Biology:Ribonuclease P
Ribonuclease P (EC 3.1.26.5, RNase P) is a type of ribonuclease which cleaves RNA. RNase P is unique from other RNases in that it is a ribozyme – a ribonucleic acid that acts as a catalyst in the same way that a protein-based enzyme would. Its function is to cleave off an extra, or precursor, sequence of RNA on tRNA molecules. Further, RNase P is one of two known multiple turnover ribozymes in nature (the other being the ribosome), the discovery of which earned Sidney Altman and Thomas Cech the Nobel Prize in Chemistry in 1989: in the 1970s, Altman discovered the existence of precursor tRNA with flanking sequences and was the first to characterize RNase P and its activity in processing of the 5' leader sequence of precursor tRNA.[1]
Its best characterised enzyme activity is the generation of mature 5′-ends of tRNAs by cleaving the 5′-leader elements of precursor-tRNAs. Cellular RNase Ps are ribonucleoproteins. The RNA from bacterial RNase P retains its catalytic activity in the absence of the protein subunit, i.e. it is a ribozyme. Similarly, archaeal RNase P RNA has been shown to be weakly catalytically active in the absence of its respective protein cofactors.[2] Isolated eukaryotic RNase P RNA has not been shown to retain its catalytic function, but is still essential for the catalytic activity of the holoenzyme. Although the archaeal and eukaryotic holoenzymes have a much greater protein content than the bacterial ones, the RNA cores from all three lineages are homologous—the helices corresponding to P1, P2, P3, P4, and P10/11 are common to all cellular RNase P RNAs. Yet there is considerable sequence variation, particularly among the eukaryotic RNAs.
In Bacteria
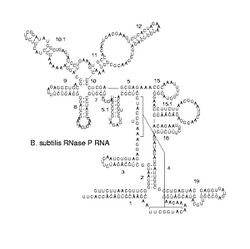
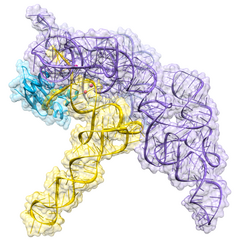
Bacterial RNase P has two components: an RNA chain, called M1 RNA, and a polypeptide chain, or protein, called C5 protein.[3][4] In vivo, both components are necessary for the ribozyme to function properly, but in vitro, the M1 RNA can act alone as a catalyst.[1] The primary role of the C5 protein is to enhance the substrate binding affinity and the catalytic rate of the M1 RNA enzyme probably by increasing the metal ion affinity in the active site. The crystal structure of a bacterial RNase P holoenzyme with tRNA has been recently resolved, showing how the large, coaxially stacked helical domains of the RNase P RNA engage in shape selective recognition of the pre-tRNA target. This crystal structure confirms earlier models of substrate recognition and catalysis, identifies the location of the active site, and shows how the protein component increases RNase P functionality.[5][6]
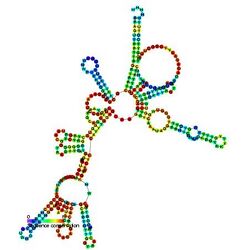
Bacterial RNase P class A and B
Ribonuclease P (RNase P) is a ubiquitous endoribonuclease, found in archaea, bacteria and eukarya as well as chloroplasts and mitochondria. Its best characterised activity is the generation of mature 5'-ends of tRNAs by cleaving the 5'-leader elements of precursor-tRNAs. Cellular RNase Ps are ribonucleoproteins (RNP). RNA from bacterial RNase Ps retains its catalytic activity in the absence of the protein subunit, i.e. it is a ribozyme. Isolated eukaryotic and archaeal RNase P RNA has not been shown to retain its catalytic function, but is still essential for the catalytic activity of the holoenzyme. Although the archaeal and eukaryotic holoenzymes have a much greater protein content than the eubacterial ones, the RNA cores from all the three lineages are homologous—helices corresponding to P1, P2, P3, P4, and P10/11 are common to all cellular RNase P RNAs. Yet, there is considerable sequence variation, particularly among the eukaryotic RNAs.
|
| ||||||||||||||||||||||||||||||||||||||||||||
In Archaea
In archaea, RNase P ribonucleoproteins consist of 4–5 protein subunits that are associated with RNA. As revealed by in vitro reconstitution experiments these protein subunits are individually dispensable for tRNA processing that is essentially mediated by the RNA component.[7][8][9] The structures of protein subunits of archaeal RNase P have been resolved by x-ray crystallography and NMR, thus revealing new protein domains and folding fundamental for function.
Using comparative genomics and improved computational methods, a radically minimized form of the RNase P RNA, dubbed "Type T", has been found in all complete genomes in the crenarchaeal phylogenetic family Thermoproteaceae, including species in the genera Pyrobaculum, Caldivirga and Vulcanisaeta.[10] All retain a conventional catalytic domain, but lack a recognizable specificity domain. 5′ tRNA processing activity of the RNA alone was experimentally confirmed. The Pyrobaculum and Caldivirga RNase P RNAs are the smallest naturally occurring form yet discovered to function as trans-acting ribozymes.[10] Loss of the specificity domain in these RNAs suggests potential altered substrate specificity.
It has recently been argued that the archaebacterium Nanoarchaeum equitans does not possess RNase P. Computational and experimental studies failed to find evidence for its existence. In this organism the tRNA promoter is close to the tRNA gene and it is thought that transcription starts at the first base of the tRNA thus removing the requirement for RNase P.[11]
|
| ||||||||||||||||||||||||||||||||||||||||||
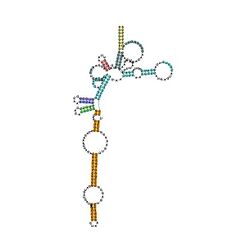
In eukaryotes
Nuclear RNase P
| Nuclear RNase P | |
|---|---|
 Predicted secondary structure and sequence conservation of RNaseP_nuc | |
| Identifiers | |
| Symbol | RNaseP_nuc |
| Rfam | RF00009 |
| Other data | |
| RNA type | Gene; ribozyme |
| Domain(s) | Eukaryota; Bacteria; Archaea |
| GO | 0008033 0004526 0030677 |
| SO | 0000386 |
| PDB structures | PDBe |
In eukaryotes, such as humans and yeast,[12] most RNase P consists of an RNA chain that is structurally similar to that found in bacteria [13] as well as nine to ten associated proteins (as opposed to the single bacterial RNase P protein, C5).[14][15] Five of these protein subunits exhibit homology to archaeal counterparts.
Recent (2007) findings also reveal that eukaryotic RNase P has a new function:[14] It has been shown that human nuclear RNase P is required for the normal and efficient transcription of various small noncoding RNAs, such as tRNA, 5S rRNA, SRP RNA and U6 snRNA genes,[16] which are transcribed by RNA polymerase III, one of three major nuclear RNA polymerases in human cells.
RNase P from eukaryotes was only recently (2007) demonstrated to be a ribozyme.[17] Accordingly, the numerous protein subunits of eukaryotic RNase P have a minor contribution to tRNA processing per se,[18] while they seem to be essential for the function of RNase P and RNase MRP in other biological settings, such as gene transcription and the cell cycle.[16][19]
| Subunit | Function/interaction (in tRNA processing) |
|---|---|
| RPP14 | RNA binding |
| RPP20 | ATPase, helicase/Hsp27, SMN, Rpp25 |
| RPP21 | RNA binding, activityg/Rpp29 |
| RPP25 | RNA binding/Rpp20 |
| RPP29 | tRNA binding, activity/Rpp21 |
| RPP30 | RNA binding, activity/Pop5 |
| RPP38 | RNA binding, activity |
| RPP40 | |
| hPop1 | |
| hPop5 | RNA binding, activity/Rpp30 |
| H1 RNA | Activity/Rpp21, Rpp29, Rpp30, Rpp38 |
RNase MRP
Protein subunits of RNase P are shared with RNase MRP,[15][20][21] an evolutionarily related catalytic ribonucleoprotein involved in processing of ribosomal RNA in the nucleolus and[22] DNA replication in the mitochondrion.[23]
Organellar RNase P
Most eukaryotes have mitochondria, an organelle derived from proteobacteria, or a reduced version. Some also have chloroplasts, an organelle derived from cyanobacteria. These organelles have their own genome and machinery for transcription and translation. They make their own tRNAs, which requires maturation by RNase P.
Bacteria-derived
As expected for the endosymbiotic theory – and similarly to other organellar genes – RNase P RNA genes (Rpm1 in yeast nomenclature or rnpB in bacterial nomenclature) have in the mitochondrial genome of the baker's yeast and most of its relatives in Saccharomycetales. These RNAs are extremely minimized ("crippled" according to Rossmanith) and do not work alone in vitro. They also show high divergence even among related yeasts. The baker's yeast version has one identified protein partner Rpm2, the only protein partner to mitochondrial RNase P RNA a known as of 2012. The identification of rnpB in the broader category of fungi remains patchy.[24]
Among Archaeplastida ("broader plants": plants, green algae, red algae), only two early-branching prasinophytes have a mtRNase P RNA gene. The secondary structure resembles α-proteobacterial RNase P RNA, but they do not work alone in vitro. It is unknown what the required protein partner is.[24] The glaucophytes, red algae, and some prasinophytes have a bacterial type A RNase P RNA in their chloroplast genomes. Other plants use the protein-only system described below.[24]
The situation among so-called protists is less clear due to the lack of data. The jakobid Reclinomonas americana is notable for having a mtRNase P RNA believed to be the closest to the version in the proto-mitochondrion, though it also does not work alone in vitro.[24] (Mixing and matching parts from this RNA with the E. coli P-RNA does produce an RNA that is active alone.)[25]
No mitochondrial mtRNase P RNA has been found among animals as of 2012. Most of them have a copy of the protein-only system identified.[24]
Eukaryotic protein-only RNase P
| Protein-only RNase P, C-terminal catalytic | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | PRORP_C | ||||||||
| Pfam | PF16953 | ||||||||
| InterPro | IPR031595 | ||||||||
| |||||||||
The alternative to a RNA-based RNase P in animals and plants is the protein-only RNase P (PRORP). Most PRORPs consist of a C-terminal metallonuclease PIN domain and an N-terminal pentatricopeptide repeat (PPR) domain,[26] but variations exist.[27]
The PRORP was originally discovered in plants, specifically in Spinach chloroplasts.[28] The model plant Arabidopsis thaliana has three protein-only RNase P genes: PRORP1, PRORP2, PRORP3. PRORP1 goes to the mitochondria and chloroplasts while PRORP2 and PRORP3 stays in the nucleus. All can cleave tRNA in vitro. The plant version has all functionalities in one chain of protein.[29]
Human mitochondrial RNase P is a trimeric protein and does not contain RNA. It consists of TRMT10C, HSD17B10, and the catalytic PRORP.[30] Its structure has been solved. The PPR domain in human PRORP does not perform base recognition, unlike in plant single-protein PRORPs.[31] Other animals have a similar setup. Even nematodes have a divergent version of this trimeric system.[24]
The kinetoplastids also have PRORP.[24]
Prokaryotic protein-only RNase P
| RNA-free ribonuclease P / PINc domain ribonuclease | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | PIN_5 | ||||||||
| Pfam | PF08745 | ||||||||
| InterPro | IPR014856 | ||||||||
| |||||||||
Some prokrayotes (bacteria and archaea) have a single-protein RNase P quite different from the eukaryotic PRORP. They are called HARP (Homologs of Aquifex RNase P). They are tiny proteins of up to 23 kDa. They only share sequence similarity with eukaryotic PRORP in one region, the metallonuclease PIN domain. Some of them form high-order oligomers.[32]
HARP does not carry out the functions of RNase P very efficiently. Many organisms that have HARP also have a typical RNase P. This hints at a different function. In 2025, it was found that HARP has a tendency to appear with the RNA ligase protein. A new theory backed by immunodepletion is that it acts with RNA ligase to mature and circularize C/D box snoRNAs.[33]
Therapies using RNase P
RNase P is now being studied as a potential therapy for diseases such as herpes simplex virus,[34] cytomegalovirus,[34][35] influenza and other respiratory infections,[36] HIV-1[37] and cancer caused by fusion gene BCR-ABL.[34][38] External guide sequences (EGSs) are formed with complementarity to viral or oncogenic mRNA and structures that mimic the T loop and acceptor stem of tRNA.[36] These structures allow RNase P to recognize the EGS and cleave the target mRNA. EGS therapies have shown to be effective in culture and in live mice.[39]
References
- ↑ 1.0 1.1 "The RNA moiety of ribonuclease P is the catalytic subunit of the enzyme". Cell 35 (3 Pt 2): 849–57. December 1983. doi:10.1016/0092-8674(83)90117-4. PMID 6197186.
- ↑ Tsai, H.Y.; Pulukkunat, D.K.; Woznick, W.K.; Gopalan, V. (2006). "Functional reconstitution and characterization of Pyrococcus furiosus RNase P". PNAS 103 (44): 16147–16152. doi:10.1073/pnas.0608000103. PMID 17053064. Bibcode: 2006PNAS..10316147T.
- ↑ "RNase P: interface of the RNA and protein worlds". Trends in Biochemical Sciences 31 (6): 333–41. June 2006. doi:10.1016/j.tibs.2006.04.007. PMID 16679018.
- ↑ "Molecular modeling of the three-dimensional structure of the bacterial RNase P holoenzyme". Journal of Molecular Biology 325 (4): 661–75. January 2003. doi:10.1016/S0022-2836(02)01267-6. PMID 12507471. http://www-ibmc.u-strasbg.fr/upr9002/westhof/PDF/2003_HTsai_JMB.pdf. Retrieved 2019-09-24.
- ↑ "Structure of a bacterial ribonuclease P holoenzyme in complex with tRNA". Nature 468 (7325): 784–9. December 2010. doi:10.1038/nature09516. PMID 21076397. Bibcode: 2010Natur.468..784R.
- ↑ "RNase P: at last, the key finds its lock". RNA 17 (9): 1615–8. September 2011. doi:10.1261/rna.2841511. PMID 21803972.
- ↑ "Archaeal RNase P has multiple protein subunits homologous to eukaryotic nuclear RNase P proteins". RNA 8 (3): 296–306. March 2002. doi:10.1017/S1355838202028492. PMID 12003490.
- ↑ "A fifth protein subunit Ph1496p elevates the optimum temperature for the ribonuclease P activity from Pyrococcus horikoshii OT3". Biochemical and Biophysical Research Communications 343 (3): 956–64. May 2006. doi:10.1016/j.bbrc.2006.02.192. PMID 16574071. Bibcode: 2006BBRC..343..956F.
- ↑ "Functional reconstitution and characterization of Pyrococcus furiosus RNase P". Proceedings of the National Academy of Sciences of the United States of America 103 (44): 16147–52. October 2006. doi:10.1073/pnas.0608000103. PMID 17053064. Bibcode: 2006PNAS..10316147T.
- ↑ 10.0 10.1 "Discovery of a minimal form of RNase P in Pyrobaculum". Proceedings of the National Academy of Sciences of the United States of America 107 (52): 22493–8. December 2010. doi:10.1073/pnas.1013969107. PMID 21135215. Bibcode: 2010PNAS..10722493L.
- ↑ "Life without RNase P". Nature 453 (7191): 120–3. May 2008. doi:10.1038/nature06833. PMID 18451863. Bibcode: 2008Natur.453..120R.
- ↑ Randall Munroe rephrased this as "You know, eukaryotes—like sourdough starter or Conan O'Brien." (Munroe, Randall (30 September 2022). "4:25 PM". https://twitter.com/xkcd/status/1575944901090455552.)
- ↑ "Structure and function of eukaryotic Ribonuclease P RNA". Molecular Cell 24 (3): 445–56. November 2006. doi:10.1016/j.molcel.2006.09.011. PMID 17081993.
- ↑ 14.0 14.1 14.2 "Human RNase P: a tRNA-processing enzyme and transcription factor". Nucleic Acids Research 35 (11): 3519–24. 2007. doi:10.1093/nar/gkm071. PMID 17483522.
- ↑ 15.0 15.1 "Purification and characterization of the nuclear RNase P holoenzyme complex reveals extensive subunit overlap with RNase MRP". Genes & Development 12 (11): 1678–90. June 1998. doi:10.1101/gad.12.11.1678. PMID 9620854.
- ↑ 16.0 16.1 "A role for the catalytic ribonucleoprotein RNase P in RNA polymerase III transcription". Genes & Development 20 (12): 1621–35. June 2006. doi:10.1101/gad.386706. PMID 16778078.
- ↑ "Eukaryotic RNase P RNA mediates cleavage in the absence of protein". Proceedings of the National Academy of Sciences of the United States of America 104 (7): 2062–7. February 2007. doi:10.1073/pnas.0607326104. PMID 17284611.
- ↑ "An important piece of the RNase P jigsaw solved". Trends in Biochemical Sciences 32 (6): 247–50. June 2007. doi:10.1016/j.tibs.2007.04.005. PMID 17485211.
- ↑ "RNase MRP cleaves the CLB2 mRNA to promote cell cycle progression: novel method of mRNA degradation". Molecular and Cellular Biology 24 (3): 945–53. February 2004. doi:10.1128/MCB.24.3.945-953.2004. PMID 14729943.
- ↑ "Characterization and purification of Saccharomyces cerevisiae RNase MRP reveals a new unique protein component". The Journal of Biological Chemistry 280 (12): 11352–60. March 2005. doi:10.1074/jbc.M409568200. PMID 15637077.
- ↑ "Differential association of protein subunits with the human RNase MRP and RNase P complexes". RNA 12 (7): 1373–82. July 2006. doi:10.1261/rna.2293906. PMID 16723659.
- ↑ "A big development for a small RNA". Nature 410 (6824): 29–31. March 2001. doi:10.1038/35065191. PMID 11242026.
- ↑ "Phylogenetic analysis of the structure of RNase MRP RNA in yeasts". RNA 8 (6): 740–51. June 2002. doi:10.1017/S1355838202022082. PMID 12088147.
- ↑ 24.0 24.1 24.2 24.3 24.4 24.5 24.6 Rossmanith, W (September 2012). "Of P and Z: mitochondrial tRNA processing enzymes.". Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms 1819 (9–10): 1017–26. doi:10.1016/j.bbagrm.2011.11.003. PMID 22137969.
- ↑ Seif, E; Cadieux, A; Lang, BF (September 2006). "Hybrid E. coli--Mitochondrial ribonuclease P RNAs are catalytically active.". RNA 12 (9): 1661–70. doi:10.1261/rna.52106. PMID 16894220.
- ↑ Harris, Michael E. (January 2016). "Theme and Variation in tRNA 5′ End Processing Enzymes: Comparative Analysis of Protein versus Ribonucleoprotein RNase P". Journal of Molecular Biology 428 (1): 5–9. doi:10.1016/j.jmb.2015.12.001. PMID 26655024.
- ↑ InterPro IPR031595, domain architecture.
- ↑ "Chloroplast ribonuclease P does not utilize the ribozyme-type pre-tRNA cleavage mechanism". RNA 6 (4): 545–53. April 2000. doi:10.1017/S1355838200991465. PMID 10786845.
- ↑ Karasik, A; Shanmuganathan, A; Howard, MJ; Fierke, CA; Koutmos, M (16 January 2016). "Nuclear Protein-Only Ribonuclease P2 Structure and Biochemical Characterization Provide Insight into the Conserved Properties of tRNA 5' End Processing Enzymes.". Journal of Molecular Biology 428 (1): 26–40. doi:10.1016/j.jmb.2015.11.025. PMID 26655022.
- ↑ "RNase P without RNA: identification and functional reconstitution of the human mitochondrial tRNA processing enzyme". Cell 135 (3): 462–74. October 2008. doi:10.1016/j.cell.2008.09.013. PMID 18984158.
- ↑ Bhatta, Arjun; Dienemann, Christian; Cramer, Patrick; Hillen, Hauke S. (September 2021). "Structural basis of RNA processing by human mitochondrial RNase P". Nature Structural & Molecular Biology 28 (9): 713–723. doi:10.1038/s41594-021-00637-y. PMID 34489609.
- ↑ Wilhelm, Catherine A.; Mallik, Leena; Kelly, Abigail L.; Brotzman, Shayna; Mendoza, Johnny; Anders, Anna G.; Leskaj, Suada; Castillo, Carmen et al. (November 2023). "Bacterial RNA-free RNase P: Structural and functional characterization of multiple oligomeric forms of a minimal protein-only ribonuclease P". Journal of Biological Chemistry 299 (11). doi:10.1016/j.jbc.2023.105327. PMID 37806495.
- ↑ Seshadri, R; Gopalan, V (29 April 2025). "An RNA ligase partner for the prokaryotic protein-only RNase P: insights into the functional diversity of RNase P from genome mining.". mBio 16 (6): e0044925. doi:10.1128/mbio.00449-25. PMID 40298408.
- ↑ 34.0 34.1 34.2 "Developing RNase P ribozymes for gene-targeting and antiviral therapy". Cellular Microbiology 6 (6): 499–508. June 2004. doi:10.1111/j.1462-5822.2004.00398.x. PMID 15104592.
- ↑ "RNase P ribozymes for the studies and treatment of human cytomegalovirus infections". Journal of Clinical Virology 25 (Suppl 2): S63-74. August 2002. doi:10.1016/s1386-6532(02)00097-5. PMID 12361758.
- ↑ 36.0 36.1 "Gene silencing in the therapy of influenza and other respiratory diseases: Targeting to RNase P by use of External Guide Sequences (EGS)". Biologics: Targets and Therapy 1 (4): 425–32. December 2007. PMID 19707312.
- ↑ "Effective inhibition of human immunodeficiency virus 1 replication by engineered RNase P ribozyme". PLOS ONE 7 (12). December 26, 2012. doi:10.1371/journal.pone.0051855. PMID 23300569. Bibcode: 2012PLoSO...751855Z.
- ↑ "In vivo inhibition by a site-specific catalytic RNA subunit of RNase P designed against the BCR-ABL oncogenic products: a novel approach for cancer treatment". Blood 95 (3): 731–7. February 2000. doi:10.1182/blood.V95.3.731.003k28_731_737. PMID 10648380.
- ↑ "A peptide-morpholino oligomer conjugate targeting Staphylococcus aureus gyrA mRNA improves healing in an infected mouse cutaneous wound model". International Journal of Pharmaceutics 453 (2): 651–5. September 2013. doi:10.1016/j.ijpharm.2013.05.041. PMID 23727592.
Further reading
- "Ribonuclease P: unity and diversity in a tRNA processing ribozyme". Annual Review of Biochemistry 67: 153–80. 1998. doi:10.1146/annurev.biochem.67.1.153. PMID 9759486.
- "The Ribonuclease P Database". Nucleic Acids Research 27 (1): 314. January 1999. doi:10.1093/nar/27.1.314. PMID 9847214.
- "Phylogenetic-comparative analysis of the eukaryal ribonuclease P RNA". RNA 6 (12): 1895–1904. December 2000. doi:10.1017/S1355838200001461. PMID 11142387.
- "Eukaryotic ribonuclease P: a plurality of ribonucleoprotein enzymes". Annual Review of Biochemistry 71: 165–189. 2002. doi:10.1146/annurev.biochem.71.110601.135352. PMID 12045094.
- "Structural implications of novel diversity in eucaryal RNase P RNA". RNA 11 (5): 739–751. May 2005. doi:10.1261/rna.7211705. PMID 15811915.
External links
- Nobel Lecture of Sidney Altman, Nobel prize in Chemistry 1989
- RNase P Database at ncsu.edu
- Page for Nuclear RNase P at Rfam
- Page for Archaeal RNase P at Rfam
- Page for Bacterial RNase P class A at Rfam
- Page for Bacterial RNase P class B at Rfam
- RNase+P at the US National Library of Medicine Medical Subject Headings (MeSH)
- EC 3.1.26.5
 |