Chemistry:Abacavir
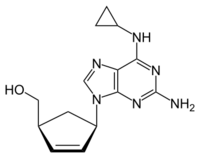 | |
 Chemical structure of abacavir | |
| Clinical data | |
|---|---|
| Pronunciation | /əˈbækəvɪər/ ( |
| Trade names | Ziagen |
| Other names | Abacavir sulfate (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699012 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 83% |
| Metabolism | Liver |
| Elimination half-life | 1.54 ± 0.63 h |
| Excretion | Kidney (1.2% abacavir, 30% 5'-carboxylic acid metabolite, 36% 5'-glucuronide metabolite, 15% unidentified minor metabolites). Fecal (16%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| Chemical and physical data | |
| Formula | C14H18N6O |
| Molar mass | 286.339 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 165 °C (329 °F) |
| |
| |
| (verify) | |
Abacavir, sold under the brand name Ziagen among others, is a medication used to treat HIV/AIDS.[2][3][4][5] Similar to other nucleoside analog reverse-transcriptase inhibitors (NRTIs), abacavir is used together with other HIV medications, and is not recommended by itself.[6] It is taken by mouth as a tablet or solution and may be used in children over the age of three months.[4][7]
Abacavir is generally well tolerated.[7] Common side effects include vomiting, insomnia (trouble sleeping), fever, and feeling tired.[4] Other common side effects include loss of appetite, headache, nausea (feeling sick), diarrhea, rash, and lethargy (lack of energy).[3] More severe side effects include hypersensitivity, liver damage, and lactic acidosis.[4] Genetic testing can indicate whether a person is at higher risk of developing hypersensitivity.[4] Symptoms of hypersensitivity include rash, vomiting, and shortness of breath.[7] Abacavir is in the NRTI class of medications, which work by blocking reverse transcriptase, an enzyme needed for HIV virus replication.[8] Within the NRTI class, abacavir is a carbocyclic nucleoside.[4]
Abacavir was patented in 1988, and approved for use in the United States in 1998.[9][10] It is on the World Health Organization's List of Essential Medicines.[11] It is available as a generic medication.[4] Abacavir is used together with other HIV medications, such as abacavir/lamivudine/zidovudine, abacavir/dolutegravir/lamivudine, and abacavir/lamivudine.[7][8] The combination abacavir/lamivudine is an essential medicine.[11]
Medical uses
Abacavir, in combination with other antiretroviral agents, is indicated for the treatment of HIV-1 infection.[2][3] Abacavir should be used in combination with other antiretroviral agents.[2][3]
Contraindications
Abacavir is contraindicated for people who have the HLA‑B*5701 allele or who have moderate or severe liver disease (hepatic impairment).[2][3]
Side effects
Common adverse reactions include nausea, headache, fatigue, vomiting, diarrhea, Anorexia (symptom) (loss of appetite), and insomnia (trouble sleeping). Rare but serious side effects include hypersensitivity reaction such as rash, elevated AST and ALT, depression, anxiety, fever/chills, URI, lactic acidosis, hypertriglyceridemia, and lipodystrophy.[12]
Hypersensitivity syndrome
Hypersensitivity to abacavir is strongly associated with a specific allele at the human leukocyte antigen B locus namely HLA-B*5701.[13][14][15] The mechanism for this hypersensitivity reaction is due to abacavir binding in the antigen-binding cleft of HLA-B*57:01, allowing alternative peptides to bind, which appear as "non-self" when presented to T cells.[16] There is an association between the prevalence of HLA-B*5701 and ancestry. The prevalence of the allele is estimated to be 3.4 to 5.8 percent on average in populations of European ancestry, 17.6 percent in Indian Americans, 3.0 percent in Hispanic Americans, and 1.2 percent in Chinese Americans.[17][18] There is significant variability in the prevalence of HLA-B*5701 among African populations. In African Americans, the prevalence is estimated to be 1.0 percent on average, 0 percent in the Yoruba from Nigeria, 3.3 percent in the Luhya from Kenya, and 13.6 percent in the Masai from Kenya, although the average values are derived from highly variable frequencies within sample groups.[19]
Common symptoms of abacavir hypersensitivity syndrome include fever, malaise, nausea, and diarrhea. Some patients may also develop a skin rash.[20] Symptoms of AHS typically manifest within six weeks of treatment using abacavir, although they may be confused with symptoms of HIV, immune reconstitution syndrome, hypersensitivity syndromes associated with other drugs, or infection.[21] The U.S. Food and Drug Administration (FDA) released an alert concerning abacavir and abacavir-containing medications on 24 July 2008,[22] and the FDA-approved drug label for abacavir recommends pre-therapy screening for the HLA-B*5701 allele and the use of alternative therapy in subjects with this allele.[2] Additionally, both the Clinical Pharmacogenetics Implementation Consortium and the Dutch Pharmacogenetics Working Group recommend use of an alternative therapy in individuals with the HLA-B*5701 allele.[23][24]
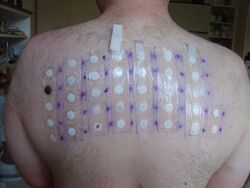
Skin-patch testing may also be used to determine whether an individual will experience a hypersensitivity reaction to abacavir, although some patients susceptible to developing AHS may not react to the patch test.[25]
The development of suspected hypersensitivity reactions to abacavir requires immediate and permanent discontinuation of abacavir therapy in all patients, including patients who do not possess the HLA-B*5701 allele. On 1 March 2011, the FDA informed the public about an ongoing safety review of abacavir and a possible increased risk of heart attack associated with the drug.[26] A meta-analysis of 26 studies conducted by the FDA, however, did not find any association between abacavir use and heart attack[27][28]
Immunopathogenesis
The mechanism underlying abacavir hypersensitivity syndrome is related to the change in the HLA-B*5701 protein product. Abacavir binds with high specificity to the HLA-B*5701 protein, changing the shape and chemistry of the antigen-binding cleft. This results in a change in immunological tolerance and the subsequent activation of abacavir-specific cytotoxic T cells, which produce a systemic reaction known as abacavir hypersensitivity syndrome.[16]
Interaction
Abacavir, and in general NRTIs, do not undergo hepatic metabolism and therefore have very limited (to none) interaction with the CYP enzymes and drugs that effect these enzymes. That being said there are still few interactions that can affect the absorption or the availability of abacavir. Below are few of the common established drug and food interaction that can take place during abacavir co-administration:
- Protease inhibitors such as tipranavir or ritonovir may decrease the serum concentration of abacavir through induction of glucuronidation. Abacavir is metabolized by both alcohol dehydrogenase and glucuronidation.[29][30]
- Ethanol may result in increased levels of abacavir through the inhibition of alcohol dehydrogenase. Abacavir is metabolized by both alcohol dehydrogenase and glucuronidation.[29][31]
- Methadone may diminish the therapeutic effect of Abacavir. Abacavir may decrease the serum concentration of Methadone.[32][33]
- Orlistat may decrease the serum concentration of antiretroviral drugs. The mechanism of this interaction is not fully established but it is suspected that it is due to the decreased absorption of abacavir by orlistat.[34]
- Cabozantinib: Drugs from the MRP2 inhibitor (Multidrug resistance-associated protein 2 inhibitors) family such as abacavir could increase the serum concentration of Cabozantinib.[35]
Mechanism of action
Abacavir is a nucleoside reverse transcriptase inhibitor that inhibits viral replication. It is a guanosine analogue that is phosphorylated to carbovir triphosphate (CBV-TP). CBV-TP competes with the viral molecules and is incorporated into the viral DNA. Once CBV-TP is integrated into the viral DNA, transcription and HIV reverse transcriptase is inhibited.[36]
Pharmacokinetics
Abacavir is given orally and is rapidly absorbed with a high bioavailability of 83%.[37] Solution and tablet have comparable concentrations and bioavailability. Abacavir can be taken with or without food.[38]
Abacavir can cross the blood–brain barrier. Abacavir is metabolized primarily through the enzymes alcohol dehydrogenase and glucuronyl transferase to an inactive carboxylate and glucuronide metabolites. It has a half-life of approximately 1.5-2.0 hours. If a person has liver failure, abacavir's half life is increased by 58%.[39]
Abacavir is eliminated via excretion in the urine (83%) and feces (16%).[40] It is unclear whether abacavir can be removed by hemodialysis or peritoneal dialysis.[36]
History
Robert Vince and Susan Daluge along with Mei Hua, a visiting scientist from China, developed the medication in the '80s.[41][42][43]
Abacavir was approved by the U.S. Food and Drug Administration (FDA) on 18 December 1998, and is thus the fifteenth approved antiretroviral drug in the United States.[44] Its patent expired in the United States on 26 December 2009.[citation needed]
Synthesis

References
- ↑ "TGA eBS - Product and Consumer Medicine Information Licence". http://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2011-PI-01963-3.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 "Ziagen- abacavir sulfate tablet, film coated label". DailyMed. 30 September 2015. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=ca73b519-015a-436d-aa3c-af53492825a1.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 "Ziagen EPAR". 17 September 2018. https://www.ema.europa.eu/en/medicines/human/EPAR/ziagen. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 "Abacavir Sulfate". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/abacavir-sulfate.html.
- ↑ "Drug Name Abbreviations Adult and Adolescent ARV Guidelines". https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv-guidelines/292/drug-name-abbreviations.
- ↑ "What Not to Use Adult and Adolescent ARV Guidelines". https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv-guidelines/12/what-not-to-use.
- ↑ 7.0 7.1 7.2 7.3 "A review of the pharmacokinetics of abacavir". Clinical Pharmacokinetics 47 (6): 351–371. 2008. doi:10.2165/00003088-200847060-00001. PMID 18479171.
- ↑ 8.0 8.1 "Nucleoside reverse transcriptase inhibitors (NRTIs or 'nukes') - HIV/AIDS". http://www.hiv.va.gov/patient/treat/NRTIs.asp.
- ↑ Analogue-based Drug Discovery. John Wiley & Sons. 2006. p. 505. ISBN 9783527607495. https://books.google.com/books?id=FjKfqkaKkAAC&pg=PA505.
- ↑ HIV/AIDS Treatment Drugs. Infobase Publishing. 2008. p. 56. ISBN 9781438102078. https://books.google.com/books?id=RFNVvStUJlkC&pg=PA56.
- ↑ 11.0 11.1 World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Abacavir Adverse Reactions". https://online.epocrates.com/noFrame/showPage.do?method=drugs&MonographId=2043&ActiveSectionId=5.
- ↑ "HLA-B*5701 screening for hypersensitivity to abacavir". The New England Journal of Medicine 358 (6): 568–579. February 2008. doi:10.1056/nejmoa0706135. PMID 18256392. http://researchrepository.murdoch.edu.au/id/eprint/8930/. Retrieved 9 July 2019.
- ↑ "Prospective genetic screening decreases the incidence of abacavir hypersensitivity reactions in the Western Australian HIV cohort study". Clinical Infectious Diseases 43 (1): 99–102. July 2006. doi:10.1086/504874. PMID 16758424.
- ↑ "Abacavir Therapy and HLA-B*57:01 Genotype". Medical Genetics Summaries. National Center for Biotechnology Information (NCBI). 2015. Bookshelf ID: NBK315783. https://www.ncbi.nlm.nih.gov/books/NBK315783/. Retrieved 2019-01-14.
- ↑ 16.0 16.1 "Immune self-reactivity triggered by drug-modified HLA-peptide repertoire". Nature 486 (7404): 554–558. June 2012. doi:10.1038/nature11147. PMID 22722860. Bibcode: 2012Natur.486..554I.
- ↑ "Genetic variations in HLA-B region and hypersensitivity reactions to abacavir". Lancet 359 (9312): 1121–1122. March 2002. doi:10.1016/s0140-6736(02)08158-8. PMID 11943262.
- ↑ "Association between presence of HLA-B*5701, HLA-DR7, and HLA-DQ3 and hypersensitivity to HIV-1 reverse-transcriptase inhibitor abacavir". Lancet 359 (9308): 727–732. March 2002. doi:10.1016/s0140-6736(02)07873-x. PMID 11888582.
- ↑ "Ancestry and disease in the age of genomic medicine". The New England Journal of Medicine 363 (16): 1551–1558. October 2010. doi:10.1056/nejmra0911564. PMID 20942671.
- ↑ "Successful translation of pharmacogenetics into the clinic: the abacavir example". Molecular Diagnosis & Therapy 13 (1): 1–9. 2009. doi:10.1007/bf03256308. PMID 19351209.
- ↑ "Drug hypersensitivity in HIV". Current Opinion in Allergy and Clinical Immunology 7 (4): 324–330. August 2007. doi:10.1097/aci.0b013e32825ea68a. PMID 17620824.
- ↑ "Information for Healthcare Professionals: Abacavir (marketed as Ziagen) and Abacavir-Containing Medications". Food and Drug Administration (FDA). 2008-07-24. https://www.fda.gov/drugs/drugsafety/postmarketdrugsafetyinformationforpatientsandproviders/ucm123927.htm.
- ↑ "Pharmacogenetics: from bench to byte--an update of guidelines". Clinical Pharmacology and Therapeutics 89 (5): 662–673. May 2011. doi:10.1038/clpt.2011.34. PMID 21412232.
- ↑ "Clinical Pharmacogenetics Implementation Consortium Guidelines for HLA-B Genotype and Abacavir Dosing: 2014 update". Clinical Pharmacology and Therapeutics 95 (5): 499–500. May 2014. doi:10.1038/clpt.2014.38. PMID 24561393.
- ↑ "A review of drug patch testing and implications for HIV clinicians". AIDS 22 (9): 999–1007. May 2008. doi:10.1097/qad.0b013e3282f7cb60. PMID 18520343.
- ↑ "FDA Drug Safety Communication: Safety Review update of Abacavir and possible increased risk of heart attack". Food and Drug Administration (FDA). 1 March 2011. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-safety-review-update-abacavir-and-possible-increased-risk-heart-attack.
- ↑ "FDA Alert: Abacavir - Ongoing Safety Review: Possible Increased Risk of Heart Attack". Drugs.com. https://www.drugs.com/fda/abacavir-ongoing-safety-review-possible-increased-risk-heart-attack-12914.html.
- ↑ "No association of abacavir use with myocardial infarction: findings of an FDA meta-analysis". Journal of Acquired Immune Deficiency Syndromes 61 (4): 441–447. December 2012. doi:10.1097/QAI.0b013e31826f993c. PMID 22932321.
- ↑ 29.0 29.1 Prescribing information. Ziagen (abacavir). Research Triangle Park, NC: GlaxoSmithKline, July 2002
- ↑ "Mechanisms of pharmacokinetic and pharmacodynamic drug interactions associated with ritonavir-enhanced tipranavir". Pharmacotherapy 27 (6): 888–909. June 2007. doi:10.1592/phco.27.6.888. PMID 17542771.
- ↑ "Pharmacokinetic interaction of abacavir (1592U89) and ethanol in human immunodeficiency virus-infected adults". Antimicrobial Agents and Chemotherapy 44 (6): 1686–1690. June 2000. doi:10.1128/aac.44.6.1686-1690.2000. PMID 10817729.
- ↑ "Effectiveness and safety of abacavir, lamivudine, and zidovudine in antiretroviral therapy-naive HIV-infected patients: results from a large multicenter observational cohort". Journal of Acquired Immune Deficiency Syndromes 41 (2): 154–159. February 2006. doi:10.1097/01.qai.0000194231.08207.8a. PMID 16394846.
- ↑ Dolophine(methadone) [prescribing information]. Columbus, OH: Roxane Laboratories, Inc.; March 2015.
- ↑ "Orlistat: weight lost at cost of HIV rebound". The Journal of Antimicrobial Chemotherapy 71 (6): 1739–1741. June 2016. doi:10.1093/jac/dkw033. PMID 26945709.
- ↑ Cometriq (cabozantinib) [prescribing information]. South San Francisco, CA: Exelixis, Inc.; May 2016.
- ↑ 36.0 36.1 Product Information: ZIAGEN(R) oral tablets, oral solution, abacavir sulfate oral tablets, oral solution. ViiV Healthcare (per Manufacturer), Research Triangle Park, NC, 2015.
- ↑ "Abacavir: absolute bioavailability, bioequivalence of three oral formulations, and effect of food". Pharmacotherapy 19 (8): 932–942. August 1999. doi:10.1592/phco.19.11.932.31568. PMID 10453964.
- ↑ "Food requirements for anti-HIV medications". aidsmap.com. May 2019. https://www.aidsmap.com/about-hiv/food-requirements-anti-hiv-medications.
- ↑ "Abacavir pharmacokinetics in hepatic dysfunction". Clinical Infectious Diseases 40 (6): 909–910. March 2005. doi:10.1086/429247. PMID 15736035.
- ↑ "A review of the pharmacokinetics of abacavir". Clinical Pharmacokinetics 47 (6): 351–71. 2008. doi:10.2165/00003088-200847060-00001. PMID 18479171.
- ↑ "Dr. Robert Vince - 2010 Inductee". Minnesota Inventors Hall of Fame. http://www.minnesotainventors.org/inductees/robert-vince.html.
- ↑ "Robert Vince, PhD (faculty listing)". University of Minnesota. http://drugdesign.umn.edu/bio/cdd-faculty-staff/robert-vince.
- ↑ "1592U89, a novel carbocyclic nucleoside analog with potent, selective anti-human immunodeficiency virus activity". Antimicrobial Agents and Chemotherapy 41 (5): 1082–1093. May 1997. doi:10.1128/AAC.41.5.1082. PMID 9145874.
- ↑ Mary Annette Banach. "How HIV Clinicians Acquire Representational Fluency: A Case Study of the HIV Resistance Preceptorship ", University of California, Berkeley, 2003.
- ↑ "An Efficient Asymmetric Approach to Carbocyclic Nucleosides: Asymmetric Synthesis of 1592U89, a Potent Inhibitor of HIV Reverse Transcriptase". The Journal of Organic Chemistry 61 (13): 4192–4193. June 1996. doi:10.1021/jo960708p. PMID 11667311.
Further reading
- "Abacavir Therapy and HLA-B*57:01 Genotype". Medical Genetics Summaries. National Center for Biotechnology Information (NCBI). April 2018. https://www.ncbi.nlm.nih.gov/books/NBK315783/. Retrieved 6 February 2020.
External links
- "Abacavir". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/rn/136470-78-5.
- "Abacavir sulfate". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/abacavir%20sulfate.
- Abacavir pathway on PharmGKB
 |