Medicine:Carnosinemia
| Carnosinemia | |
|---|---|
| Other names | Carnosinase deficiency[1] or Aminoacyl-histidine dipeptidase deficiency,[2] |
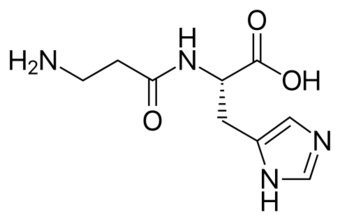 | |
| Carnosine | |
Carnosinemia is a rare autosomal recessive[3] metabolic disorder[4] caused by a deficiency of carnosinase, a dipeptidase (a type of enzyme that splits dipeptides into their two amino acid constituents).[5]
Carnosine is a dipeptide composed of beta-alanine and histidine, and is found in skeletal muscle and cells of the nervous system.[6] This disorder results in an excess of carnosine in the urine, cerebrospinal fluid, blood, and nervous tissue.[7] Neurological disorders associated with a deficiency of carnosinase, and the resulting carnosinemia ("carnosine in the blood") are common.[3][8][9]
Symptoms and signs
A variety of neurological symptoms have been associated with carnosinemia. They include: hypotonia, developmental delay, intellectual disability, degeneration of axons, sensory neuropathy, tremors, demyelinization, gray matter anomalies, myoclonic seizures, and loss of purkinje fibers.[3][4][8][9]
Genetics
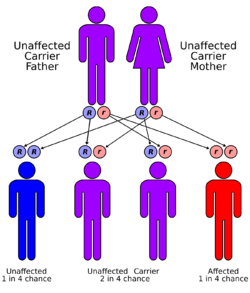
The gene for carnosinase is located on chromosome 18,[3] an autosome. The carnosine dipeptidase-1 gene (CNDP1) controls tissue and serum carnosinase.[10] Mutations in CNDP1 are responsible for carnosinase deficiency, resulting in carnosinemia.[3]
Carnosinemia is an autosomal recessive disorder,[3] which means the defective gene is located on an autosome, and two copies of the defective gene - one from each parent - are required to inherit the disorder. The parents of an individual with an autosomal recessive disorder both carry one copy of the defective gene, but usually do not experience any signs or symptoms of the disorder.[citation needed]
Diagnosis
Types
Carnosinase in humans has two forms:[11][12][13][14]
1. Cellular, or tissue carnosinase:[12] This form of the enzyme is found in every bodily tissue. It is a dimer, and hydrolyzes both carnosine and anserine, preferring dipeptides that have a histidine monomer in the C-terminus position.[11][12] Tissue carnosinase is often considered a "nonspecific dipeptidase",[13][15] based in part on its ability to hydrolyze a range of dipeptide substrates, including those belonging to prolinase.[16]
2. Serum carnosinase:[14] This is the carnosinase found in the blood plasma. Deficiency of this form of carnosinase, along with carnosinuria ("carnosine in the urine"), is the usual metabolic indicator of systemic carnosinase deficiency.[3][8][17] Serum carnosinase is a glycoprotein, and splits free carnosine and anserine in the blood.[11] This form of the dipeptidase is not found in human blood until late infancy, slowly rising to adult levels by age 15.[14] Unlike tissue carnosinase, serum carnosinase also hydrolyzes the GABA metabolite homocarnosine.[11] Homocarnosinosis, a neurological disorder resulting in an excess of homocarnosine in the brain, though unaffected by tissue carnosinase, is caused by a deficiency of serum carnosinase in its ability to hydrolyze homocarnosine.[18]
A deficiency of tissue and serum carnosinase, with serum being an indicator, is the underlying metabolic cause of carnosinemia.[7][9]
Treatment
See also
References
- ↑ Online Mendelian Inheritance in Man (OMIM) 212200
- ↑ DDB 29672
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 "A deletion in the long arm of chromosome 18 in a child with serum carnosinase deficiency". Pediatr. Res. 41 (2): 210–213. 1997. doi:10.1203/00006450-199702000-00009. PMID 9029640.
- ↑ 4.0 4.1 "Carnosinemia. A new metabolic disorder associated with neurological disease and mental defect". N. Engl. J. Med. 277 (23): 1219–1227. 1967. doi:10.1056/NEJM196712072772302. PMID 6058610.
- ↑ "L-carnosine, a substrate of carnosinase-1, influences glucose metabolism". Diabetes 56 (10): 2425–2432. 2007. doi:10.2337/db07-0177. PMID 17601992.
- ↑ "Carnosine and its constituents inhibit glycation of low-density lipoproteins that promotes foam cell formation in vitro". FEBS Lett. 581 (5): 1067–1070. 2007. doi:10.1016/j.febslet.2007.01.082. PMID 17316626.
- ↑ 7.0 7.1 "Inborn errors of carnosine and homocarnosine metabolism". J Neural Transm Suppl 29: 91–106. 1990. doi:10.1007/978-3-7091-9050-0_10. ISBN 978-3-211-82142-8. PMID 2358806.
- ↑ 8.0 8.1 8.2 "Histopathology of the nervous system in carnosinase enzyme deficiency with mental retardation". Neurology 22 (6): 644–655. 1972. doi:10.1212/wnl.22.6.644. PMID 4673339.
- ↑ 9.0 9.1 9.2 "Neurological diseases in a child with carnosinase deficiency". Neuropediatrics 12 (2): 143–151. 1981. doi:10.1055/s-2008-1059647. PMID 7266778.
- ↑ "Allelic variation in the CNDP1 gene and its lack of association with longevity and coronary heart disease". Mech Ageing Dev. 127 (11): 817–820. 2006. doi:10.1016/j.mad.2006.08.002. PMID 16965804.
- ↑ 11.0 11.1 11.2 11.3 "Purification and properties of human serum carnosinase". Clin Chim Acta 196 (2–3): 193–205. 1991. doi:10.1016/0009-8981(91)90073-L. PMID 1903095.
- ↑ 12.0 12.1 12.2 "Characterization of human tissue carnosinase". Biochem. J. 228 (3): 653–660. 1985. doi:10.1042/bj2280653. PMID 4026801.
- ↑ 13.0 13.1 Lenney JF (1990). "Separation and characterization of two carnosine-splitting cytosolic dipeptides from hog kidney (carnosinase and non-specific dipeptidase)". Biol Chem Hoppe-Seyler 371 (5): 433–440. doi:10.1515/bchm3.1990.371.1.433. PMID 2378680.
- ↑ 14.0 14.1 14.2 "Human serum carnosinase: characterization, distinction from cellular carnosinase and activation by cadmium". Clin Chim Acta 123 (3): 221–231. 1982. doi:10.1016/0009-8981(82)90166-8. PMID 7116644.
- ↑ "Bestatin inhibition of human tissue carnosinase, a non-specific cytosolic dipeptidase". Biol Chem Hoppe-Seyler 369 (12): 1281–1286. 1988. doi:10.1515/bchm3.1988.369.2.1281. PMID 3242551.
- ↑ Lenney JF (1990). "Human cytosolic carnosinase: evidence of identity with prolinase, a non-specific dipeptidase". Biol Chem Hoppe-Seyler 371 (2): 167–171. doi:10.1515/bchm3.1990.371.1.167. PMID 2334521.
- ↑ "A patient with a deficiency of serum-carnosinase activity". Acta Paediatr. Scand. 58 (6): 584–592. 1969. doi:10.1111/j.1651-2227.1969.tb04766.x. PMID 5378348.
- ↑ "Homocarnosinosis: lack of serum carnosinase is the deficiency probably responsible for elevated brain and CSF homocarnosine". Clin Chim Acta 132 (2): 157–165. 1983. doi:10.1016/0009-8981(83)90243-7. PMID 6616870.
External links
| Classification |
|---|
 |
