Biology:Chordate
A chordate (/ˈkɔːrdeɪt/ Template:Respelling) is a bilaterian animal belonging to the phylum Chordata (/kɔːrˈdeɪtə/ Template:Respelling). All chordates possess, at some point during their larval or adult stages, five distinctive physical characteristics (synapomorphies) that distinguish them from other taxa: a notochord, a hollow dorsal nerve cord, an endostyle or thyroid, pharyngeal slits, and a post-anal tail.[1]
In addition to the morphological characteristics used to define chordates, analysis of genome sequences has identified two conserved signature indels (CSIs) in their proteins: cyclophilin-like protein and inner mitochondrial membrane protease ATP23, which are exclusively shared by all vertebrates, tunicates and cephalochordates.[2] These CSIs provide molecular means to reliably distinguish chordates from all other animals.
Chordates are divided into three subphyla: Vertebrata (fish, amphibians, reptiles, birds and mammals), which possess a skull and whose notochords are replaced by a cartilaginous/bony axial endoskeleton (spine); Tunicata or Urochordata (sea squirts, salps, and larvaceans), which only retain the synapomorphies during their larval stage; and Cephalochordata (lancelets), which resemble jawless fish but have no gills or a distinct head. The vertebrates and tunicates compose the clade Olfactores, which is sister to Cephalochordata (see diagram under Phylogeny). Extinct taxa such as the conodonts are chordates, but their internal placement is less certain. Hemichordata (which includes the acorn worms) was previously considered a fourth chordate subphylum, but now is treated as a separate phylum which are now thought to be closer to the echinoderms, and together they form the clade Ambulacraria, the sister phylum of the chordates. Chordata, Ambulacraria, and possibly Xenacoelomorpha are believed to form the superphylum Deuterostomia, although this is called into doubt in a 2021 publication.[3]
Chordata is the third-largest phylum of the animal kingdom (behind only the protostomal phyla Arthropoda and Mollusca) and is also one of the most ancient animal taxa. Chordate fossils have been found from as early as the Cambrian explosion over 539 million years ago.[4] Of the more than 81,000 living species of chordates, about half are ray-finned fishes (class Actinopterygii) and the vast majority of the rest are tetrapods, a terrestrial clade of lobe-finned fishes (Sarcopterygii) who evolved air-breathing using lungs.[5]
Etymology
The name "chordate" comes from the first of these synapomorphies, the notochord, which plays a significant role in chordate body plan structuring and movements. Chordates are also bilaterally symmetric, have a coelom, possess a closed circulatory system, and exhibit metameric segmentation. Although the name Chordata is attributed to William Bateson (1885), it was already in prevalent use by 1880. Ernst Haeckel described a taxon comprising tunicates, cephalochordates, and vertebrates in 1866. Though he used the German vernacular form, it is allowed under the ICZN code because of its subsequent latinization.[6]
Anatomy

Chordates form a phylum of animals that are defined by having at some stage in their lives all of the following anatomical features:[7]
- A notochord, a stiff but elastic rod of glycoprotein wrapped in two collagen helices, which extends along the central axis of the body. Among members of the subphylum Vertebrata (vertebrates), the notochord gets replaced by hyaline cartilage or osseous tissue of the spine, and notochord remnants develop into the intervertebral discs, which allow adjacent spinal vertebrae to bend and twist relative to each other. In wholly aquatic species, this helps the animal swim efficiently by flexing its tail side-to-side.
- A hollow dorsal nerve cord, also known as the neural tube, which develops into the spinal cord, the main communications trunk of the nervous system. In vertebrates, the rostral end of the neural tube enlarges into several vesicles during embryonic development, which give rise to the brain.
- Pharyngeal slits. The pharynx is the part of the throat immediately behind the mouth. In fish, the slits are modified to form gills, but in some other chordates they are part of a filter-feeding system that extracts food particles from ingested water. In tetrapods, they are only present during embryonic stages of the development.
- A post-anal tail. A muscular tail that extends backwards beyond the location of the anus. In some chordates such as hominids, this is only present in the embryonic stage.
- An endostyle. This is a groove in the ventral wall of the pharynx. In filter-feeding species it produces mucus to gather food particles, which helps in transporting food to the esophagus.[8] It also stores iodine, and may be a precursor of the vertebrate thyroid gland.[7]
There are soft constraints that separate chordates from other biological lineages, but are not part of the formal definition:
- All chordates are deuterostomes, which means that, during embryonic development, the anus forms before the mouth does.
- All chordates are based on a bilateral body plan.[9]
- All chordates are coelomates, and have a fluid-filled body cavity (coelom) with a complete serosal lining derived from mesoderm called mesothelium (see Brusca and Brusca).[10]
Classification
The following schema is from the 2015 edition of Vertebrate Palaeontology.[12][13] The invertebrate chordate classes are from Fishes of the World.[14] While it is generally structured so as to reflect evolutionary relationships (similar to a cladogram), it also retains the traditional ranks used in Linnaean taxonomy.
- Phylum Chordata
- Subphylum Cephalochordata (Acraniata) – (lancelets; 30 species)
- Class Leptocardii (lancelets)
- Subphylum Tunicata (Urochordata) – (tunicates; 3,000 species)
- Class "Ascidiacea" (sea squirts; paraphyletic as thaliaceans are excluded)
- Class Thaliacea (salps, doliolids and pyrosomes)
- Class Appendicularia (larvaceans)
- Subphylum Vertebrata (Craniata) (vertebrates – animals with backbones; 66,100+ species)
- Infraphylum "Agnatha" paraphyletic (jawless vertebrates; 100+ species)
- Superclass Cyclostomata
- Class Myxinoidea or Myxini (hagfish; 65 species)
- Class Petromyzontida or Hyperoartia (lampreys)
- Class †Conodonta
- Class †Myllokunmingiida
- Class †Pteraspidomorphi
- Class †Thelodonti
- Class †Anaspida
- Class †Cephalaspidomorphi
- Superclass Cyclostomata
- Infraphylum Gnathostomata (jawed vertebrates)
- Class †"Placodermi" (Paleozoic armoured forms; paraphyletic in relation to all other gnathostomes)
- Class Chondrichthyes (cartilaginous fish; 900+ species)
- Class †"Acanthodii" (Paleozoic "spiny sharks"; paraphyletic in relation to Chondrichthyes)
- Class "Osteichthyes" (bony fish; 30,000+ species; paraphyletic when tetrapods are excluded)
- Subclass Actinopterygii (ray-finned fish; about 30,000 species)
- Subclass "Sarcopterygii" (lobe-finned fish: 8 species; paraphyletic when tetrapods are excluded)
- Superclass Tetrapoda (four-limbed vertebrates; 35,100+ species)[lower-alpha 1][15]
- Infraphylum "Agnatha" paraphyletic (jawless vertebrates; 100+ species)
- Subphylum Cephalochordata (Acraniata) – (lancelets; 30 species)
Subphyla

Cephalochordata: Lancelets
Cephalochordates, one of the three subdivisions of chordates, are small, "vaguely fish-shaped" animals that lack brains, clearly defined heads and specialized sense organs.[20] These burrowing filter-feeders compose the earliest-branching chordate subphylum.[21][22]
Tunicata (Urochordata)

The tunicates have three distinct adult shapes. Each is a member of one of three monophyletic clades. All tunicate larvae have the standard chordate features, including long, tadpole-like tails. Their larva also have rudimentary brains, light sensors and tilt sensors.[23]
The smallest of the three groups of tunicates is the Appendicularia. They retain tadpole-like shapes and active swimming all their lives, and were for a long time regarded as larvae of the other two groups.[24]
The other two groups, the sea squirts and the salps, metamorphize into adult forms which lose the notochord, nerve cord, and post-anal tail. Both are soft-bodied filter feeders with multiple gill slits. They feed on plankton which they collect in their mucus.
Sea squirts are sessile and consist mainly of water pumps and filter-feeding apparatus.[23] Most attach firmly to the sea floor, where they remain in one place for life, feeding on plankton.
The salps float in mid-water, feeding on plankton, and have a two-generation cycle in which one generation is solitary and the next forms chain-like colonies.[25]
The etymology of the term Urochordata (Balfour 1881) is from the ancient Greek οὐρά (oura, "tail") + Latin chorda ("cord"), because the notochord is only found in the tail.[26] The term Tunicata (Lamarck 1816) is recognised as having precedence and is now more commonly used.[23]
--------------------------------------------------------
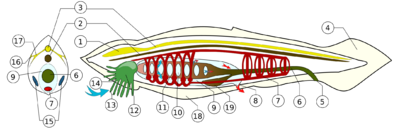
1. Notochord, 2. Nerve chord, 3. Buccal cirri, 4. Pharynx, 5. Gill slit, 6. Gonad, 7. Gut, 8. V-shaped muscles, 9. Anus, 10. Inhalant syphon, 11. Exhalant syphon, 12. Heart, 13. Stomach, 14. Esophagus, 15. Intestines, 16. Tail, 17. Atrium, 18. Tunic
Vertebrata (Craniata)

Craniates all have distinct skulls. They include the hagfish, which have no vertebrae. Michael J. Benton commented that "craniates are characterized by their heads, just as chordates, or possibly all deuterostomes, are by their tails".[27]
The notochord is accompanied or replaced by the vertebral column in most craniates.[28] It consists of a series of bony or cartilaginous cylindrical vertebrae, generally with neural arches that protect the spinal cord, and with projections that link the vertebrae. Hagfishes have incomplete braincases and no vertebrae, and were therefore regarded as non-vertebrate craniates.[29][30] However the cladistic exclusion of hagfish from the vertebrates is controversial, as they may instead be degenerate vertebrates who have secondarily lost their vertebral columns.[31]
Molecular phylogenetics, which uses DNA sequences to classify organisms, has generally supported the grouping of hagfish and lampreys (which have vertebrae) in a clade named Cyclostomata,[32][33] suggesting that the hagfish are indeed degenerate vertebrates.[31] Examination of fossil hagfish also supports this conclusion.[34] As a result, Craniata and Vertebrata are no longer different groups, as far as extant animals are concerned.[31]
Phylogeny
Overview

There is still much ongoing differential (DNA sequence based) comparison research that is trying to separate out the simplest forms of chordates. As some lineages of the 90% of species that lack a backbone or notochord might have lost these structures over time, this complicates the classification of chordates. Some chordate lineages may only be found by DNA analysis, when there is no physical trace of any chordate-like structures.[36]
Attempts to work out the evolutionary relationships of the chordates have produced several hypotheses. The current consensus is that chordates are monophyletic (meaning that the Chordata includes all and only the descendants of a single common ancestor, which is itself a chordate) and that the vertebrates' nearest relatives are tunicates. In 2016, identification of two conserved signature indels (CSIs) in the proteins cyclophilin-like protein and mitochondrial inner membrane protease ATP23, which are exclusively shared by all vertebrates, tunicates and cephalochordates also provided strong evidence of the monophyly of Chordata.[2]
All of the earliest chordate fossils have been found in the Early Cambrian Chengjiang fauna, and include three species that are regarded as fish, and hence vertebrates. Because the fossil record of early chordates is poor, only molecular phylogenetics offers a reasonable prospect of dating their emergence. However, the use of molecular phylogenetics for dating evolutionary transitions is controversial. It has proven difficult to produce a detailed classification within the living chordates. Attempts to produce evolutionary "family trees" shows that many of the traditional classes are paraphyletic.[citation needed]
| Deuterostomes |
| |||||||||||||||||||||||||||
While this has been well known since the 19th century, an insistence on only monophyletic taxa has resulted in vertebrate classification being in a state of flux.[37]
The majority of animals more complex than jellyfish and other cnidarians are split into two groups, the protostomes and deuterostomes, the latter of which contains chordates.[38] It seems very likely the 555 million-year-old Kimberella was a member of the protostomes.[39][40] If so, this means the protostome and deuterostome lineages must have split some time before Kimberella appeared—at least 558 million years ago, and hence well before the start of the Cambrian 541 million years ago.[38] Three enigmatic species that are possible very early tunicates, and therefore deuterostomes, were also found from the Ediacaran period – Ausia fenestrata from the Nama Group of Namibia, the sac-like Yarnemia ascidiformis, and one from a second new Ausia-like genus from the Onega Peninsula of northern Russia, Burykhia hunti. Results of a new study have shown possible affinity of these Ediacaran organisms to the ascidians.[41][42] Ausia and Burykhia lived in shallow coastal waters slightly more than 555 to 548 million years ago, and are believed to be the oldest evidence of the chordate lineage of metazoans.[42] The Russian Precambrian fossil Yarnemia is identified as a tunicate only tentatively, because its fossils are nowhere near as well-preserved as those of Ausia and Burykhia, so this identification has been questioned.


Fossils of one major deuterostome group, the echinoderms (whose modern members include starfish, sea urchins and crinoids), are quite common from the start of the Cambrian, 542 million years ago.[43] The Mid Cambrian fossil Rhabdotubus johanssoni has been interpreted as a pterobranch hemichordate.[44] Opinions differ about whether the Chengjiang fauna fossil Yunnanozoon, from the earlier Cambrian, was a hemichordate or chordate.[45][46] Another fossil, Haikouella lanceolata, also from the Chengjiang fauna, is interpreted as a chordate and possibly a craniate, as it shows signs of a heart, arteries, gill filaments, a tail, a neural chord with a brain at the front end, and possibly eyes—although it also had short tentacles round its mouth.[46] Haikouichthys and Myllokunmingia, also from the Chengjiang fauna, are regarded as fish.[35][47] Pikaia, discovered much earlier (1911) but from the Mid Cambrian Burgess Shale (505 Ma), is also regarded as a primitive chordate.[48] On the other hand, fossils of early chordates are very rare, since invertebrate chordates have no bones or teeth, and only one has been reported for the rest of the Cambrian.[49] The best known and earliest unequivocally identified Tunicate is Shankouclava shankouense from the Lower Cambrian Maotianshan Shale at Shankou village, Anning, near Kunming (South China).[50]
The evolutionary relationships between the chordate groups and between chordates as a whole and their closest deuterostome relatives have been debated since 1890. Studies based on anatomical, embryological, and paleontological data have produced different "family trees". Some closely linked chordates and hemichordates, but that idea is now rejected.[8] Combining such analyses with data from a small set of ribosome RNA genes eliminated some older ideas, but opened up the possibility that tunicates (urochordates) are "basal deuterostomes", surviving members of the group from which echinoderms, hemichordates and chordates evolved.[51] Some researchers believe that, within the chordates, craniates are most closely related to cephalochordates, but there are also reasons for regarding tunicates (urochordates) as craniates' closest relatives.[8][52]
Since early chordates have left a poor fossil record, attempts have been made to calculate the key dates in their evolution by molecular phylogenetics techniques—by analyzing biochemical differences, mainly in RNA. One such study suggested that deuterostomes arose before 900 million years ago and the earliest chordates around 896 million years ago.[52] However, molecular estimates of dates often disagree with each other and with the fossil record,[52] and their assumption that the molecular clock runs at a known constant rate has been challenged.[53][54]
A specific relationship between vertebrates and tunicates is also strongly supported by two CSIs found in the proteins predicted exosome complex RRP44 and serine palmitoyltransferase, that are exclusively shared by species from these two subphyla but not cephalochordates, indicating vertebrates are more closely related to tunicates than cephalochordates.[2]
Cladogram
Below is a phylogenetic tree of the phylum. Lines of the cladogram show probable evolutionary relationships between both extinct taxa, which are denoted with a dagger (†), and extant taxa.[55][56][57][58][59]
| Chordata |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Closest non-chordate relatives
The closest relatives of the chordates are believed to be the hemichordates and Echinodermata, which together form the Ambulacraria. The Chordata and Ambulacraria together form the superphylum Deuterostomia.
Hemichordates
Hemichordates ("half chordates") have some features similar to those of chordates: branchial openings that open into the pharynx and look rather like gill slits; stomochords, similar in composition to notochords, but running in a circle round the "collar", which is ahead of the mouth; and a dorsal nerve cord—but also a smaller ventral nerve cord.
There are two living groups of hemichordates. The solitary enteropneusts, commonly known as "acorn worms", have long proboscises and worm-like bodies with up to 200 branchial slits, are up to 2.5 metres (8.2 ft) long, and burrow though seafloor sediments. Pterobranchs are colonial animals, often less than 1 millimetre (0.039 in) long individually, whose dwellings are interconnected. Each filter feeds by means of a pair of branched tentacles, and has a short, shield-shaped proboscis. The extinct graptolites, colonial animals whose fossils look like tiny hacksaw blades, lived in tubes similar to those of pterobranchs.[60]
Echinoderms

Echinoderms differ from chordates and their other relatives in three conspicuous ways: they possess bilateral symmetry only as larvae – in adulthood they have radial symmetry, meaning that their body pattern is shaped like a wheel; they have tube feet; and their bodies are supported by dermal skeletons made of calcite, a material not used by chordates. Their hard, calcified shells keep their bodies well protected from the environment, and these skeletons enclose their bodies, but are also covered by thin skins. The feet are powered by another unique feature of echinoderms, a water vascular system of canals that also functions as a "lung" and surrounded by muscles that act as pumps. Crinoids are typically sessile and look rather like flowers (hence the common name "sea lilies"), and use their feather-like arms to filter food particles out of the water; most live anchored to rocks, but a few species can move very slowly. Other echinoderms are mobile and take a variety of body shapes, for example starfish and brittle stars, sea urchins and sea cucumbers.[61]
See also
- Biology:Chordate genomics – Study of the evolution of the chordate clade
- Biology:List of chordate orders – All the classes and orders of phylum Chordata
Notes
- ↑ The classification below follows Benton 2004, and uses a synthesis of rank-based Linnaean taxonomy and also reflects evolutionary relationships. Benton included the superclass Tetrapoda in the subclass Sarcopterygii in order to reflect the direct descent of tetrapods from lobe-finned fish, despite the former being assigned a higher taxonomic rank
References
- ↑ Freeborn, Michelle (2015-01-01). Roberts, Clive Douglas. ed. The fishes of New Zealand. Two. Te Papa Press. p. 6. ISBN 978-0-9941041-6-8. https://www.wikidata.org/wiki/Q58012425. Retrieved 29 May 2024.
- ↑ 2.0 2.1 2.2 Gupta, Radhey S. (January 2016). "Molecular signatures that are distinctive characteristics of the vertebrates and chordates and supporting a grouping of vertebrates with the tunicates". Molecular Phylogenetics and Evolution 94 (Pt A): 383–391. doi:10.1016/j.ympev.2015.09.019. ISSN 1055-7903. PMID 26419477. Bibcode: 2016MolPE..94..383G.
- ↑ Kapli, Paschalia; Natsidis, Paschalis; Leite, Daniel J.; Fursman, Maximilian; Jeffrie, Nadia; Rahman, Imran A.; Philippe, Hervé; Copley, Richard R. et al. (2021-03-19). "Lack of support for Deuterostomia prompts reinterpretation of the first Bilateria" (in en). Science Advances 7 (12). doi:10.1126/sciadv.abe2741. ISSN 2375-2548. PMID 33741592. Bibcode: 2021SciA....7.2741K.
- ↑ "Stratigraphic Chart 2022". International Stratigraphic Commission. February 2022. https://stratigraphy.org/ICSchart/ChronostratChart2022-02.pdf.
- ↑ "Chordates". https://eol.org/pages/694#:~:text=Chordata%20(Chordates)%20is%20a%20phylum,Vertebrates%2C%20Cephalochordates%2C%20and%20Tunicates..
- ↑ Nielsen, C. (July 2012). "The authorship of higher chordate taxa". Zoologica Scripta 41 (4): 435–436. doi:10.1111/j.1463-6409.2012.00536.x.
- ↑ 7.0 7.1 Rychel, A.L.; Smith, S.E.; Shimamoto, H.T.; Swalla, B.J. (March 2006). "Evolution and Development of the Chordates: Collagen and Pharyngeal Cartilage". Molecular Biology and Evolution 23 (3): 541–549. doi:10.1093/molbev/msj055. PMID 16280542.
- ↑ 8.0 8.1 8.2 8.3 Ruppert, E. (January 2005). "Key characters uniting hemichordates and chordates: homologies or homoplasies?". Canadian Journal of Zoology 83 (1): 8–23. doi:10.1139/Z04-158. Bibcode: 2005CaJZ...83....8R. http://article.pubs.nrc-cnrc.gc.ca/RPAS/RPViewDoc?_handler_=HandleInitialGet&articleFile=z04-158.pdf&journal=cjz&volume=83. Retrieved 2008-09-22.
- ↑ Valentine, J.W. (2004). On the Origin of Phyla. Chicago: University of Chicago Press. p. 7. ISBN 978-0-226-84548-7.
- ↑ R.C.Brusca, G.J.Brusca. Invertebrates. Sinauer Associates, Sunderland Mass 2003 (2nd ed.), p. 47, ISBN 0-87893-097-3.
- ↑ "Molecular regionalization of the developing amphioxus neural tube challenges major partitions of the vertebrate brain". PLOS Biology 15 (4). 2017. doi:10.1371/journal.pbio.2001573. PMID 28422959.
- ↑ Benton, M.J. (2004). Vertebrate Palaeontology, Third Edition. Blackwell Publishing. The classification scheme is available online
- ↑ Benton, Michael J. (2014). Vertebrate Palaeontology (4th ed.). John Wiley & Sons. ISBN 978-1-118-40764-6. https://books.google.co.uk/books?id=qak-BAAAQBAJ&pg=PP1.
- ↑ Nelson, J. S. (2006). Fishes of the World (4th ed.). New York: John Wiley and Sons, Inc. ISBN 978-0-471-25031-9.
- ↑ Benton, M.J. (2004). Vertebrate Paleontology. 3rd ed.. Blackwell Science Ltd.
- ↑ Frost, Darrel R.. "ASW Home". American Museum of Natural History, New York. http://research.amnh.org/herpetology/amphibia/index.html.
- ↑ "Reptiles face risk of extinction". BBC Nature. 15 February 2013. https://www.bbc.co.uk/nature/21458115.
- ↑ "New Study Doubles the Estimate of Bird Species in the World". Amnh.org. https://www.amnh.org/about-the-museum/press-center/new-study-doubles-the-estimate-of-bird-species-in-the-world.
- ↑ "Species Statistics Aug 2019". http://www.reptile-database.org/db-info/SpeciesStat.html.
- ↑ Benton, M.J. (14 April 2000). Vertebrate Palaeontology: Biology and Evolution. Blackwell Publishing. p. 6. ISBN 978-0-632-05614-9. https://books.google.com/books?id=PQuKO7xqjNQC&q=vertebrate. Retrieved 2008-09-22.
- ↑ Gee, H. (19 June 2008). "Evolutionary biology: The amphioxus unleashed". Nature 453 (7198): 999–1000. doi:10.1038/453999a. PMID 18563145. Bibcode: 2008Natur.453..999G.
- ↑ "Branchiostoma". Lander University. http://lanwebs.lander.edu/faculty/rsfox/invertebrates/branchiostoma.html.
- ↑ 23.0 23.1 23.2 Benton, M.J. (14 April 2000). Vertebrate Palaeontology: Biology and Evolution. Blackwell Publishing. p. 5. ISBN 978-0-632-05614-9. https://books.google.com/books?id=PQuKO7xqjNQC&q=vertebrate.
- ↑ "Appendicularia". Australian Government Department of the Environment, Water, Heritage and the Arts. http://www.environment.gov.au/biodiversity/abrs/publications/electronic-books/pubs/tunicates/05-appendicularia.pdf.
- ↑ "Animal fact files: salp". BBC. https://www.bbc.co.uk/nature/blueplanet/factfiles/jellies/salp_bg.shtml.
- ↑ Oxford English Dictionary, Third Edition, January 2009: Urochordata
- ↑ Benton, M.J. (14 April 2000). Vertebrate Palaeontology: Biology and Evolution. Blackwell Publishing. pp. 12–13. ISBN 978-0-632-05614-9. https://books.google.com/books?id=PQuKO7xqjNQC&q=vertebrate. Retrieved 2008-09-22.
- ↑ "Morphology of the Vertebrates". University of California Museum of Paleontology. http://www.ucmp.berkeley.edu/vertebrates/vertmm.html.
- ↑ "Introduction to the Myxini". University of California Museum of Paleontology. http://www.ucmp.berkeley.edu/vertebrates/basalfish/myxini.html.
- ↑ Campbell, N.A.; Reece, J.B. (2005). Biology (7th ed.). San Francisco, California: Benjamin Cummings. ISBN 978-0-8053-7171-0.
- ↑ 31.0 31.1 31.2 Janvier, P. (2010). "MicroRNAs revive old views about jawless vertebrate divergence and evolution". Proceedings of the National Academy of Sciences 107 (45): 19137–19138. doi:10.1073/pnas.1014583107. PMID 21041649. Bibcode: 2010PNAS..10719137J. "Although I was among the early supporters of vertebrate paraphyly, I am impressed by the evidence provided by Heimberg et al. and prepared to admit that cyclostomes are, in fact, monophyletic. The consequence is that they may tell us little, if anything, about the dawn of vertebrate evolution, except that the intuitions of 19th century zoologists were correct in assuming that these odd vertebrates (notably, hagfishes) are strongly degenerate and have lost many characters over time".
- ↑ Shigehiro Kuraku, S.; Hoshiyama, D.; Katoh, K.; Suga, H.; Miyata, T. (December 1999). "Monophyly of Lampreys and Hagfishes Supported by Nuclear DNA-Coded Genes". Journal of Molecular Evolution 49 (6): 729–735. doi:10.1007/PL00006595. PMID 10594174. Bibcode: 1999JMolE..49..729K.
- ↑ Delabre, Christiane (2002). "Complete Mitochondrial DNA of the Hagfish, Eptatretus burgeri: The Comparative Analysis of Mitochondrial DNA Sequences Strngly Supports the Cyclostome Monophyly". Molecular Phylogenetics and Evolution 22 (2): 184–192. doi:10.1006/mpev.2001.1045. PMID 11820840. Bibcode: 2002MolPE..22..184D.
- ↑ Miyashita, Tetsuto; Coates, Michael I.; Farrar, Robert; Larson, Peter; Manning, Phillip L. et al. (5 February 2019). "Hagfish from the Cretaceous Tethys Sea and a reconciliation of the morphological–molecular conflict in early vertebrate phylogeny". Proceedings of the National Academy of Sciences of the United States of America 116 (6): 2146–2151. doi:10.1073/pnas.1814794116. PMID 30670644. Bibcode: 2019PNAS..116.2146M.
- ↑ 35.0 35.1 Shu, D-G.; Conway Morris, S.; Han, J. (January 2003). "Head and backbone of the Early Cambrian vertebrate Haikouichthys". Nature 421 (6922): 526–529. doi:10.1038/nature01264. PMID 12556891. Bibcode: 2003Natur.421..526S.
- ↑ Josh Gabbatiss (15 August 2016), Why we have a spine when over 90% of animals don't, BBC, http://www.bbc.com/earth/story/20160812-why-we-have-a-spine-when-over-90-of-animals-dont, retrieved 10 September 2016
- ↑ Holland, N. D. (22 November 2005). "Chordates". Curr. Biol. 15 (22): R911–4. doi:10.1016/j.cub.2005.11.008. PMID 16303545. Bibcode: 2005CBio...15.R911H.
- ↑ 38.0 38.1 Erwin, Douglas H.; Eric H. Davidson (1 July 2002). "The last common bilaterian ancestor". Development 129 (13): 3021–3032. doi:10.1242/dev.129.13.3021. PMID 12070079. http://dev.biologists.org/cgi/content/full/129/13/3021. Retrieved 22 September 2008.
- ↑ {{{1}}} (2007), "{{{2}}}", in Vickers-Rich, Patricia; Komarower, Patricia, The Rise and Fall of the Ediacaran Biota, Special publications, 286, London: Geological Society, pp. {{{3}}}–{{{4}}}, doi:10.1144/SP286.{{{5}}}, ISBN 9781862392335, OCLC 156823511
- ↑ Butterfield, N.J. (December 2006). "Hooking some stem-group "worms": fossil lophotrochozoans in the Burgess Shale". BioEssays 28 (12): 1161–6. doi:10.1002/bies.20507. PMID 17120226. Bibcode: 2006BiEss..28.1161B.
- ↑ Vickers-Rich P. (2007). "Chapter 4. The Nama Fauna of Southern Africa". In: Fedonkin, M. A.; Gehling, J. G.; Grey, K.; Narbonne, G. M.; Vickers-Rich, P. "The Rise of Animals: Evolution and Diversification of the Kingdom Animalia", Johns Hopkins University Press. pp. 69–87
- ↑ 42.0 42.1 Fedonkin, M. A.; Vickers-Rich, P.; Swalla, B.; Trusler, P.; Hall, M. (2008). "A Neoproterozoic chordate with possible affinity to the ascidians: New fossil evidence from the Vendian of the White Sea, Russia and its evolutionary and ecological implications". HPF-07 Rise and fall of the Ediacaran (Vendian) biota. International Geological Congress - Oslo 2008.
- ↑ Bengtson, S. (2004). "Early skeletal fossils". The Paleontological Society Papers: Neoproterozoic – Cambrian Biological Revolutions 10: 67–78. doi:10.1017/S1089332600002345. http://www.nrm.se/download/18.4e32c81078a8d9249800021554/Bengtson2004ESF.pdf. Retrieved 2008-07-18.
- ↑ Bengtson, S.; Urbanek, A. (October 2007). "Rhabdotubus, a Middle Cambrian rhabdopleurid hemichordate". Lethaia 19 (4): 293–308. doi:10.1111/j.1502-3931.1986.tb00743.x. http://www3.interscience.wiley.com/journal/120025616/abstract.
- ↑ Shu, D.; Zhang, X.; Chen, L. (April 1996). "Reinterpretation of Yunnanozoon as the earliest known hemichordate". Nature 380 (6573): 428–430. doi:10.1038/380428a0. Bibcode: 1996Natur.380..428S.
- ↑ 46.0 46.1 Chen, J-Y.; Hang, D-Y.; Li, C.W. (December 1999). "An early Cambrian craniate-like chordate". Nature 402 (6761): 518–522. doi:10.1038/990080. Bibcode: 1999Natur.402..518C.
- ↑ Shu, D-G.; Conway Morris, S.; Zhang, X-L. (November 1999). "Lower Cambrian vertebrates from south China". Nature 402 (6757): 42. doi:10.1038/46965. Bibcode: 1999Natur.402...42S. http://www.bios.niu.edu/davis/bios458/Shu1.pdf. Retrieved 2008-09-23.
- ↑ Shu, D-G.; Conway Morris, S.; Zhang, X-L. (November 1996). "A Pikaia-like chordate from the Lower Cambrian of China". Nature 384 (6605): 157–158. doi:10.1038/384157a0. Bibcode: 1996Natur.384..157S.
- ↑ Conway Morris, S. (2008). "A Redescription of a Rare Chordate, Metaspriggina walcotti Simonetta and Insom, from the Burgess Shale (Middle Cambrian), British Columbia, Canada". Journal of Paleontology 82 (2): 424–430. doi:10.1666/06-130.1. Bibcode: 2008JPal...82..424M. http://jpaleontol.geoscienceworld.org/cgi/content/extract/82/2/424. Retrieved 2009-04-28.
- ↑ Chen, Jun-Yuan; Huang, Di-Ying; Peng, Qing-Qing; Chi, Hui-Mei; Wang, Xiu-Qiang; Feng, Man (2003). "The first tunicate from the Early Cambrian of South China". Proceedings of the National Academy of Sciences 100 (14): 8314–8318. doi:10.1073/pnas.1431177100. PMID 12835415. Bibcode: 2003PNAS..100.8314C.
- ↑ Winchell, C. J.; Sullivan, J.; Cameron, C. B.; Swalla, B. J.; Mallatt, J. (1 May 2002). "Evaluating Hypotheses of Deuterostome Phylogeny and Chordate Evolution with New LSU and SSU Ribosomal DNA Data". Molecular Biology and Evolution 19 (5): 762–776. doi:10.1093/oxfordjournals.molbev.a004134. PMID 11961109.
- ↑ 52.0 52.1 52.2 Blair, J. E.; Hedges, S. B. (November 2005). "Molecular Phylogeny and Divergence Times of Deuterostome Animals". Molecular Biology and Evolution 22 (11): 2275–2284. doi:10.1093/molbev/msi225. PMID 16049193.
- ↑ Ayala, F. J. (January 1999). "Molecular clock mirages". BioEssays 21 (1): 71–75. doi:10.1002/(SICI)1521-1878(199901)21:1<71::AID-BIES9>3.0.CO;2-B. PMID 10070256.
- ↑ Schwartz, J. H.; Maresca, B. (December 2006). "Do Molecular Clocks Run at All? A Critique of Molecular Systematics". Biological Theory 1 (4): 357–371. doi:10.1162/biot.2006.1.4.357.
- ↑ Putnam, N. H.; Butts, T.; Ferrier, D. E. K.; Furlong, R. F.; Hellsten, U.; Kawashima, T.; Robinson-Rechavi, M.; Shoguchi, E. et al. (June 2008). "The amphioxus genome and the evolution of the chordate karyotype". Nature 453 (7198): 1064–1071. doi:10.1038/nature06967. PMID 18563158. Bibcode: 2008Natur.453.1064P.
- ↑ Ota, K. G.; Kuratani, S. (September 2007). "Cyclostome embryology and early evolutionary history of vertebrates". Integrative and Comparative Biology 47 (3): 329–337. doi:10.1093/icb/icm022. PMID 21672842.
- ↑ "A phylogenomic framework and timescale for comparative studies of tunicates". BMC Biology 16 (1). April 2018. doi:10.1186/s12915-018-0499-2. PMID 29653534. Bibcode: 2018BMCB...16...39D.
- ↑ Goujet, Daniel F (2015-02-16), "Placodermi (Armoured Fishes)" (in en), ELS, John Wiley & Sons, Ltd, pp. 1–7, doi:10.1002/9780470015902.a0001533.pub2, ISBN 978-0-470-01590-2
- ↑ Miyashita, Tetsuto; Coates, Michael I.; Farrar, Robert; Larson, Peter; Manning, Phillip L.; Wogelius, Roy A. et al. (2019). "Hagfish from the Cretaceous Tethys Sea and a reconciliation of the morphological–molecular conflict in early vertebrate phylogeny". Proceedings of the National Academy of Sciences of the United States of America 116 (6): 2146–2151. doi:10.1073/pnas.1814794116. PMID 30670644. Bibcode: 2019PNAS..116.2146M.
- ↑ "Introduction to the Hemichordata". University of California Museum of Paleontology. http://www.ucmp.berkeley.edu/chordata/hemichordata.html.
- ↑ Cowen, R. (2000). History of Life (3rd ed.). Blackwell Science. p. 412. ISBN 978-0-632-04444-3.
External links
| Wikispecies has information related to Chordata |
| The Wikibook Dichotomous Key has a page on the topic of: Chordata |
Wikidata ☰ Q10915 entry
 |