Biology:Cholesteryl ester transfer protein
 Generic protein structure example |
Cholesteryl ester transfer protein (CETP), also called plasma lipid transfer protein, is a plasma protein that facilitates the transport of cholesteryl esters and triglycerides between the lipoproteins. It collects triglycerides from very-low-density lipoproteins (VLDL) or chylomicrons and exchanges them for cholesteryl esters from high-density lipoproteins (HDL), and vice versa. Most of the time, however, CETP does a heteroexchange, trading a triglyceride for a cholesteryl ester or a cholesteryl ester for a triglyceride.
Genetics
The CETP gene is located on chromosome 16 (16q21).
Protein fold
The crystal structure of CETP is that of dimer of two TUbular LIPid (TULIP) binding domains.[1][2] Each domain consists of a core of 6 elements: 4 beta-sheets forming an extended superhelix; 2 flanking elements that tend to include some alpha helix. The sheets wrap around the helices to produce a cylinder 6 x 2.5 x 2.5 nm. CETP contains two of these domains that interact head-to-head via an interface made of 6 beta-sheets, 3 from each protomer. The same fold is shared by Bacterial Permeability Inducing proteins (examples: BPIFP1 BPIFP2 BPIFA3 and BPIFB4), phospholipid transfer protein (PLTP), and long-Palate Lung, and Nasal Epithelium protein (L-PLUNC). The fold is similar to intracellular SMP domains,[3] and originated in bacteria.[4][5][6] The crystal structure of CETP has been obtained with bound CETP inhibitors.[7] However, this has not resolved the doubt over whether CETP function as a lipid tube or shuttle.[8]
Role in disease
Rare mutations leading to reduced function of CETP have been linked to accelerated atherosclerosis.[9] In contrast, a polymorphism (I405V) of the CETP gene leading to lower serum levels has also been linked to exceptional longevity[10] and to metabolic response to nutritional intervention.[11] However, this mutation also increases the prevalence of coronary heart disease in patients with hypertriglyceridemia.[12] The D442G mutation, which lowers CETP levels and increases HDL levels also increases coronary heart disease.[9]
Elaidic acid, a major component of trans fat, increases CETP activity.[13]
Pharmacology
As HDL can alleviate atherosclerosis and other cardiovascular diseases, and certain disease states such as the metabolic syndrome feature low HDL, pharmacological inhibition of CETP is being studied as a method of improving HDL levels.[14] To be specific, in a 2004 study, the small molecular agent torcetrapib was shown to increase HDL levels, alone and with a statin, and lower LDL when co-administered with a statin.[15] Studies into cardiovascular endpoints, however, were largely disappointing. While they confirmed the change in lipid levels, most reported an increase in blood pressure, no change in atherosclerosis,[16][17] and, in a trial of a combination of torcetrapib and atorvastatin, an increase in cardiovascular events and mortality.[18]
A compound related to torcetrapib, Dalcetrapib (investigative name JTT-705/R1658), was also studied, but trials have ceased.[19] It increases HDL levels by 30%, as compared to 60% by torcetrapib.[20] Two CETP inhibitors were previously under development. One was Merck's MK-0859 anacetrapib, which in initial studies did not increase blood pressure.[21] In 2017, its development was abandoned by Merck.[22] The other was Eli Lilly's evacetrapib, which failed in Phase 3 trials.
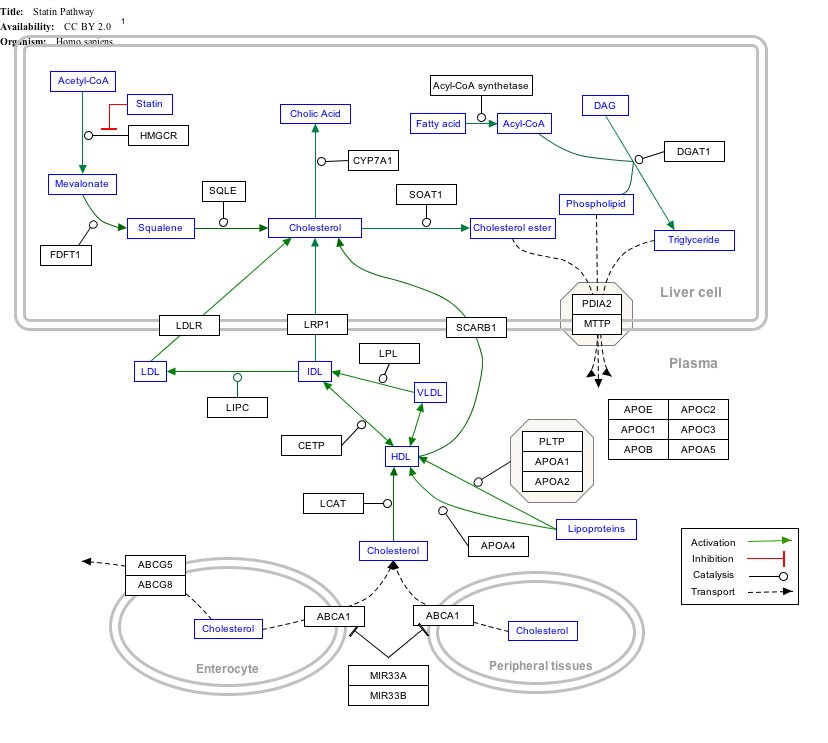
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ↑ The interactive pathway map can be edited at WikiPathways: "Statin_Pathway_WP430". http://www.wikipathways.org/index.php/Pathway:WP430.
References
- ↑ "Crystal structure of cholesteryl ester transfer protein reveals a long tunnel and four bound lipid molecules". Nature Structural & Molecular Biology 14 (2): 106–13. February 2007. doi:10.1038/nsmb1197. PMID 17237796.
- ↑ "The TULIP superfamily of eukaryotic lipid-binding proteins as a mediator of lipid sensing and transport". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 1861 (8 Pt B): 913–923. August 2016. doi:10.1016/j.bbalip.2016.01.016. PMID 26825693.
- ↑ "SMP-domain proteins at membrane contact sites: Structure and function". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 1861 (8 Pt B): 924–927. August 2016. doi:10.1016/j.bbalip.2015.12.003. PMID 26686281.
- ↑ "Tubular lipid binding proteins (TULIPs) growing everywhere". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 1864 (9): 1439–1449. September 2017. doi:10.1016/j.bbamcr.2017.05.019. PMID 28554774.
- ↑ "The hypothetical protein P47 of Clostridium botulinum E1 strain Beluga has a structural topology similar to bactericidal/permeability-increasing protein". Toxicon 147: 19–26. June 2018. doi:10.1016/j.toxicon.2017.10.012. PMID 29042313.
- ↑ "Crystal structures of OrfX2 and P47 from a Botulinum neurotoxin OrfX-type gene cluster". FEBS Letters 591 (22): 3781–3792. November 2017. doi:10.1002/1873-3468.12889. PMID 29067689.
- ↑ "Crystal structures of cholesteryl ester transfer protein in complex with inhibitors". The Journal of Biological Chemistry 287 (44): 37321–9. October 2012. doi:10.1074/jbc.M112.380063. PMID 22961980.
- ↑ "Cholesteryl ester transfer between lipoproteins does not require a ternary tunnel complex with CETP". Journal of Structural Biology 194 (2): 191–8. May 2016. doi:10.1016/j.jsb.2016.02.016. PMID 26876146.
- ↑ 9.0 9.1 "Increased coronary heart disease in Japanese-American men with mutation in the cholesteryl ester transfer protein gene despite increased HDL levels". The Journal of Clinical Investigation 97 (12): 2917–23. June 1996. doi:10.1172/JCI118751. PMID 8675707.
- ↑ "Unique lipoprotein phenotype and genotype associated with exceptional longevity". JAMA 290 (15): 2030–40. October 2003. doi:10.1001/jama.290.15.2030. PMID 14559957.
- ↑ "Cholesteryl ester transfer protein I405V polymorphism influences apolipoprotein A-I response to a change in dietary fatty acid composition". Hormone and Metabolic Research 41 (7): 554–8. July 2009. doi:10.1055/s-0029-1192034. PMID 19242900.
- ↑ "Relationship of HDL and coronary heart disease to a common amino acid polymorphism in the cholesteryl ester transfer protein in men with and without hypertriglyceridemia". Journal of Lipid Research 39 (5): 1071–8. May 1998. doi:10.1016/S0022-2275(20)33876-1. PMID 9610775.
- ↑ "Plasma cholesteryl ester transfer protein activity is increased when trans-elaidic acid is substituted for cis-oleic acid in the diet". Atherosclerosis 106 (1): 99–107. March 1994. doi:10.1016/0021-9150(94)90086-8. PMID 8018112.
- ↑ "Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis". Arteriosclerosis, Thrombosis, and Vascular Biology 23 (2): 160–7. February 2003. doi:10.1161/01.ATV.0000054658.91146.64. PMID 12588754.
- ↑ "Effects of an inhibitor of cholesteryl ester transfer protein on HDL cholesterol". The New England Journal of Medicine 350 (15): 1505–15. April 2004. doi:10.1056/NEJMoa031766. PMID 15071125.
- ↑ "Effect of torcetrapib on the progression of coronary atherosclerosis". The New England Journal of Medicine 356 (13): 1304–16. March 2007. doi:10.1056/NEJMoa070635. PMID 17387129.
- ↑ "Effect of torcetrapib on carotid atherosclerosis in familial hypercholesterolemia". The New England Journal of Medicine 356 (16): 1620–30. April 2007. doi:10.1056/NEJMoa071359. PMID 17387131.
- ↑ "Pfizer Stops All Torcetrapib Clinical Trials in Interest of Patient Safety" (Press release). U.S. Food and Drug Administration. 2006-12-03. Archived from the original on 5 December 2006.
- ↑ "The role of CETP inhibition in dyslipidemia". Current Atherosclerosis Reports 9 (2): 125–33. August 2007. doi:10.1007/s11883-007-0008-5. PMID 17877921.
- ↑ "Efficacy and safety of a novel cholesteryl ester transfer protein inhibitor, JTT-705, in humans: a randomized phase II dose-response study". Circulation 105 (18): 2159–65. May 2002. doi:10.1161/01.CIR.0000015857.31889.7B. PMID 11994249.
- ↑ "Merck announces its investigational CETP-Inhibitor, MK-0859, produced positive effects on lipids with no observed blood pressure changes". Reuters (Reuters, Inc.). 2007-10-04. https://www.reuters.com/article/inPlayBriefing/idUSIN20071004163052MRK20071004.
- ↑ "Merck says will not seek approval of cholesterol treatment". Reuters. 2017. https://www.reuters.com/article/us-merck-cholesterol/merck-says-will-not-seek-approval-of-cholesterol-treatment-idUSKBN1CG2W1.
Further reading
- "[Distribution of sphingosine 1-phosphate in plasma lipoproteins and its role in the regulation of the vascular cell functions]". Tanpakushitsu Kakusan Koso. Protein, Nucleic Acid, Enzyme 47 (4 Suppl): 480–7. March 2002. PMID 11915346.
- "Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis". Arteriosclerosis, Thrombosis, and Vascular Biology 23 (2): 160–7. February 2003. doi:10.1161/01.ATV.0000054658.91146.64. PMID 12588754.
- "Concerted actions of cholesteryl ester transfer protein and phospholipid transfer protein in type 2 diabetes: effects of apolipoproteins". Current Opinion in Lipidology 18 (3): 251–7. June 2007. doi:10.1097/MOL.0b013e3280e12685. PMID 17495597.
External links
- Cholesterol+ester+transfer+proteins at the US National Library of Medicine Medical Subject Headings (MeSH)
 |