Chemistry:Meclofenoxate
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
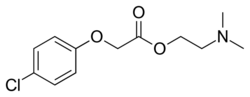
| Formula | C12H16ClNO3 |
| Molar mass | 257.71 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Meclofenoxate (INN, BAN; brand name Lucidril, also known as centrophenoxine) is a cholinergic nootropic used as a dietary supplement.[1][2][3] It is an ester of dimethylethanolamine (DMAE) and 4-chlorophenoxyacetic acid (pCPA).
In elderly patients, meclofenoxate has been shown to improve performance on certain memory tests.[4] Meclofenoxate also increases cellular membrane phospholipids.[citation needed] It is sold in Japan and some European countries, such as Germany , Hungary, and Austria, as a prescription drug.
Side effects
Meclofenoxate is considered to be safe and high in tolerability. However, possible side effects may include, rarely, insomnia, dizziness, restlessness, muscle tremor, depression, nausea, muscle tension, and headache; these side effects may be due to overdosage and may indicate the need for the dosage to be reduced.
Research
Meclofenoxate, as well as DMAE, have been found to increase the lifespans of mice by 26.5%.[5][6]
Brand names
In addition to Lucidril, meclofenoxate has also been marketed under the brand names Amipolen, Analux, Brenal, Cellative, Centrophenoxin, Cerebron, Cerutil, Closete, Helfergin, Lucidryl, Lutiaron, Marucotol, Proserout, Proseryl, and Ropoxyl.[7] In the US, meclofenoxate is sold as a dietary supplement, although it is an unapproved drug.[3]
See also
References
- ↑ The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. 14 November 2014. pp. 758–. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA758.
- ↑ Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 636–. ISBN 978-3-88763-075-1. https://books.google.com/books?id=5GpcTQD_L2oC&pg=PA636.
- ↑ 3.0 3.1 "The unapproved drug centrophenoxine (meclofenoxate) in cognitive enhancement dietary supplements". Clinical Toxicology 60 (10): 1156–1158. October 2022. doi:10.1080/15563650.2022.2109485. PMID 35959800.
- ↑ "The differential effects of meclofenoxate on memory loss in the elderly". Age and Ageing 6 (2): 123–31. May 1977. doi:10.1093/ageing/6.2.123. PMID 329662.
- ↑ "A Brief History of Anti-aging Drugs". The Science of Anti-aging Medicine. American Academy of Anti-Aging Med. 1 January 2003. pp. 66–. ISBN 978-0-9668937-3-1. https://books.google.com/books?id=RI8ZyNo5KCkC&pg=PA66.
- ↑ "Effect of dimethylamnioethyl p-chlorophenoxyacetate on the life span of male Swiss Webster Albino mice". Experimental Gerontology 8 (4): 177–183. August 1973. doi:10.1016/0531-5565(73)90024-7. PMID 4147092.
- ↑ Drugs in Psychiatric Practice. Elsevier. 22 October 2013. ISBN 978-1-4831-9193-5. https://books.google.com/books?id=6gglBQAAQBAJ.
 |
