Patient Data". JAMA Internal Medicine 175 (5): 745–754. May 2015. doi:10.1001/jamainternmed.2015.0237. PMID 25775274.
- ↑ "Effect of Vitamin D on Blood Pressure and Hypertension in the General Population: An Update Meta-Analysis of Cohort Studies and Randomized Controlled Trials". Preventing Chronic Disease 17: E03. January 2020. doi:10.5888/pcd17.190307. PMID 31922371.
- ↑ "Vitamin D status and blood pressure in children and adolescents: a systematic review of observational studies". Systematic Reviews 10 (1). February 2021. doi:10.1186/s13643-021-01584-x. PMID 33618764.
- ↑ "Effects of Nutritional Supplements and Dietary Interventions on Cardiovascular Outcomes: An Umbrella Review and Evidence Map". Annals of Internal Medicine 171 (3): 190–198. August 2019. doi:10.7326/m19-0341. PMID 31284304.
- ↑ "Effect of vitamin D on inflammatory and clinical outcomes in patients with rheumatoid arthritis: a systematic review and dose-response meta-analysis of randomized controlled trials". Nutrition Reviews 82 (5): 600–11. April 2024. doi:10.1093/nutrit/nuad083. PMID 37437898.
- ↑ 126.0 126.1 "Vitamin D3 as an add-on treatment for multiple sclerosis: A systematic review and meta-analysis of randomized controlled trials". Multiple Sclerosis and Related Disorders 82. February 2024. doi:10.1016/j.msard.2024.105433. PMID 38211504. https://www.sciencedirect.com/science/article/abs/pii/S2211034824000129.
- ↑ 127.0 127.1 "Vitamin D and the risk of latent tuberculosis infection: a systematic review and meta-analysis". BMC Pulmonary Medicine 22 (1). January 2022. doi:10.1186/s12890-022-01830-5. PMID 35045861.
- ↑ 128.0 128.1 "Efficacy and safety of vitamin D in tuberculosis patients: a systematic review and meta-analysis". Expert Review of Anti-Infective Therapy 20 (7): 1049–1059. July 2022. doi:10.1080/14787210.2022.2071702. PMID 35477334.
- ↑ "Vitamin D and thyroid disorders: a systematic review and meta-analysis of observational studies". BMC Endocrine Disorders 21 (1). August 2021. doi:10.1186/s12902-021-00831-5. PMID 34425794.
- ↑ "Vitamin D status in patients with systemic lupus erythematosus (SLE): A systematic review and meta-analysis". Autoimmunity Reviews 18 (11). November 2019. doi:10.1016/j.autrev.2019.102392. PMID 31520805.
- ↑ "Myasthenia gravis and vitamin D serum levels: A systematic review and meta-analysis". CNS & Neurological Disorders Drug Targets 22 (5): 752–60. 2023. doi:10.2174/1871527321666220707111344. PMID 35796450.
- ↑ "Vitamin D level in rheumatoid arthritis and its correlation with the disease activity: a meta-analysis". Clinical and Experimental Rheumatology 34 (5): 827–33. September 2016. PMID 27049238.
- ↑ "The association between vitamin D deficiency and multiple sclerosis: an updated systematic review and meta-analysis". Multiple Sclerosis and Related Disorders 90. October 2024. doi:10.1016/j.msard.2024.105804. PMID 39180838. https://www.msard-journal.com/article/S2211-0348(24)00381-X/fulltext.
- ↑ "Vitamin D status and risk of rheumatoid arthritis: systematic review and meta-analysis". BMC Rheumatology 7 (1). March 2023. doi:10.1186/s41927-023-00325-y. PMID 36918989.
- ↑ "Vitamin D supplementation to prevent acute respiratory infections: a systematic review and meta-analysis of aggregate data from randomised controlled trials". Lancet Diabetes Endocrinol 9 (5): 276–92. May 2021. doi:10.1016/S2213-8587(21)00051-6. PMID 33798465.
- ↑ "Vitamin D and innate and adaptive immunity". Vitamins and the Immune System. Vitamins & Hormones. 86. Academic Press. 2011. pp. 23–62. doi:10.1016/B978-0-12-386960-9.00002-2. ISBN 978-0-12-386960-9.
- ↑ "Vitamin D and immune regulation: Antibacterial, antiviral, anti-Inflammatory". Journal of Bone and Mineral Research Plus 5 (1). January 2021. doi:10.1002/jbm4.10405. PMID 32904944.
- ↑ "Low serum vitamin D levels and tuberculosis: a systematic review and meta-analysis". International Journal of Epidemiology 37 (1): 113–119. February 2008. doi:10.1093/ije/dym247. PMID 18245055.
- ↑ "Association Between Inflammatory Bowel Disease and Vitamin D Deficiency: A Systematic Review and Meta-analysis". Inflammatory Bowel Diseases 21 (11): 2708–2717. November 2015. doi:10.1097/MIB.0000000000000546. PMID 26348447.
- ↑ 140.0 140.1 140.2 "Vitamin D for the treatment of inflammatory bowel disease". The Cochrane Database of Systematic Reviews 2023 (10). October 2023. doi:10.1002/14651858.CD011806.pub2. PMID 37781953.
- ↑ "Vitamin D Therapy in Adults With Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis". Inflammatory Bowel Diseases 26 (12): 1819–1830. November 2020. doi:10.1093/ibd/izaa087. PMID 32385487.
- ↑ "Vitamin D for the management of asthma". The Cochrane Database of Systematic Reviews 2023 (2). February 2023. doi:10.1002/14651858.CD011511.pub3. PMID 36744416.
- ↑ 143.0 143.1 "Vitamin D". Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health (NIH). 17 July 2020. https://www.covid19treatmentguidelines.nih.gov/supplements/vitamin-d/.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 144.0 144.1 COVID-19 rapid guideline: vitamin D (PDF) (Technical report). National Institute for Health and Care Excellence (NICE). December 2020. ISBN 978-1-4731-3942-8. NG187. Archived from the original on 3 December 2021. Retrieved 22 February 2021.
- ↑ "COVID-19 rapid guideline: managing COVID-19". 8 May 2024. https://www.nice.org.uk/guidance/ng191.
- ↑ "Low vitamin D status is associated with coronavirus disease 2019 outcomes: a systematic review and meta-analysis". International Journal of Infectious Diseases 104: 58–64. March 2021. doi:10.1016/j.ijid.2020.12.077. PMID 33401034.
- ↑ "Association of Vitamin D Status with SARS-CoV-2 Infection or COVID-19 Severity: A Systematic Review and Meta-analysis". Advances in Nutrition 12 (5): 1636–1658. October 2021. doi:10.1093/advances/nmab012. PMID 33751020.
- ↑ "Therapeutic and prognostic role of vitamin D for COVID-19 infection: A systematic review and meta-analysis of 43 observational studies". The Journal of Steroid Biochemistry and Molecular Biology 211. July 2021. doi:10.1016/j.jsbmb.2021.105883. PMID 33775818.
- ↑ "The link between COVID-19 and VItamin D (VIVID): A systematic review and meta-analysis". Metabolism 119. June 2021. doi:10.1016/j.metabol.2021.154753. PMID 33774074.
- ↑ "Prognostic and Therapeutic Role of Vitamin D in COVID-19: Systematic Review and Meta-analysis". The Journal of Clinical Endocrinology and Metabolism 107 (5): 1484–1502. April 2022. doi:10.1210/clinem/dgab892. PMID 34894254.
- ↑ "The Effect of Vitamin D Supplementation Post COVID-19 Infection and Related Outcomes: A Systematic Review and Meta-Analysis". Nutrients 16 (22): 3794. November 2024. doi:10.3390/nu16223794. PMID 39599582.
- ↑ "The impact of vitamin D administration on mortality in COVID-19 patients: a systematic review and meta-analysis of randomized controlled trials". Inflammopharmacology 32 (5): 3205–12. October 2024. doi:10.1007/s10787-024-01564-2. PMID 39225947. https://pure.hud.ac.uk/en/publications/14482531-5730-4e82-be48-1766b4070e6d.
- ↑ "Effect of Vitamin D3 Supplementation on Severe COVID-19: A Meta-Analysis of Randomized Clinical Trials". Nutrients 16 (10): 1402. May 2024. doi:10.3390/nu16101402. PMID 38794642.
- ↑ "Vitamin D to prevent exacerbations of COPD: systematic review and meta-analysis of individual participant data from randomised controlled trials". Thorax 74 (4): 337–345. April 2019. doi:10.1136/thoraxjnl-2018-212092. PMID 30630893.
- ↑ "Effects of Vitamin D Supplementation on Prevention of Type 2 Diabetes in Patients With Prediabetes: A Systematic Review and Meta-analysis". Diabetes Care 43 (7): 1650–1658. July 2020. doi:10.2337/dc19-1708. PMID 33534730.
- ↑ "The effects of vitamin D supplementation on indices of glycemic control in Iranian diabetics: A systematic review and meta-analysis". Complementary Therapies in Clinical Practice 34: 294–304. February 2019. doi:10.1016/j.ctcp.2018.12.009. PMID 30712741.
- ↑ "Serum vitamin D levels in relation to type-2 diabetes and prediabetes in adults: a systematic review and dose-response meta-analysis of epidemiologic studies". Critical Reviews in Food Science and Nutrition 62 (29): 8178–8198. June 2021. doi:10.1080/10408398.2021.1926220. PMID 34076544.
- ↑ "Interventions for latent autoimmune diabetes (LADA) in adults". The Cochrane Database of Systematic Reviews 2011 (9). September 2011. doi:10.1002/14651858.cd006165.pub3. PMID 21901702.
- ↑ "Vitamin D Status and Attention Deficit Hyperactivity Disorder: A Systematic Review and Meta-Analysis of Observational Studies". Advances in Nutrition 9 (1): 9–20. January 2018. doi:10.1093/advances/nmx002. PMID 29438455.
- ↑ "The Effect of Vitamin D Supplementation on Attention-Deficit/Hyperactivity Disorder: A Systematic Review and Meta-Analysis of Randomized Controlled Trials". Journal of Child and Adolescent Psychopharmacology 29 (9): 670–687. November 2019. doi:10.1089/cap.2019.0059. PMID 31368773.
- ↑ "Vitamin D supplementation for depressive symptoms: a systematic review and meta-analysis of randomized controlled trials". Psychosomatic Medicine 76 (3): 190–196. April 2014. doi:10.1097/psy.0000000000000044. PMID 24632894.
- ↑ "Vitamin D, cognition, and dementia: a systematic review and meta-analysis". Neurology 79 (13): 1397–1405. September 2012. doi:10.1212/WNL.0b013e31826c197f. PMID 23008220.
- ↑ 163.0 163.1 "Vitamin D and schizophrenia: 20 years on". Molecular Psychiatry 26 (7): 2708–2720. July 2021. doi:10.1038/s41380-021-01025-0. PMID 33500553.
- ↑ "Vitamin D deficiency and Schizophrenia in Adults: A Systematic Review and Meta-analysis of Observational Studies". Psychiatry Research 288. June 2020. doi:10.1016/j.psychres.2020.112959. PMID 32335466.
- ↑ "Mechanisms Suggesting a Relationship between Vitamin D and Erectile Dysfunction: An Overview". Biomolecules 13 (6): 930. June 2023. doi:10.3390/biom13060930. PMID 37371510.
- ↑ "Vitamin D and Male Erectile Function: An Updated Review". The World Journal of Men's Health 39 (1): 31–37. January 2021. doi:10.5534/wjmh.190151. PMID 32009309.
- ↑ "Therapeutic Effects of Vitamin D on Vaginal, Sexual, and Urological Functions in Postmenopausal Women". Nutrients 15 (17): 3804. August 2023. doi:10.3390/nu15173804. PMID 37686835.
- ↑ "Vitamin D and its role during pregnancy in attaining optimal health of mother and fetus". Nutrients 4 (3): 208–230. March 2012. doi:10.3390/nu4030208. PMID 22666547.
- ↑ 169.0 169.1 "Association between maternal serum 25-hydroxyvitamin D level and pregnancy and neonatal outcomes: systematic review and meta-analysis of observational studies". BMJ 346. March 2013. doi:10.1136/bmj.f1169. PMID 23533188.
- ↑ "Vitamin D supplementation during pregnancy: state of the evidence from a systematic review of randomised trials". BMJ 359. November 2017. doi:10.1136/bmj.j5237. PMID 29187358.
- ↑ "Vitamin D supplementation for women during pregnancy". The Cochrane Database of Systematic Reviews 2024 (7). July 2024. doi:10.1002/14651858.CD008873.pub5. PMID 39077939.
- ↑ "Effect of Oral Consumption of Vitamin D on Uterine Fibroids: A Systematic Review and Meta-Analysis of Randomized Clinical Trials". Nutrition and Cancer 76 (3): 226–235. 2024. doi:10.1080/01635581.2023.2288716. PMID 38234246.
- ↑ "A Systematic Review of Vitamin D and Fibroids: Pathophysiology, Prevention, and Treatment". Reproductive Sciences 30 (4): 1049–1064. April 2023. doi:10.1007/s43032-022-01011-z. PMID 35960442.
- ↑ Li, Zizhan; Wei, Xiao; Shao, Zhongjun; Liu, Huan; Bai, Shizhu (2023-10-19). "Correlation between vitamin D levels in serum and the risk of dental caries in children: a systematic review and meta-analysis". BMC Oral Health 23 (1): 768. doi:10.1186/s12903-023-03422-z. ISSN 1472-6831. PMID 37858104.
- ↑ 175.0 175.1 175.2 ((European Food Safety Authority (EFSA) Panel on Dietetic Products, Nutrition and Allergies (NDA) )) (2010). "Scientific opinion on the substantiation of health claims related to vitamin D and normal function of the immune system and inflammatory response (ID 154, 159), maintenance of normal muscle function (ID 155) and maintenance of normal cardiovascular function (ID 159) according to Article 13(1) of Regulation (EC) No 1924/2006". EFSA Journal 8 (2): 1468–85. doi:10.2903/j.efsa.2010.1468.
- ↑ ((European Food Safety Authority (EFSA) Panel on Dietetic Products, Nutrition and Allergies (NDA) )) (2011). "Scientific opinion on the substantiation of a health claim related to vitamin D and risk of falling according to Article 14 of Regulation (EC) No 1924/2006". EFSA Journal 9 (9): 2382–2400. doi:10.2903/j.efsa.2011.2382. http://orbit.dtu.dk/files/6355745/prod11324398702885.Scientific.pdf. Retrieved 20 August 2019.
- ↑ "Health Canada Scientific Summary on the U. S. Health Claim Regarding Calcium and Osteoporosis". Bureau of Nutritional Sciences Food Directorate, Health Products and Food Branch Health Canada. 1 May 2000. http://www.hc-sc.gc.ca/fn-an/label-etiquet/claims-reclam/assess-evalu/calcium_osteo-eng.php.
- ↑ "Newly established regulation in Japan: foods with health claims". Asia Pacific Journal of Clinical Nutrition 11 (2): S94–S96. 2002. doi:10.1046/j.1440-6047.2002.00007.x. PMID 12074195.
- ↑ 179.0 179.1 179.2 179.3 179.4 Nutrient Reference Values for Australia and New Zealand Including Recommended Dietary Intakes. Canberra: National Health and Medical Research Council. 2006. ISBN 1-86496-243-7. https://www.nhmrc.gov.au/about-us/publications/nutrient-reference-values-australia-and-new-zealand-including-recommended-dietary-intakes#block-views-block-file-attachments-content-block-1. Retrieved 19 March 2023.
- ↑ 180.0 180.1 EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA) (2012). "Scientific Opinion on the Tolerable Upper Intake Level of vitamin D". EFSA Journal 10 (7): 2813. doi:10.2903/j.efsa.2012.2813.
- ↑ Institute of Medicine (1997). "Vitamin D". Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D and Fluoride. Washington, DC: The National Academies Press. pp. 250–87. ISBN 0-309-06403-1. https://nap.nationalacademies.org/read/5776/chapter/9. Retrieved 28 December 2024.
- ↑ "PHE publishes new advice on vitamin D". Public Health England. 21 July 2016. https://www.gov.uk/government/news/phe-publishes-new-advice-on-vitamin-d.
- ↑ "Federal Register May 27, 2016 Food Labeling: Revision of the Nutrition and Supplement Facts Labels. FR page 33982.". https://www.gpo.gov/fdsys/pkg/FR-2016-05-27/pdf/2016-11867.pdf.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Daily Value Reference of the Dietary Supplement Label Database (DSLD)". https://www.dsld.nlm.nih.gov/dsld/dailyvalue.jsp.
- ↑ "Vitamin D food fortification on the table". Australian Broadcasting Corporation. 12 June 2012. http://www.abc.net.au/science/articles/2012/06/12/3522708.htm.
- ↑ "Australian Health Survey: Biomedical Results for Nutrients, 2011–12". 21 December 2011. https://www.abs.gov.au/statistics/health/health-conditions-and-risks/australian-health-survey-biomedical-results-nutrients/latest-release.
- ↑ "nutrition labelling for foodstuffs as regards recommended daily allowances, energy conversion factors, and definitions". 28 October 2008. https://eur-lex.europa.eu/legal-content/En/TXT/PDF/?uri=CELEX:32008L0100.
- ↑ "Vitamin D (translated)" (in sv). Swedish National Food Agency. https://www.livsmedelsverket.se/livsmedel-och-innehall/naringsamne/vitaminer-och-antioxidanter/d-vitamin.
- ↑ Vitamin-D-Bedarf bei fehlender endogener Synthese Deutsche Gesellschaft für Ernährung, January 2012
- ↑ "EMAS position statement: Vitamin D and postmenopausal health". Maturitas 71 (1): 83–88. January 2012. doi:10.1016/j.maturitas.2011.11.002. PMID 22100145.
- ↑ "Vitamin D in mushrooms". Nutrient Data Laboratory, US Department of Agriculture. 2009. https://www.ars.usda.gov/ARSUserFiles/80400525/Articles/AICR09_Mushroom_VitD.pdf.
- ↑ 192.0 192.1 "Natural vitamin D content in animal products". Advances in Nutrition 4 (4): 453–462. July 2013. doi:10.3945/an.113.003780. PMID 23858093.
- ↑ "Vitamin food fortification today". Food & Nutrition Research 56: 5459. 2012. doi:10.3402/fnr.v56i0.5459. PMID 22481896.
- ↑ "What You Need to Know When Choosing Milk and Milk Alternatives". 11 November 2021. https://health.clevelandclinic.org/what-you-need-to-know-when-choosing-milk-and-milk-alternatives.
- ↑ "Serum concentrations of lipids, vitamin d metabolites, retinol, retinyl esters, tocopherols and selected carotenoids in twelve captive wild felid species at four zoos". The Journal of Nutrition 133 (1): 160–6. January 2003. doi:10.1093/jn/133.1.160. PMID 12514284.
- ↑ "Vitamin D and the skin". Journal of Bone and Mineral Metabolism 28 (2): 117–30. March 2010. doi:10.1007/s00774-009-0153-8. PMID 20107849.
- ↑ "Sunlight, UV Radiation, Vitamin D, and Skin Cancer: How Much Sunlight do We Need?". Sunlight, Vitamin D and Skin Cancer. Advances in Experimental Medicine and Biology. 1268. Springer. 2020. pp. 19–36. doi:10.1007/978-3-030-46227-7_2. ISBN 978-3-030-46226-0. "108 references"
- ↑ "Vitamin D in plants: a review of occurrence, analysis, and biosynthesis". Frontiers in Plant Science 4: 136. May 2013. doi:10.3389/fpls.2013.00136. PMID 23717318. Bibcode: 2013FrPS....4..136J.
- ↑ "Vitamin D in Nature: A Product of Synthesis and/or Degradation of Cell Membrane Components". Biochemistry. Biokhimiia 83 (11): 1350–1357. November 2018. doi:10.1134/S0006297918110056. PMID 30482146.
- ↑ "Vitamin D in an ecological context". International Journal of Circumpolar Health 59 (1): 26–32. January 2000. PMID 10850004.
- ↑ 201.0 201.1 "Vitamin D: calcium and bone homeostasis during evolution". BoneKEy Reports 3: 480. January 2014. doi:10.1038/bonekey.2013.214. PMID 24466411.
- ↑ 202.0 202.1 202.2 "Vitamin D and evolution: Pharmacologic implications". Biochemical Pharmacology 173. March 2020. doi:10.1016/j.bcp.2019.07.024. PMID 31377232.
- ↑ 203.0 203.1 203.2 "Vitamin D in the Context of Evolution". Nutrients 14 (15): 3018. July 2022. doi:10.3390/nu14153018. PMID 35893872. Bibcode: 2022Nutri..14.3018C.
- ↑ "The pathology of vitamin D deficiency in domesticated animals: An evolutionary and comparative overview". International Journal of Paleopathology 23: 100–09. December 2018. doi:10.1016/j.ijpp.2018.03.001. PMID 29544996. Bibcode: 2018IJPal..23..100U.
- ↑ Nijboer, Joeke (September 2024). "Nutrition in Reptiles: Vitamin D and Ultraviolet Light". https://www.merckvetmanual.com/management-and-nutrition/nutrition-exotic-and-zoo-animals/nutrition-in-reptiles.
- ↑ Hoppes, Sharman (September 2024). "Nutritional Diseases of Pet Birds: Phosphorus and vitamin D3 imbalance". https://www.merckvetmanual.com/exotic-and-laboratory-animals/pet-birds/nutritional-diseases-of-pet-birds.
- ↑ "Ultraviolet radiation and Vitamin D3 in amphibian health, behaviour, diet and conservation". Comp Biochem Physiol A 154 (2): 184–90. October 2009. doi:10.1016/j.cbpa.2009.06.008. PMID 19555772. http://usir.salford.ac.uk/id/eprint/36713/1/Ultraviolet%20radiation%20and%20Vitamin%20D3%20in%20amphibian.pdf.
- ↑ "Vitamin D metabolism in dogs and cats and its relation to diseases not associated with bone metabolism". Journal of Animal Physiology and Animal Nutrition 104 (1): 322–42. January 2020. doi:10.1111/jpn.13259. PMID 31803981.
- ↑ "The Role of Vitamin D in Small Animal Bone Metabolism". Metabolites 10 (12): 496. December 2020. doi:10.3390/metabo10120496. PMID 33287408.
- ↑ "Physiological function of vitamin D3 in fish". Reviews in Aquaculture 15 (4): 1732–1748. March 2023. doi:10.1111/raq.12814. Bibcode: 2023RvAq...15.1732C.
- ↑ 211.0 211.1 211.2 211.3 211.4 "Evolution, Prehistory and Vitamin D". International Journal of Environmental Research and Public Health 17 (2): 646. January 2020. doi:10.3390/ijerph17020646. PMID 31963858.
- ↑ "Why Humans and Their Fur Parted Ways" (in en-US). The New York Times. 19 August 2003. ISSN 0362-4331. https://www.nytimes.com/2003/08/19/science/why-humans-and-their-fur-parted-ways.html.
- ↑ "The protective role of melanin against UV damage in human skin". Photochemistry and Photobiology 84 (3): 539–549. 2008. doi:10.1111/j.1751-1097.2007.00226.x. PMID 18435612.
- ↑ "A Single Migration From Africa Populated the World, Studies Find". The New York Times. 22 September 2016. https://www.nytimes.com/2016/09/22/science/ancient-dna-human-history.html.
- ↑ "A revised action spectrum for vitamin D synthesis by suberythemal UV radiation exposure in humans in vivo". Proceedings of the National Academy of Sciences of the United States of America 118 (40). October 2021. doi:10.1073/pnas.2015867118. PMID 34580202. Bibcode: 2021PNAS..11815867Y.
- ↑ 216.0 216.1 "The vitamin D epidemic and its health consequences". The Journal of Nutrition 135 (11): 2739S–2748S. November 2005. doi:10.1093/jn/135.11.2739S. PMID 16251641. http://jn.nutrition.org/content/135/11/2739S.full.pdf. "Vitamin D3 is produced commercially by extracting 7-dehydrocholesterol from wool fat, followed by UVB irradiation and purification [...] [Vitamin D2] is commercially made by irradiating and then purifying the ergosterol extracted from yeast".
- ↑ "Chapter 6: Industrial Aspects of Vitamin D". Vitamin D: Two-Volume Set. Academic Press. 12 May 2011. ISBN 978-0-12-381978-9. https://books.google.com/books?id=w7hMAFmsM84C&pg=PA79.
- ↑ "Genetic evidence that the human CYP2R1 enzyme is a key vitamin D 25-hydroxylase". Proceedings of the National Academy of Sciences of the United States of America 101 (20): 7711–15. May 2004. doi:10.1073/pnas.0402490101. PMID 15128933. Bibcode: 2004PNAS..101.7711C.
- ↑ "Section I: Ch. 8: Vitamin D Binding Protein". Vitamin D. 1 (2 ed.). Academic Press. 2004. pp. 117–134. ISBN 978-0-12-252687-9. https://books.google.com/books?id=5c66r0KrPUMC. Retrieved 9 April 2017.
- ↑ 220.0 220.1 "Update in vitamin D". The Journal of Clinical Endocrinology and Metabolism 95 (2): 471–478. February 2010. doi:10.1210/jc.2009-1773. PMID 20133466.
- ↑ 221.0 221.1 221.2 221.3 "Exploring vitamin D metabolism and function in cancer". Experimental & Molecular Medicine 50 (4): 1–14. April 2018. doi:10.1038/s12276-018-0038-9. PMID 29657326.
- ↑ "Vitamin D: Production, Metabolism and Mechanisms of Action". Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.. 31 December 2021. https://www.ncbi.nlm.nih.gov/books/NBK278935/.
- ↑ "Alternative pathways for vitamin D metabolism". Feldman and Pike's Vitamin D. Academic Press. January 2024. pp. 85–109. doi:10.1016/B978-0-323-91386-7.00001-5. ISBN 978-0-323-91390-4.
- ↑ "Membrane-mediated actions of 1,25-dihydroxy vitamin D3: a review of the roles of phospholipase A2 activating protein and Ca(2+)/calmodulin-dependent protein kinase II". The Journal of Steroid Biochemistry and Molecular Biology 147: 81–84. March 2015. doi:10.1016/j.jsbmb.2014.11.002. PMID 25448737.
- ↑ "The Non-Genomic Actions of Vitamin D". Nutrients 8 (3): 135. March 2016. doi:10.3390/nu8030135. PMID 26950144.
- ↑ "Nongenomic Activities of Vitamin D". Nutrients 14 (23): 5104. December 2022. doi:10.3390/nu14235104. PMID 36501134.
- ↑ 227.0 227.1 227.2 227.3 227.4 227.5 227.6 "Vitamin D and Its Target Genes". Nutrients 14 (7): 1354. March 2022. doi:10.3390/nu14071354. PMID 35405966.
- ↑ 228.00 228.01 228.02 228.03 228.04 228.05 228.06 228.07 228.08 228.09 "The vitamin D receptor: new paradigms for the regulation of gene expression by 1,25-dihydroxyvitamin D(3)". Endocrinology and Metabolism Clinics of North America 39 (2): 255–69, table of contents. June 2010. doi:10.1016/j.ecl.2010.02.007. PMID 20511050.
- ↑ 229.0 229.1 "The function of vitamin D receptor in vitamin D action". Journal of Biochemistry 127 (5): 717–22. May 2000. doi:10.1093/oxfordjournals.jbchem.a022662. PMID 10788778.
- ↑ 230.0 230.1 230.2 "The Biological Activities of Vitamin D and Its Receptor in Relation to Calcium and Bone Homeostasis, Cancer, Immune and Cardiovascular Systems, Skin Biology, and Oral Health". BioMed Research International 2018. 2018. doi:10.1155/2018/9276380. PMID 29951549.
- ↑ "The role of vitamin D in the endocrinology controlling calcium homeostasis". Molecular and Cellular Endocrinology 453: 36–45. September 2017. doi:10.1016/j.mce.2017.04.008. PMID 28400273.
- ↑ 232.0 232.1 "Genomic Effects of the Vitamin D Receptor: Potentially the Link between Vitamin D, Immune Cells, and Multiple Sclerosis". Frontiers in Immunology 9. 2018. doi:10.3389/fimmu.2018.00477. PMID 29593729.
- ↑ 233.0 233.1 Medical Physiology E-Book. Elsevier Health Sciences. 29 March 2016. ISBN 978-1-4557-3328-6. https://books.google.com/books?id=6QzhCwAAQBAJ. Retrieved 9 April 2017.
- ↑ 234.0 234.1 234.2 "Parathyroid hormone – a bone anabolic and catabolic agent". Current Opinion in Pharmacology 5 (6): 612–7. Dec 2005. doi:10.1016/j.coph.2005.07.004. PMID 16181808.
- ↑ "Renal control of calcium, phosphate, and magnesium homeostasis". Clinical Journal of the American Society of Nephrology 10 (7): 1257–72. 2015. doi:10.2215/CJN.09750913. PMID 25287933.
- ↑ "On the Molecular Mechanism of Intestinal Calcium Transport". Mineral Absorption in the Monogastric GI Tract. Advances in Experimental Medicine and Biology. 249. 1989. pp. 45–65. doi:10.1007/978-1-4684-9111-1_5. ISBN 978-1-4684-9113-5.
- ↑ "The vitamin D receptor in osteoblastic cells but not secreted parathyroid hormone is crucial for soft tissue calcification induced by the proresorptive activity of 1,25(OH)2D3". J Steroid Biochem Mol Biol 232. September 2023. doi:10.1016/j.jsbmb.2023.106351. PMID 37352941.
Further reading
- Feldman and Pike's Vitamin D (5th Edition) Volume One: Biochemistry, Physiology and Diagnostics; Editors: Hewison M, Bouillon R, Giovannucci E, Goltzman D,Meyer MB, Welsh J. Academic Press (October 2023) ISBN 978-0-32-391386-7
- Feldman and Pike's Vitamin D (5th Edition) Volume Two: Diseases and Therapeutics; Editors: Hewison M, Bouillon R, Giovannucci E, Goltzman D,Meyer MB, Welsh J. Academic Press (January 2024) ISBN 978-0-32-391338-6
- Dietary Reference Intakes for Vitamin D and Calcium Editors: Ross AC, Taylor CL, Yaktine AL, Del Valle HB. National Academies Press (US) (2011) ISBN 978-0-30-916394-1 Available from: Dietary Reference Intakes for Calcium and Vitamin D doi: 10.17226/13050
| Scholia has a profile for Vitamin D (). |
 |


