Biology:Erythropoietin receptor
 Generic protein structure example |
The erythropoietin receptor (EpoR) is a protein that in humans is encoded by the EPOR gene.[1] EpoR is a 52kDa peptide with a single carbohydrate chain resulting in an approximately 56-57 kDa protein found on the surface of EPO responding cells. It is a member of the cytokine receptor family. EpoR pre-exists as dimers. These dimers were originally thought to be formed by extracellular domain interactions,[2] however, it is now assumed that it is formed by interactions of the transmembrane domain[3][4] and that the original structure of the extracellular interaction site was due to crystallisation conditions and does not depict the native conformation.[5] Binding of a 30 kDa ligand erythropoietin (Epo), changes the receptor's conformational change, resulting in the autophosphorylation of Jak2 kinases that are pre-associated with the receptor (i.e., EpoR does not possess intrinsic kinase activity and depends on Jak2 activity).[6][7] At present, the best-established function of EpoR is to promote proliferation and rescue of erythroid (red blood cell) progenitors from apoptosis.[1]
Function and mechanism of action
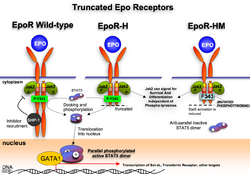
The cytoplasmic domains of the EpoR contain a number of phosphotyrosines that are phosphorylated by Jak2 and serve as docking sites for a variety of intracellular pathway activators and Stats (such as Stat5). In addition to activating Ras/AKT and ERK/MAP kinase, phosphatidylinositol 3-kinase/AKT pathway and STAT transcription factors, phosphotyrosines also serve as docking sites for phosphatases that negatively affect EpoR signaling in order to prevent overactivation that may lead to such disorders as erythrocytosis. In general, the defects in the erythropoietin receptor may produce erythroleukemia and familial erythrocytosis. Mutations in Jak2 kinases associated with EpoR can also lead to polycythemia vera.[8]
Erythroid survival
Primary role of EpoR is to promote proliferation of erythroid progenitor cells and rescue erythroid progenitors from cell death.[9] EpoR induced Jak2-Stat5 signaling, together with transcriptional factor GATA-1, induces the transcription of pro-survival protein Bcl-xL.[10] Additionally, EpoR has been implicated in suppressing expression of death receptors Fas, Trail and TNFa that negatively affect erythropoiesis.[11][12][13]
Based on current evidence, it is still unknown whether Epo/EpoR directly cause "proliferation and differentiation" of erythroid progenitors in vivo, although such direct effects have been described based on in vitro work.
Erythroid differentiation
It is thought that erythroid differentiation is primarily dependent on the presence and induction of erythroid transcriptional factors such as GATA-1, FOG-1 and EKLF, as well as the suppression of myeloid/lymphoid transcriptional factors such as PU.1.[14] Direct and significant effects of EpoR signaling specifically upon the induction of erythroid-specific genes such as beta-globin, have been mainly elusive. It is known that GATA-1 can induce EpoR expression.[15] In turn, EpoR's PI3-K/AKT signaling pathway augments GATA-1 activity.[16]
Erythroid cell cycle/proliferation
Induction of proliferation by the EpoR is likely cell type-dependent. It is known that EpoR can activate mitogenic signaling pathways and can lead to cell proliferation in erythroleukemic cell lines in vitro, various non-erythroid cells, and cancer cells. So far, there is no sufficient evidence that in vivo, EpoR signaling can induce erythroid progenitors to undergo cell division, or whether Epo levels can modulate the cell cycle.[9] EpoR signaling may still have a proliferation effect upon BFU-e progenitors, but these progenitors cannot be directly identified, isolated and studied. CFU-e progenitors enter the cell cycle at the time of GATA-1 induction and PU.1 suppression in a developmental manner rather than due to EpoR signaling.[17] Subsequent differentiation stages (proerythroblast to orthochromatic erythroblast) involve a decrease in cell size and eventual expulsion of the nucleus, and are likely dependent upon EpoR signaling only for their survival. In addition, some evidence on macrocytosis in hypoxic stress (when Epo can increase 1000-fold) suggests that mitosis is actually skipped in later erythroid stages, when EpoR expression is low/absent, in order to provide emergency reserve of red blood cells as soon as possible.[18][19] Such data, though sometimes circumstantial, argue that there is limited capacity to proliferate specifically in response to Epo (and not other factors). Together, these data suggest that EpoR in erythroid differentiation may function primarily as a survival factor, while its effect on the cell cycle (for example, rate of division and corresponding changes in the levels of cyclins and Cdk inhibitors) in vivo awaits further work. In other cell systems, however, EpoR may provide a specific proliferative signal.
Commitment of multipotent progenitors to the erythroid lineage
EpoR's role in lineage commitment is currently unclear. EpoR expression can extend as far back as the hematopoietic stem cell compartment.[20] It is unknown whether EpoR signaling plays a permissive (i.e. induces only survival) or an instructive (i.e. upregulates erythroid markers to lock progenitors to a predetermined differentiation path) role in early, multipotent progenitors in order to produce sufficient erythroblast numbers. Current publications in the field suggest that it is primarily permissive. The generation of BFU-e and CFU-e progenitors was shown to be normal in rodent embryos knocked out for either Epo or EpoR.[21] An argument against such lack of requirement is that in response to Epo or hypoxic stress, the number of early erythroid stages, the BFU-e and CFU-e, increases dramatically. However, it is unclear if it is an instructive signal or, again, a permissive signal. One additional point is that signaling pathways activated by the EpoR are common to many other receptors; replacing EpoR with prolactin receptor supports erythroid survival and differentiation in vitro.[22][23] Together, these data suggest that commitment to erythroid lineage likely does not happen due to EpoR's as-yet-unknown instructive function, but possibly due to its role in survival at the multipotent progenitor stages.
Animal studies on Epo Receptor mutations
Mice with truncated EpoR[24] are viable, which suggests Jak2 activity is sufficient to support basal erythropoiesis by activating the necessary pathways without phosphotyrosine docking sites being needed. EpoR-H form of EpoR truncation contains the first, and, what can be argued, the most important tyrosine 343 that serves as a docking site for the Stat5 molecule, but lacks the rest of the cytoplasmic tail. These mice exhibit elevated erythropoiesis consistent with the idea that phosphatase recruitment (and therefore the shutting down of signaling) is aberrant in these mice.
The EpoR-HM receptor also lacks the majority of the cytoplasmic domain, and contains the tyrosine 343 that was mutated to phenylalanine, making it unsuitable for efficient Stat5 docking and activation. These mice are anemic and show poor response to hypoxic stress, such as phenylhydrazine treatment or erythropoietin injection.[24]
EpoR knockout mice have defects in heart, brain and the vasculature. These defects may be due to blocks in RBC formation and thus insufficient oxygen delivery to developing tissues because mice engineered to express Epo receptors only in erythroid cells develop normally.
Clinical significance
Defects in the erythropoietin receptor may produce erythroleukemia and familial erythrocytosis.[1] Overproduction of red blood cells increases a chance of adverse cardiovascular event, such as thrombosis and stroke.
Rarely, seemingly beneficial mutations in the EpoR may arise, where increased red blood cell number allows for improved oxygen delivery in athletic endurance events with no apparent adverse effects upon the athlete's health (as for example in the Finnish athlete Eero Mäntyranta).[25]
Erythropoietin was reported to maintain endothelial cells and to promote tumor angiogenesis, hence the dysregulation of EpoR may affect the growth of certain tumors.[26][27] However this hypothesis is not universally accepted.
Interactions
Erythropoietin receptor has been shown to interact with:
- CRKL,[28][29]
- Erythropoietin,[30][31]
- Grb2,[32][33]
- Janus kinase 2,[34][35]
- LYN,[36]
- PIK3R1,[37][38]
- PTPN6,[39]
- SOCS2,[40]
- SOCS3,[41][42] and
- STAT5A.[43]
References
- ↑ 1.0 1.1 1.2 "Entrez Gene: EPOR erythropoietin receptor". https://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=2057.
- ↑ "Crystallographic evidence for preformed dimers of erythropoietin receptor before ligand activation". Science 283 (5404): 987–90. Feb 1999. doi:10.1126/science.283.5404.987. PMID 9974392. Bibcode: 1999Sci...283..987L.
- ↑ Ebie, Alexandra Z.; Fleming, Karen G. (February 2007). "Dimerization of the Erythropoietin Receptor Transmembrane Domain in Micelles" (in en). Journal of Molecular Biology 366 (2): 517–524. doi:10.1016/j.jmb.2006.11.035. PMID 17173930. https://linkinghub.elsevier.com/retrieve/pii/S0022283606015750.
- ↑ Li, Qingxin; Wong, Ying Lei; Huang, Qiwei; Kang, CongBao (November 2014). "Structural Insight into the Transmembrane Domain and the Juxtamembrane Region of the Erythropoietin Receptor in Micelles" (in en). Biophysical Journal 107 (10): 2325–2336. doi:10.1016/j.bpj.2014.10.013. PMID 25418301. Bibcode: 2014BpJ...107.2325L.
- ↑ Pang, Xiaodong; Zhou, Huan-Xiang (2012-03-08). "A Common Model for Cytokine Receptor Activation: Combined Scissor-Like Rotation and Self-Rotation of Receptor Dimer Induced by Class I Cytokine" (in en). PLOS Computational Biology 8 (3): e1002427. doi:10.1371/journal.pcbi.1002427. ISSN 1553-7358. PMID 22412367. Bibcode: 2012PLSCB...8E2427P.
- ↑ "Structure, function, and activation of the erythropoietin receptor". Blood 81 (9): 2223–36. May 1993. doi:10.1182/blood.V81.9.2223.2223. PMID 8481505.
- ↑ "The structure, organization, activation and plasticity of the erythropoietin receptor". Current Opinion in Structural Biology 9 (6): 696–704. Dec 1999. doi:10.1016/S0959-440X(99)00032-9. PMID 10607675.
- ↑ "A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera". Nature 434 (7037): 1144–8. Apr 2005. doi:10.1038/nature03546. PMID 15793561. Bibcode: 2005Natur.434.1144J.
- ↑ 9.0 9.1 "Erythropoietin retards DNA breakdown and prevents programmed death in erythroid progenitor cells". Science 248 (4953): 378–81. Apr 1990. doi:10.1126/science.2326648. PMID 2326648. Bibcode: 1990Sci...248..378K.
- ↑ "Fetal anemia and apoptosis of red cell progenitors in Stat5a-/-5b-/- mice: a direct role for Stat5 in Bcl-X(L) induction". Cell 98 (2): 181–91. Jul 1999. doi:10.1016/S0092-8674(00)81013-2. PMID 10428030.
- ↑ "Apoptotic role of Fas/Fas ligand system in the regulation of erythropoiesis". Blood 93 (3): 796–803. Feb 1999. doi:10.1182/blood.V93.3.796. PMID 9920828.
- ↑ "Suppression of Fas-FasL coexpression by erythropoietin mediates erythroblast expansion during the erythropoietic stress response in vivo". Blood 108 (1): 123–33. Jul 2006. doi:10.1182/blood-2005-11-4458. PMID 16527892.
- ↑ "Multiple members of the TNF superfamily contribute to IFN-gamma-mediated inhibition of erythropoiesis". Journal of Immunology 175 (3): 1464–72. Aug 2005. doi:10.4049/jimmunol.175.3.1464. PMID 16034083.
- ↑ "Transcriptional regulation of erythropoiesis: an affair involving multiple partners". Oncogene 21 (21): 3368–76. May 2002. doi:10.1038/sj.onc.1205326. PMID 12032775.
- ↑ "Activation of the erythropoietin receptor promoter by transcription factor GATA-1". Proceedings of the National Academy of Sciences of the United States of America 88 (23): 10638–41. Dec 1991. doi:10.1073/pnas.88.23.10638. PMID 1660143. Bibcode: 1991PNAS...8810638Z.
- ↑ "Erythropoietin stimulates phosphorylation and activation of GATA-1 via the PI3-kinase/AKT signaling pathway". Blood 107 (3): 907–15. Feb 2006. doi:10.1182/blood-2005-06-2516. PMID 16204311.
- ↑ "A key commitment step in erythropoiesis is synchronized with the cell cycle clock through mutual inhibition between PU.1 and S-phase progression". PLOS Biology 8 (9): e1000484. 2010. doi:10.1371/journal.pbio.1000484. PMID 20877475.
- ↑ "Macrocytosis Resulting from Early Denucleation of Erythroid Precursors". Blood 24 (5): 582–93. Nov 1964. doi:10.1182/blood.V24.5.582.582. PMID 14236733.
- ↑ "Synthesis of haemoglobin in relation to the maturation of erythroid cells". Nature 196 (4852): 347–50. Oct 1962. doi:10.1038/196347a0. PMID 14014098. Bibcode: 1962Natur.196..347B.
- ↑ "New evidence supporting megakaryocyte-erythrocyte potential of flk2/flt3+ multipotent hematopoietic progenitors". Cell 126 (2): 415–26. Jul 2006. doi:10.1016/j.cell.2006.06.037. PMID 16873070.
- ↑ "Generation of committed erythroid BFU-E and CFU-E progenitors does not require erythropoietin or the erythropoietin receptor". Cell 83 (1): 59–67. Oct 1995. doi:10.1016/0092-8674(95)90234-1. PMID 7553874.
- ↑ "The prolactin receptor rescues EpoR-/- erythroid progenitors and replaces EpoR in a synergistic interaction with c-kit". Blood 92 (5): 1491–6. Sep 1998. doi:10.1182/blood.V92.5.1491. PMID 9716574.
- ↑ "The prolactin receptor and severely truncated erythropoietin receptors support differentiation of erythroid progenitors". The Journal of Biological Chemistry 272 (22): 14009–12. May 1997. doi:10.1074/jbc.272.22.14009. PMID 9162017.
- ↑ 24.0 24.1 "The distal region and receptor tyrosines of the Epo receptor are non-essential for in vivo erythropoiesis". The EMBO Journal 20 (12): 3156–66. Jun 2001. doi:10.1093/emboj/20.12.3156. PMID 11406592.
- ↑ "Truncated erythropoietin receptor causes dominantly inherited benign human erythrocytosis". Proceedings of the National Academy of Sciences of the United States of America 90 (10): 4495–9. May 1993. doi:10.1073/pnas.90.10.4495. PMID 8506290. Bibcode: 1993PNAS...90.4495D.
- ↑ "The erythropoietin receptor and its expression in tumor cells and other tissues". The Oncologist 9 (Suppl 5): 18–30. 2004. doi:10.1634/theoncologist.9-90005-18. PMID 15591419.
- ↑ "The erythropoietin receptor in normal and cancer tissues". Critical Reviews in Oncology/Hematology 67 (1): 39–61. Jul 2008. doi:10.1016/j.critrevonc.2008.03.006. PMID 18434185.
- ↑ "Erythropoietin and IL-3 induce tyrosine phosphorylation of CrkL and its association with Shc, SHP-2, and Cbl in hematopoietic cells". Biochemical and Biophysical Research Communications 239 (2): 412–7. Oct 1997. doi:10.1006/bbrc.1997.7480. PMID 9344843.
- ↑ "CrkL is recruited through its SH2 domain to the erythropoietin receptor and plays a role in Lyn-mediated receptor signaling". The Journal of Biological Chemistry 276 (35): 33282–90. Aug 2001. doi:10.1074/jbc.M102924200. PMID 11443118.
- ↑ "Shared and unique determinants of the erythropoietin (EPO) receptor are important for binding EPO and EPO mimetic peptide". The Journal of Biological Chemistry 274 (20): 14163–9. May 1999. doi:10.1074/jbc.274.20.14163. PMID 10318834.
- ↑ "An antagonist peptide-EPO receptor complex suggests that receptor dimerization is not sufficient for activation". Nature Structural Biology 5 (11): 993–1004. Nov 1998. doi:10.1038/2965. PMID 9808045.
- ↑ "Involvement of SH2-containing phosphotyrosine phosphatase Syp in erythropoietin receptor signal transduction pathways". The Journal of Biological Chemistry 270 (10): 5631–5. Mar 1995. doi:10.1074/jbc.270.10.5631. PMID 7534299.
- ↑ "Grap is a novel SH3-SH2-SH3 adaptor protein that couples tyrosine kinases to the Ras pathway". The Journal of Biological Chemistry 271 (21): 12129–32. May 1996. doi:10.1074/jbc.271.21.12129. PMID 8647802.
- ↑ "The N-terminal domain of Janus kinase 2 is required for Golgi processing and cell surface expression of erythropoietin receptor". Molecular Cell 8 (6): 1327–38. Dec 2001. doi:10.1016/S1097-2765(01)00401-4. PMID 11779507.
- ↑ "JAK2 associates with the erythropoietin receptor and is tyrosine phosphorylated and activated following stimulation with erythropoietin". Cell 74 (2): 227–36. Jul 1993. doi:10.1016/0092-8674(93)90414-L. PMID 8343951.
- ↑ "Lyn physically associates with the erythropoietin receptor and may play a role in activation of the Stat5 pathway". Blood 91 (10): 3734–45. May 1998. doi:10.1182/blood.V91.10.3734. PMID 9573010.
- ↑ "Role of the vav proto-oncogene product (Vav) in erythropoietin-mediated cell proliferation and phosphatidylinositol 3-kinase activity". The Journal of Biological Chemistry 272 (22): 14334–40. May 1997. doi:10.1074/jbc.272.22.14334. PMID 9162069.
- ↑ "Phosphorylation of tyrosine 503 in the erythropoietin receptor (EpR) is essential for binding the P85 subunit of phosphatidylinositol (PI) 3-kinase and for EpR-associated PI 3-kinase activity". The Journal of Biological Chemistry 270 (40): 23402–8. Oct 1995. doi:10.1074/jbc.270.40.23402. PMID 7559499.
- ↑ "Specific recruitment of SH-PTP1 to the erythropoietin receptor causes inactivation of JAK2 and termination of proliferative signals". Cell 80 (5): 729–38. Mar 1995. doi:10.1016/0092-8674(95)90351-8. PMID 7889566.
- ↑ "Design and application of a cytokine-receptor-based interaction trap". Nature Cell Biology 3 (12): 1114–9. Dec 2001. doi:10.1038/ncb1201-1114. PMID 11781573.
- ↑ "CIS3/SOCS-3 suppresses erythropoietin (EPO) signaling by binding the EPO receptor and JAK2". The Journal of Biological Chemistry 275 (38): 29338–47. Sep 2000. doi:10.1074/jbc.M003456200. PMID 10882725.
- ↑ "A new high affinity binding site for suppressor of cytokine signaling-3 on the erythropoietin receptor". European Journal of Biochemistry 269 (10): 2516–26. May 2002. doi:10.1046/j.1432-1033.2002.02916.x. PMID 12027890.
- ↑ "Physical and functional interactions between Stat5 and the tyrosine-phosphorylated receptors for erythropoietin and interleukin-3". Blood 88 (12): 4415–25. Dec 1996. doi:10.1182/blood.V88.12.4415.bloodjournal88124415. PMID 8977232.
Further reading
- "The molecular physiology of erythropoietin and the erythropoietin receptor". Current Opinion in Hematology 1 (2): 113–8. Mar 1994. PMID 9371269.
- "Biology of erythropoietin". Haematologica 83 (8): 724–32. Aug 1998. PMID 9793257.
- "Quality control of receptor-kinase signaling complexes". Developmental Cell 2 (1): 1–2. Jan 2002. doi:10.1016/S1534-5807(01)00114-9. PMID 11782306.
- "Erythropoietin receptor in myelodysplastic syndrome and leukemia". Leukemia & Lymphoma 43 (2): 261–4. Feb 2002. doi:10.1080/10428190290006026. PMID 11999556.
- "Molecular pathogenesis of Philadelphia chromosome negative myeloproliferative disorders". Blood Reviews 19 (1): 1–13. Jan 2005. doi:10.1016/j.blre.2004.02.002. PMID 15572213.
- "Switching on reparative angiogenesis: essential role of the vascular erythropoietin receptor". Circulation Research 100 (5): 599–601. Mar 2007. doi:10.1161/01.RES.0000261610.11754.b1. PMID 17363704.
External links
- Erythropoietin+Receptors at the US National Library of Medicine Medical Subject Headings (MeSH)
This article incorporates text from the United States National Library of Medicine, which is in the public domain.
 |