Chemistry:Lapaquistat
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
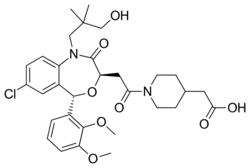
| Formula | C31H39ClN2O8 |
| Molar mass | 603.11 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Lapaquistat (TAK-475) is a cholesterol-lowering drug candidate that was abandoned before being marketed.
Unlike statins, which inhibit HMG-CoA reductase, lapaquistat metabolites inhibit squalene synthase, which is further downstream in the synthesis of cholesterol. It is hoped that side effects can be reduced by not disturbing the mevalonate pathway, which is important for other biochemical molecules besides cholesterol. However, there is increasing evidence that statins (which inhibit the mevalonate pathway) may be clinically useful because they affect these other molecules (including protein prenylation).[1]
On March 28, 2008, Takeda halted further development of lapaquistat.[2] While effective at lowering low-density lipoprotein cholesterol in a dose-dependent manner, development of the drug was ceased due to observations in clinical trials that it might cause liver damage in the high dose trial groups.[3] Data from knockout mouse studies suggests that accumulation of high levels of the metabolic substrate of squalene synthase and derivatives thereof account for the liver toxicity of squalene synthase inhibitors,[4] and efforts to mitigate this substrate accumulation would likely be necessary for clinical success of a squalene synthase inhibitor [5]
References
- ↑ "Statin therapy and autoimmune disease: from protein prenylation to immunomodulation". Nature Reviews. Immunology 6 (5): 358–370. May 2006. doi:10.1038/nri1839. PMID 16639429.
- ↑ "Discontinuation of Development of TAK-475, A Compound for Treatment of Hypercholesterolemia". Takeda Pharmaceutical Company Limited press release. http://www.takeda.com/press/article_29153.html.
- ↑ "Lapaquistat acetate: development of a squalene synthase inhibitor for the treatment of hypercholesterolemia". Circulation 123 (18): 1974–1985. May 2011. doi:10.1161/CIRCULATIONAHA.110.975284. PMID 21518985.
- ↑ "Plasma cholesterol-lowering and transient liver dysfunction in mice lacking squalene synthase in the liver". Journal of Lipid Research 56 (5): 998–1005. May 2015. doi:10.1194/jlr.M057406. PMID 25755092.
- ↑ "A novel bisphosphonate inhibitor of squalene synthase combined with a statin or a nitrogenous bisphosphonate in vitro". Journal of Lipid Research 52 (11): 1957–1964. November 2011. doi:10.1194/jlr.M016089. PMID 21903868.
Further reading
- "Squalene synthase inhibition: a novel target for the management of dyslipidemia". Current Atherosclerosis Reports 9 (1): 78–80. January 2007. doi:10.1007/BF02693932. PMID 17169251.
 |
