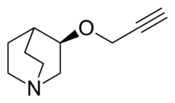
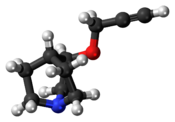
Chemistry:Talsaclidine
From HandWiki
Short description: Chemical compound
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Pharmacokinetic data | |
| Bioavailability | 70% |
| Protein binding | 7% |
| Excretion | Renal (86%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C10H15NO |
| Molar mass | 165.236 g·mol−1 |
| 3D model (JSmol) | |
| |
Talsaclidine (WAL-2014) is a non-selective muscarinic acetylcholine receptor agonist which acts as a full agonist at the M1 subtype, and as a partial agonist at the M2 and M3 subtypes.[1][2][3] It was under development for the treatment of Alzheimer's disease but showed only modest or poor efficacy in rhesus monkeys and humans, respectively,[3][4] perhaps due to an array of dose-limiting side effects including increased heart rate and blood pressure, increased salivation, urinary frequency and burning upon urination, increased lacrimation and nasal secretion, abnormal accommodation, heartburn, upset stomach as well as cramps, nausea, vomiting and diarrhea, excessive sweating and palpitations.[5]
See also
References
- ↑ "WAL 2014--a muscarinic agonist with preferential neuron-stimulating properties". Life Sciences 52 (5–6): 473–80. 1993. doi:10.1016/0024-3205(93)90304-L. PMID 8441328.
- ↑ "In vivo consequences of M1-receptor activation by talsaclidine". Life Sciences 60 (13–14): 977–84. 1997. doi:10.1016/S0024-3205(97)00037-4. PMID 9121364.
- ↑ 3.0 3.1 "Pharmacodynamic profile of the M1 agonist talsaclidine in animals and man". Life Sciences 68 (22–23): 2593–600. April 2001. doi:10.1016/S0024-3205(01)01057-8. PMID 11392631.
- ↑ "Memory-related task performance by aged rhesus monkeys administered the muscarinic M(1)-preferring agonist, talsaclidine". Psychopharmacology 162 (3): 292–300. July 2002. doi:10.1007/s00213-002-1105-3. PMID 12122487.
- ↑ "Phase I clinical trials with WAL 2014, a new muscarinic agonist for the treatment of Alzheimer's disease". Life Sciences 56 (11–12): 883–90. 1995. doi:10.1016/0024-3205(95)00024-Z. PMID 10188789.
 |
