Medicine:Trimethylaminuria
Trimethylaminuria (TMAU), also known as fish odor syndrome or fish malodor syndrome,[1] is a rare metabolic disorder that causes a defect in the normal production of an enzyme named flavin-containing monooxygenase 3 (FMO3).[2][3][4] When FMO3 is not working correctly or if not enough enzyme is produced, the body loses the ability to properly convert the rotting fish smelling chemical trimethylamine (TMA) from precursor compounds in food digestion into trimethylamine oxide (TMAO), through a process called N-oxidation.
Trimethylamine can temprorarily build up in the bloodstream and is released in the person's urine, sweat, and breath, giving off a rotten fish odor. Primary trimethylaminuria is caused by genetic mutations that affect the FMO3 function of the liver. Symptoms matching TMAU can also occur when there is no genetic cause, yet excessive TMA is excreted - this has been described as secondary trimethylaminuria (TMAU2).
Symptoms and signs
People with TMAU may have an intermittent rotten fish-like body odor, depending on diet and the severity of their FM03 mutation. In a 2011 study by Wise and colleagues of 115 positively identified TMAU subjects,[5] after a choline challenge load test (intentionally ingesting a TMA precursor), only 10% expressed a smell at a social distance. In a fasted state (12 hours), 0% had a smell detectable at a social distance, and only 5% had some minor malodour at an intimate distance. These findings suggested that those who produced an odour had a more severe form of FMO3 impairment. [citation needed]
Individuals with this condition do not have any physical symptoms, and they typically appear healthy.[6]
For unknown reasons, the condition seems more common in women than in men. Scientists suspect that such female sex hormones as progesterone and estrogen aggravate the condition. According to several reports, the condition worsens around puberty. In women, symptoms may worsen just before and during menstrual periods, after taking oral contraceptives, and around menopause.[6]
Genetics
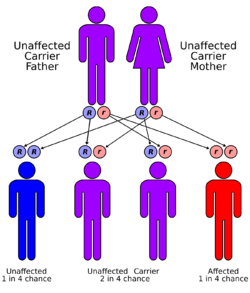
Most cases of trimethylaminuria appear to be inherited in an autosomal recessive pattern, which means two copies of the gene in each cell are altered. The parents of an individual with an autosomal recessive disorder are both carriers of one copy of the altered gene. Carriers are asymptomatic and are not at risk of developing the disorder.[7]
Mutations in the FMO3 gene, which is found on the long arm of chromosome 1, cause trimethylaminuria.[2] The FMO3 gene makes an enzyme that breaks down nitrogen-containing compounds from the diet, including trimethylamine. These compounds are produced by bacteria in the intestine as they digest proteins from eggs, meat, soy, and other foods. Normally, the FMO3 enzyme converts fishy-smelling trimethylamine into trimethylamine N-oxide which has no odor. If the enzyme is missing or its activity is reduced because of a mutation in the FMO3 gene, trimethylamine is not broken down and instead builds up in the body. As the compound is released in a person's sweat, urine, and breath, it causes the strong odor characteristic of trimethylaminuria. Researchers believe that stress and diet also play a role in triggering symptoms.[8]
There are more than 40 known mutations associated with TMAU.[9][10][11] Loss-of-function mutations, nonsense mutations, and missense mutations are three of the most common. Nonsense and missense mutations cause the most severe phenotypes. In 2007 the evolution of the FMO3 gene was studied, including the evolution of some mutations associated with TMAU.[12]
Metabolic pathway
- When food is consumed that contains TMA and/or TMAO (predominately seafood; saltwater fish, shellfish, seaweed and kelp). TMAO is converted by bacteria in the lower gastrointestinal tract (gut) into TMA.
- When a food substance, supplement or medicine that contains a TMA precursor (choline or carnitine) is ingested. Some precursor is absorbed into the bloodstream in the small intestine before reaching the gut (the RDI of choline is 450–550 mg per day,[13] which is absorbed this way), however there is a limit to the transport capacity of the intestine, and not all precursor is exposed to the process. Unabsorbed precursor ends up in the gut.[14] Certain bacteria[15] in the gut can convert those precursors to TMA,[16] the proportion of precursor converted to TMA is related to the amount of specific bacteria in the gut[17] but on average 63% of excess choline, and 31% of carnitine (±13%) are converted to TMA.[18]
TMA in the gut is absorbed through the intestinal lining and enters the bloodstream, where it is processed by the liver. A healthy liver produces an abundance of the enzyme FMO3, which neutralises the TMA by oxidising it to an odourless TMAO. If FMO3 enzyme production is compromised, or there is too much TMA for the amount of enzyme, then TMA will continue to circulate in the bloodstream until enough enzyme is produced.
Although lecithin, creatinine and betaine are technically precursors to TMA, pilot studies have shown no significant effect on the production of excess TMA/TMAO in urinary analysis at normal dietary levels of consumption.[14] When taken in large quantities (12g/day) betaine has been known to cause fish odor symptoms,[19] meaning that there is some conversion of betaine to TMA if supplements are taken regularly.
Diagnosis
False positives can occur in the following conditions, where elevated TMA can be present in the urine without any underlying TMAU:
- Urinary tract infection[20]
- Bacterial vaginosis[20]
- Cervical cancer[20]
- Advanced liver or kidney disease[20]
A similar foul-smelling odor of the urine has also been associated with colonization of the urinary tract with a bacterium called Aerococcus urinae, especially in children.[21][22]
Consumption of certain Brassica vegetables, in particular brussels sprouts, due to dietary indoles reducing FMO3 activity - research found that a diet including 300g of brussels sprouts per day for 3 weeks temporarily reduced FMO3 capability from 90%+ to ~70%. No odour was reported on any of the participants.[23] This may result in minor secondary TMAU result.
Olfactory reference syndrome is a condition where there is a persistent false belief and preoccupation with the idea of emitting an abnormal body odor. According to McNiven[24] at a Canadian genetics clinic, 83% of referrals for genetic testing for TMAU were deemed likely to instead have ORS. Findings found that the use of "fecal/sewage" as a description, and the use of multiple descriptors of the smell, and 'incorrect' locations of smell origin effectively differentiated ORS from TMAU. In the literature on body odour identification, emphasis is frequently placed on multiple consultations to reduce the risk of misdiagnosis, and also asking the individual to have a reliable confidant accompany them to the consultation who can confirm the reality of the reported symptom. ORS patients are unable to provide such confidants as they have no objective odor.[25][26]
Unrelated fecal smells are an often misinterpreted self reported symptom associated with TMAU,[24] Cashman JR[27] found that 53% of TMAU and 59% of non-TMAU subjects suffered from regular halitosis, caused by dental plaque on the back of the tongue, which produced on average "200-600 ppb of sulfurous/fecal smelling volatile sulfur compounds (i.e., VSC: hydrogen sulfide; methylmercaptan; dimethylsulfide) with each exhalation, creating a 'malodorous cloud' in their vicinity". It is possible that other causes such as halitosis, haemorrhoids, regular bromhidrosis, ORS or in severe cases, a bowel obstruction leading to fecal vomiting may be the cause of fecal smells. There is the possibility that someone may suffer from both Trimethylaminuria and ORS-like paranoia, due to the potential lack of ability to smell the odour oneself and the worry that it generates. It is recommended to organise reliable confidants, colleagues, friends or relatives ("odor buddies"[28]) to work with the sufferer to discreetly inform them if they are presenting an odour.
Affected individuals experience shame and embarrassment, fail to maintain relationships, avoid contact with people who comment on their condition, and are obsessive about masking the odour with hygiene products and even smoking. The malodorous aspect can have serious and destructive effects on schooling, personal life, career and relationships, resulting in social isolation, low self-esteem, depression, paranoid behaviour, and suicide. Delayed diagnosis, body odour and the lack of cure may lead to psychosocial issues. When the condition is suspected or known to occur in a family, genetic testing can be helpful in identifying the specific individuals who have or carry the disorder.[29]
The metabolic and clinical manifestations of TMAU are generally regarded as benign, as there is no associated organ dysfunction. This designation, and the fact that the condition is often unrecognised by doctors, misdiagnosed and can have important ramifications including missed or delayed diagnosis.[29]
Treatment
There is no known permanent cure for primary trimethylaminuria, but symptoms can in most cases be managed via diet,[30] and sometimes by certain supplements (as below).
Ways of reducing the fishy odor may include:
- Avoiding all seafood, including fish, shellfish, kelp, and seaweed. Sea life contains TMAO, which is used as an osmolyte to counter hydrostatic pressure underwater.[31]
- Reducing the consumption of foods and supplements that contain carnitine,[32] such as red meat (beef, lamb and pork), liver, and offal. A study performed by Wang Z[33] found that when comparing diets where the main protein source was red meat, white meat and non-meat protein sources, consumption of red meat increased the production of TMAO, whereas white meat and non-meat protein diets generated only low to negligible amounts TMAO. This study indicates that red meat is a major driver of TMA production by altering the balance of microbiota in the stomach due to the carnitine found in red meat. A further study by Crimarco A[17] found that a 8 week plant based diet significantly reduced TMAO production, and further, that after switching the diet to include animal based protein, TMAO production was less than the participants who had only been given an animal protein based diet. The findings suggest that the microbiome in the gut is modified by a plant based diet, and for a time a person will lack the bacteria required to convert choline and carnitine into TMA at the same rate of an animal protein based diet.
- If not pregnant or breastfeeding, reducing the consumption of foods and supplements containing choline[34] - fish, red meat, white meat, offal, egg yolks, legumes, beans, whey products, milk, and other foods that contain high levels of choline. Choline is an essential nutrient so complete elimination of choline is unadvised. As above, white meat (chicken, turkey) and plant based products may be fine to consume if red meat is predominantly avoided. Note, while raw ingredients like soybeans have a relatively high choline content, some processed products like soy sauce, soy milk and tofu have low choline content, due to dilution of ingredients, small serving size, or removal as a byproduct during the manufacturing process. It's best to check the choline content[34] of food and the portion size for a better understanding of how much choline is being consumed.
- Vitamin B2 at 50 mg per day in combination with diet resolved smell issues for 2 children with TMAU.[35] B2 was found to increase residual FMO3 performance in the liver, meaning more TMA is neutralised.
- Taking low doses of antibiotics such as neomycin and metronidazole[36] in order to reduce the amount of bacteria in the gut, although this is not recommended as a long term solution due to antibiotic resistance and other side effects.
- Using slightly acidic detergent and body washes with a pH between 5.5 and 6.5
Additionally, at least one study[37] has suggested that daily intake of the supplements activated charcoal and copper chlorophyllin may temporarily improve the quality of life of individuals afflicted with TMAU by helping their bodies to oxidize and convert TMA to the odorless N-oxide (TMAO) metabolite. Study participants experienced subjective reduction in odor as well as objective reduction in TMA and increase in TMAO concentration measured in their urine. The study found that:[citation needed]
- 85% of test participants experienced complete loss of detectable "fishy" odor
- 10% experienced some reduction in detectable odor
- 5% did not experience any detectable odor reduction
Secondary trimethylaminuria
A fish-like body odor can result from extreme excess consumption of TMA precursors such as choline, carnitine and betaine (usually unobtainable via regular dietary intake, it requires high levels of supplement intake). 900mg of trimethylamine,[38] 8g-20g of choline,[39] 3g of carnitine[40] or 20g betatine[41] has been known to cause temporary TMAU-like fish odour symptoms. These symptoms only last until supplement intake has ceased. Note, the RDI for choline is 450-550mg and most typical diets fall below this value,[42] so an excessive amount of precursor is required.
Two cases of the disorder have been identified in adults with liver damage caused by hepatitis. It is unknown if these cases were temporary or not as the individuals did not return for subsequent testing.[39]
In the case where someone is experiencing liver failure or kidney failure,[39] TMA is sometimes an element present as part of fetor hepaticus, the "breath of the dead".
TMAU2 and gut dysbiosis
There are no studies or case reports that identify gut dysbiosis as a cause for secondary trimethylaminuria. Between the years of 1997 and 2017, Sheffield Children's Hospital in England diagnosed several hundred people with TMAU2, and suggested the majority to be caused by dysbiosis in the gut.[43] There is no reference to scientific research confirming gut dysbiosis alone can cause TMAU2, and this type of diagnosis was not produced at other testing sites. Cases were claimed to be successfully cured with antibiotics and dietary changes as above. In 2017 a new gas chromatography machine[44] and testing procedure was introduced, which has far fewer false positive results,[45][46] and very few (if any) TMAU2 results.
While the balance of gut bacteria does play a part in the amount of TMA being produced from precursors in the gut[47] (antibiotic treatment temporarily stops odour), gut bacteria in general convert a significant proportion of dietary TMA precursors already. For example, on average 63% of excess choline, and 31% of carnitine (+-13%) are converted to TMA[18] by an average gut profile - even if dysbiosis raised these values to 100%, it would still not meet the thresholds as above for precursor overload on a regular diet. TMAU2 by gut dysbiosis is currently a hypothetical[39] condition requiring further research.
History
The first clinical case of TMAU was described in 1970.[48] Her mother related that her child, a 6 year old girl, had intermittently had a peculiar "fishy" odour. Analysis of her urine showed an elevated level of trimethylamine, and that a chemically pure free base sample of trimethylamine smelled similar to the patient's fishy odor. They tested her condition by giving her more trimethylamine, which substantially increased her odour (which it did not in control subjects).
Society and culture
In 2014, singer/songwriter Cassie Graves was first featured in the Daily Mail, the Daily Mirror, and The Metro UK newspapers in both print and Online,[49] giving an interview about her experiences with Trimethylaminuria. The article was later repurposed in media across the globe, most notably by HuffPost.[50] The condition featured as a minor storyline in an episode of the hit U.K. drama Doc Martin series two, episode 4.[51]
In Alexander Payne’s 2023 movie The Holdovers, the central character Paul Hunham (played by Paul Giamatti), has the condition.[52]
References
- ↑ "Trimethylaminuria: the fish malodor syndrome". Drug Metab Dispos 29 (4 Pt 2): 517–21. 2001. PMID 11259343.
- ↑ 2.0 2.1 Dolphin, Colin T.; Janmohamed, Azara; Smith, Robert L.; Shephard, Elizabeth A.; Phillips, lan R. (1997). "Missense mutation in flavin-containing mono-oxygenase 3 gene, FMO3, underlies fish-odour syndrome" (in en). Nature Genetics 17 (4): 491–494. doi:10.1038/ng1297-491. ISSN 1061-4036. https://www.nature.com/articles/ng1297-491.
- ↑ "Mutations of the flavin-containing monooxygenase gene (FMO3) cause trimethylaminuria, a defect in detoxication". Human Molecular Genetics 7 (5): 839–45. 1998. doi:10.1093/hmg/7.5.839. PMID 9536088.
- ↑ "Mild trimethylaminuria caused by common variants in FMO3 gene". Lancet 354 (9181): 834–5. 1999. doi:10.1016/S0140-6736(99)80019-1. PMID 10485731.
- ↑ "Individuals reporting idiopathic malodor production: demographics and incidence of trimethylaminuria". Am. J. Med. 124 (11): 1058–1063. 2011. doi:10.1016/j.amjmed.2011.05.030. PMID 21851918.
- ↑ 6.0 6.1 "Learning About Trimethylaminuria". http://www.genome.gov/11508983.
- ↑ Phillips, Ian R.; Shephard, Elizabeth A. (1993). "Primary Trimethylaminuria". GeneReviews® (University of Washington, Seattle). https://www.ncbi.nlm.nih.gov/books/NBK1103/.
- ↑ "Trimethylaminuria". National Library of Medicine. http://ghr.nlm.nih.gov/condition=trimethylaminuria.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Trimethylaminuria and a human FMO3 mutation database". Hum Mutat 22 (3): 209–13. 2003. doi:10.1002/humu.10252. PMID 12938085.
- ↑ "Identification of novel variants of the flavin-containing monooxygenase gene family in African Americans". Drug Metab Dispos 31 (2): 187–93. 2003. doi:10.1124/dmd.31.2.187. PMID 12527699.
- ↑ Guo, Yiran; Hwang, Liang-Dar; Li, Jiankang; Eades, Jason; Yu, Chung Wen; Mansfield, Corrine; Burdick-Will, Alexis; Chang, Xiao et al. (15 February 2017). "Genetic analysis of impaired trimethylamine metabolism using whole exome sequencing" (in en). BMC Medical Genetics 18 (1). doi:10.1186/s12881-017-0369-8. ISSN 1471-2350. PMID 28196478.
- ↑ "Molecular evolution and balancing selection in the flavin-containing monooxygenase 3 gene (FMO3)". Pharmacogenet Genomics 17 (10): 827–39. 2007. doi:10.1097/FPC.0b013e328256b198. PMID 17885620.
- ↑ "Office of Dietary Supplements - Choline" (in en). https://ods.od.nih.gov/factsheets/Choline-HealthProfessional/.
- ↑ 14.0 14.1 "Treatments of trimethylaminuria: where we are and where we might be heading". Drug Discov Today 25 (9): 1710–1717. 2020. doi:10.1016/j.drudis.2020.06.026. PMID 32615074.
- ↑ Kalnins, Gints; Kuka, Janis; Grinberga, Solveiga; Makrecka-Kuka, Marina; Liepinsh, Edgars; Dambrova, Maija; Tars, Kaspars (August 2015). "Structure and Function of CutC Choline Lyase from Human Microbiota Bacterium Klebsiella pneumoniae" (in en). Journal of Biological Chemistry 290 (35): 21732–21740. doi:10.1074/jbc.M115.670471. PMID 26187464.
- ↑ "Implication of Trimethylamine N-Oxide (TMAO) in Disease: Potential Biomarker or New Therapeutic Target". Nutrients 10 (10): 1398. 2018. doi:10.3390/nu10101398. PMID 30275434.
- ↑ 17.0 17.1 "A randomized crossover trial on the effect of plant-based compared with animal-based meat on trimethylamine-N-oxide and cardiovascular disease risk factors in generally healthy adults: Study With Appetizing Plantfood-Meat Eating Alternative Trial (SWAP-MEAT)". Am J Clin Nutr 112 (5): 1188–1199. 2020. doi:10.1093/ajcn/nqaa203. PMID 32780794.
- ↑ 18.0 18.1 Zhang, A. Q.; Mitchell, S. C.; Smith, R. L. (May 1999). "Dietary precursors of trimethylamine in man: a pilot study". Food and Chemical Toxicology 37 (5): 515–520. doi:10.1016/s0278-6915(99)00028-9. ISSN 0278-6915. PMID 10456680.
- ↑ Manning, Nigel J.; Allen, Elizabeth K.; Kirk, Richard J.; Sharrard, Mark J.; Smith, Edwin J. (2011-11-20). "Riboflavin-Responsive Trimethylaminuria in a Patient with Homocystinuria on Betaine Therapy". JIMD Reports 5: 71–75. doi:10.1007/8904_2011_99. ISBN 978-3-642-28095-5. ISSN 2192-8304. PMID 23430919.
- ↑ 20.0 20.1 20.2 20.3 Shephard, Elizabeth A; Treacy, Eileen P; Phillips, Ian R (30 November 2011). "Clinical utility gene card for: Trimethylaminuria". European Journal of Human Genetics 20 (3): 4–5. doi:10.1038/ejhg.2011.214. PMID 22126753.
- ↑ Lenherr, Nina; Berndt, Anita; Ritz, Nicole; Rudin, Christoph (10 June 2014). "Aerococcus urinae: a possible reason for malodorous urine in otherwise healthy children" (in en). European Journal of Pediatrics 173 (8): 1115–1117. doi:10.1007/s00431-014-2348-9. ISSN 0340-6199. PMID 24913181. http://link.springer.com/10.1007/s00431-014-2348-9.
- ↑ Gibb, Alan Patrick; Sivaraman, Baskaran (November 2013). "A Second Case of Foul Smelling Urine in a Boy Caused by Aerococcus urinae" (in en). Pediatric Infectious Disease Journal 32 (11): 1300–1301. doi:10.1097/INF.0b013e3182a64054. ISSN 0891-3668. PMID 24141805.
- ↑ Cashman, John R; Xiong, Yeng; Lin, Jing; Verhagen, Hans; van Poppel, Geert; van Bladeren, Peter J; Larsen-Su, Shelley; Williams, David E (1999-09-15). "In vitro and in vivo inhibition of human flavin-containing monooxygenase form 3 (FMO3) in the presence of dietary indoles". Biochemical Pharmacology 58 (6): 1047–1055. doi:10.1016/S0006-2952(99)00166-5. ISSN 0006-2952. PMID 10509757. https://www.sciencedirect.com/science/article/abs/pii/S0006295299001665.
- ↑ 24.0 24.1 "The Nose Knows... or Does it? Olfactory Reference Syndrome in Patients Presenting for Assessment of Unusual Body Odor". J Nerv Ment Dis 207 (3): 145–151. 2019. doi:10.1097/NMD.0000000000000933. PMID 30720598.
- ↑ Richter, JL (Apr 1996). "Diagnosis and treatment of halitosis.". Compendium of Continuing Education in Dentistry 17 (4): 370–2, 374–6 passim; quiz 388. PMID 9051972.
- ↑ Carranza's clinical periodontology (11th ed.). St. Louis, Mo.: Elsevier/Saunders. 2012. pp. 1333, 1334. ISBN 978-1-4377-0416-7.
- ↑ "Biochemical and clinical aspects of the human flavin-containing monooxygenase form 3 (FMO3) related to trimethylaminuria". Curr Drug Metab 4 (2): 151–170. 2003. doi:10.2174/1389200033489505. PMID 12678693.
- ↑ (in en) Trimethylaminuria (TMAU): history, diagnosis and genetics, 8 February 2013, https://www.youtube.com/watch?v=TZExedNx5Vo, retrieved 2023-10-02
- ↑ "Trimethylaminuria". Tidsskr Nor Laegeforen 141. September 2021. doi:10.4045/tidsskr.21.0142. PMID 34597008.
- ↑ Bockus, Abigail B.; Seibel, Brad A. (2016-06-01). "Trimethylamine oxide accumulation as a function of depth in Hawaiian mid-water fishes". Deep Sea Research Part I: Oceanographic Research Papers 112: 37–44. doi:10.1016/j.dsr.2016.03.005. ISSN 0967-0637. Bibcode: 2016DSRI..112...37B.
- ↑ Demarquoy, Jean; Georges, Béatrice; Rigault, Caroline; Royer, Marie-Charlotte; Clairet, Amélie; Soty, Maud; Lekounoungou, Serge; Le Borgne, Françoise (2004). "Radioisotopic determination of l-carnitine content in foods commonly eaten in Western countries" (in en). Food Chemistry 86 (1): 137–142. doi:10.1016/j.foodchem.2003.09.023. https://linkinghub.elsevier.com/retrieve/pii/S0308814603004886.
- ↑ "Impact of chronic dietary red meat, white meat, or non-meat protein on trimethylamine N-oxide metabolism and renal excretion in healthy men and women". European Heart Journal 40 (7): 583–594. 2019. doi:10.1093/eurheartj/ehy799. PMID 30535398.
- ↑ 34.0 34.1 "Choline : USDA ARS". https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/methods-and-application-of-food-composition-laboratory/mafcl-site-pages/choline/.
- ↑ "Diagnosis and phenotypic assessment of trimethylaminuria, and its treatment with riboflavin: 1H NMR spectroscopy and genetic testing". Orphanet J Rare Dis 18 (1): 14(1):222. 2019. doi:10.1186/s13023-019-1174-6. PMID 31533761.
- ↑ "Trimethylaminuria, fish odour syndrome: A new method of detection and response to treatment with metronidazole". J Inherit Metab Dis 18 (3): 306–312. 1995. doi:10.1007/bf00710420. PMID 7474897.
- ↑ "Effects of the dietary supplements, activated charcoal and copper chlorophyllin, on urinary excretion of trimethylamine in Japanese trimethylaminuria patients". Life Sci. 74 (22): 2739–2747. 2004. doi:10.1016/j.lfs.2003.10.022. PMID 15043988.
- ↑ al-Waiz, M.; Ayesh, R.; Mitchell, S. C.; Idle, J. R.; Smith, R. L. (1989). "Trimethylaminuria: the detection of carriers using a trimethylamine load test". Journal of Inherited Metabolic Disease 12 (1): 80–85. doi:10.1007/BF01805534. ISSN 0141-8955. PMID 2501587.
- ↑ 39.0 39.1 39.2 39.3 Mackay, Richard J.; McEntyre, Christopher J.; Henderson, Caroline; Lever, Michael; George, Peter M. (2011). "Trimethylaminuria: causes and diagnosis of a socially distressing condition". The Clinical Biochemist. Reviews 32 (1): 33–43. ISSN 1838-0212. PMID 21451776.
- ↑ "Office of Dietary Supplements - Carnitine" (in en). https://ods.od.nih.gov/factsheets/Carnitine-HealthProfessional/.
- ↑ Manning, Nigel J.; Allen, Elizabeth K.; Kirk, Richard J.; Sharrard, Mark J.; Smith, Edwin J. (2012). "Riboflavin-responsive trimethylaminuria in a patient with homocystinuria on betaine therapy". JIMD Reports 5: 71–75. doi:10.1007/8904_2011_99. ISBN 978-3-642-28095-5. ISSN 2192-8304. PMID 23430919.
- ↑ "Office of Dietary Supplements - Choline" (in en). https://ods.od.nih.gov/factsheets/Choline-Consumer/.
- ↑ Manning, Nigel. "TMAU – diagnostic testing at Sheffield Children's Hospital". https://www.rareconnect.org/uploads/documents/tmau-diagnostic-testing-at-sheffield-children-s-hospital.pdf.
- ↑ "Sheffield TMAU test back : And new ref ranges". https://www.meboblog.com/2017/02/sheffield-tmau-test-back-and-new-ref.html.
- ↑ "Comment on the NEW UK TMAU Test (Sheffield)" (in en-US). https://systemicbodyodor.blogspot.com/2018/04/comment-on-new-uk-tmau-test-sheffield.html.
- ↑ "UK TMAU Test Stats 2017-2021 : Activist uses FOI act". https://www.meboblog.com/2021/07/uk-tmau-test-stats-2017-2021-activist.html.
- ↑ Fraser-Andrews, E. A.; Manning, N. J.; Ashton, G. H. S.; Eldridge, P.; McGrath, J.; Menagé, H. du P. (2003). "Fish odour syndrome with features of both primary and secondary trimethylaminuria". Clinical and Experimental Dermatology 28 (2): 203–205. doi:10.1046/j.1365-2230.2003.01230.x. ISSN 0307-6938. PMID 12653714.
- ↑ "Trimethylaminuria: the fish-odour syndrome". Lancet 2 (7676): 770–1. 1970. doi:10.1016/S0140-6736(70)90241-2. PMID 4195988.
- ↑ Reporter, Metro News (July 2, 2014). "Rare condition leaves singer smelling of rotting fish". https://metro.co.uk/2014/07/02/trimethylaminuria-singer-cassie-graves-has-fish-odour-syndrome-4784276/.
- ↑ "'I Smell Like Rotting Fish': Woman Has Rare Fish Odour Syndrome". July 3, 2014. https://www.huffingtonpost.co.uk/2014/07/03/trimethylaminuria-woman-fish-syndrome-smell-rotting_n_5554223.html.
- ↑ "Aromatherapy". December 1, 2005. https://www.youtube.com/watch?v=vxzai4d2Mbw&pp=ygURI3NlcGl0bWF1YmFnaWR1aXQ%3D.
- ↑ "'Bah, humbug! 'The Holdovers' is a clunky, phony white-elephant gift of a movie". Oct 26, 2023. https://www.latimes.com/entertainment-arts/movies/story/2023-10-26/the-holdovers-review-alexander-payne-paul-giamatti.
External links
| Classification | |
|---|---|
| External resources |
This article incorporates public domain text from The U.S. National Library of Medicine and The National Human Genome Research Institute
 |
