Chemistry:Xenon fluoride nitrate
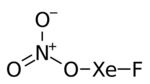
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| |
| |
| Properties | |
| FNO3Xe | |
| Molar mass | 212.295 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Xenon fluoride nitrate, also known as fluoroxenonium nitrate, is the chemical compound with formula FXeONO2.[1][2]
Synthesis
This compound is formed via the reaction:[3]
- [FXeOXeFXeF][AsF6] + 2NO2F → FXeONO2 + NO2AsF6.
Purification of FXeONO2 can then take place by dissolving in SO2ClF, which leaves the nitronium arsenic hexafluoride behind as a solid.[3]
An alternate low yield method to make FXeONO2 is to dissolve xenon difluoride in liquid dinitrogen tetroxide at 0 °C.[3]
- XeF2 + NO+ + NO3− → FXeONO2 + NOF
This method is inefficient as not much nitrate ion exists in the liquid and the xenon fluoride nitrate decomposes.[3]
Another method claimed to make this substance is:[3]
- XeF2 + HNO3 → FXeONO2 + HF
Properties
FXeONO2 is a white crystalline material.[3] The space group of the crystals is P21/c, which is monoclinic. The unit cell contains four molecules with a total volume of 386.6 Å3. The unit cell dimensions are a = 4.6663 Å, b = 8.799 Å c = 9.415 Å, with non-perpendicular angle β = 90.325°.[3] With a molecular weight of 212.3, the crystal has density 3.648. (These measurements at −173 °C.)[3]
The bond lengths in the molecule are 1.992 Å for Xe–F, 2.126 Å for Xe–O, 1.36 Å for O–NO2, 1.199 for N–Ocis and 1.224 Å for N–Otrans.[3] The bond angles are 177.6° for F–Xe–O, 114.7° for Xe-O-N, 114.5° for (Xe)O–N–Ocis, 118.4° for (Xe)O–N–Otrans and 127.1° for Ocis–N–Otrans.[3] The bond lengths and angles on the xenon atom are similar to that in FXeOSO2F and FXeOTeF5, indicating a polar oxygen bond. The Xe–O–N angle is larger than those in halogen nitrates, which indicates a lower bond density for the Xe–O bond. The N–Ocis bond length is longer than the N–Otrans bond length, opposite to other halogen nitrates.[3]
FXeONO2 is not particularly stable and slowly breaks down at −78 °C, yielding XeF2·N2O4. This happens on a timescale of several days.[3] At 0 °C, FXeONO2 has a half life of seven hours, decomposing to XeF2.[3]
References
- ↑ Moran, Matthew D.; David S. Brock; Hélène P. A. Mercier; Gary J. Schrobilgen (2010). "Xe3OF3+, a Precursor to a Noble-Gas Nitrate; Syntheses and Structural Characterizations of FXeONO2, XeF2·HNO3, and XeF2·N2O4". Journal of the American Chemical Society 132 (39): 13823–13839. doi:10.1021/ja105618w. ISSN 0002-7863. PMID 20843046. Bibcode: 2010JAChS.13213823M.
- ↑ Atta-ur-Rahman (2006-01-01). Advances in Organic Synthesis: Modern Organofluorine Chemistry-Synthetic Aspects. Bentham Science Publishers. p. 78. ISBN 9781608051984. https://books.google.com/books?id=vkcLOFEo9xIC&pg=PA78. Retrieved 5 October 2014.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 Moran, Matthew D. (2007). Synthesis and Structural Characterization of new Xenon(II) Compounds and the Use of a Xenon(II) Cation as an Oxidant for the Preparation of Halogenated Hydrocarbons. McMaster University. pp. 42, 99–145. https://macsphere.mcmaster.ca/bitstream/11375/14167/1/fulltext.pdf. Retrieved 4 Oct 2014.
| HNO3 | He | ||||||||||||||||
| LiNO3 | Be(NO3)2 | B(NO3)−4 | C | NO−3, NH4NO3 |
O | FNO3 | Ne | ||||||||||
| NaNO3 | Mg(NO3)2 | Al(NO3)3 | Si | P | S | ClONO2 | Ar | ||||||||||
| KNO3 | Ca(NO3)2 | Sc(NO3)3 | Ti(NO3)4 | VO(NO3)3 | Cr(NO3)3 | Mn(NO3)2 | Fe(NO3)3, Fe(NO3)2 |
Co(NO3)2, Co(NO3)3 |
Ni(NO3)2 | Cu(NO3)2 | Zn(NO3)2 | Ga(NO3)3 | Ge | As | Se | Br | Kr |
| RbNO3 | Sr(NO3)2 | Y(NO3)3 | Zr(NO3)4 | Nb | Mo | Tc | Ru | Rh | Pd(NO3)2 | AgNO3 | Cd(NO3)2 | In | Sn | Sb(NO3)3 | Te | I | Xe(NO3)2 |
| CsNO3 | Ba(NO3)2 | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg2(NO3)2, Hg(NO3)2 |
Tl(NO3)3, TlNO3 |
Pb(NO3)2 | Bi(NO3)3 BiO(NO3) |
Po | At | Rn | |
| FrNO3 | Ra(NO3)2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La(NO3)3 | Ce(NO3)3, Ce(NO3)4 |
Pr | Nd(NO3)3 | Pm | Sm | Eu(NO3)3 | Gd(NO3)3 | Tb(NO3)3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac(NO3)3 | Th(NO3)4 | Pa | UO2(NO3)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||
 |
