Biology:Thraustochytrids
| Thraustochytrids | |
|---|---|

| |
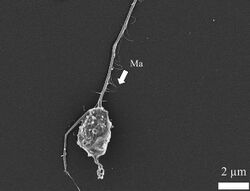
| SEM image of thraustochytrid showing ectoplasmic net. | |
| Scientific classification | |
| Domain: | Eukaryota |
| Clade: | Diaphoretickes |
| Clade: | SAR |
| Clade: | Stramenopiles |
| Phylum: | Bigyra |
| Class: | Labyrinthulea |
| Order: | Thraustochytrida Sparrow, 1973 |
| Families | |
| |
| Synonyms | |
|
Labyrinthulales | |
Thraustochytrids are single-celled saprotrophic eukaryotes (decomposers) that are widely distributed in marine ecosystems, and which secrete enzymes including, but not limited to amylases, proteases, phosphatases.[1][2][3][4][5][6] They are most abundant in regions with high amounts of detritus and decaying plant material.[1] They play an important ecological role in mangroves, where they aid in nutrient cycling by decomposing decaying matter.[7][8][9][10] Additionally, they contribute significantly to the synthesis of omega-3 polyunsaturated fatty acids (PUFAs): docosahexaenoic acid (DHA), and eicosapentaenoic acid (EPA), which are essential fatty acids for the growth and reproduction of crustaceans.[11][12][13] Thraustochytrids are members of the class Labyrinthulea, a group of protists that had previously been incorrectly categorized as fungi due to their similar appearance and lifestyle.[14] With the advent of DNA sequencing technology, labyrinthulomycetes were appropriately placed with other stramenopiles and subsequently categorized as a group of Labyrinthulomycetes.
There are several characteristics which are unique to Thraustochytrids, including their cell wall made of extracellular non-cellulosic scales, zoospores with characteristic heterokont flagella, and a bothrosome-produced ectoplasmic net, which is used for extracellular digestion.[15][16][17][18][19][20][21][22][23] Thraustochytrids are morphologically variable throughout their life cycle. They have a main vegetative asexual cycle, which can vary depending on the genus.[24][10] While sexual reproduction has been observed in this group, it remains poorly understood.[25]
Thraustochytrids are of particular biotechnical interest due to their high concentrations of docosahexaenoic acid (DHA), palmitic acid, carotenoids, and sterols, all of which have beneficial effects to human health.[10][17][26][27][28][29][30][31][32] Thraustochytrids rely on a plethora of resources such as various sources of organic carbon (vitamins and sugars), and inorganic salts throughout their life cycle.[33][34][35][36][37][38][39][40][41] Scientists have devised several potential uses for thraustochytrids stemming around increasing DHA, fatty acids, and squalene concentrations in vivo by either changing the genetic makeup or medium composition/conditioning.[42][43][44][45][46][47][48] There have also been some breakthroughs which have resulted in gene transfers to plant species in order to make isolation of certain oils easier and cost effective.[49][50] Thraustochytrids are currently cultured for use in fish feed and production of dietary supplements for humans and animals.[51][52][53] In addition, scientists are currently researching new methodologies to convert waste water into useful products like squalene, which can then be utilized for the production of biofuel.[54][55][56][57][58]
Morphology
As labyrintulomycetes,[14] thraustochytrids share distinct characteristics with other organisms in this group. These include, but are not limited to: biflagellate zoospores which have an anterior flagellum containing mastigonemes, a bothrosome-produced ectoplasmic net, and multilamellate cell walls with scales derived from Golgi bodies.[19][20][21][22][18][16][15][23][17] Thraustochytrids are single-celled protists, characterized with only one sporangium (monocentric),[59][60] an ectoplasmic net, and a multi-layered, non-cellulosic cell wall made of overlapping circular scales.[59][61] Despite often being referred to as algae, they do not have a plastid, making them obligate heterotrophs.[62]
At their vegetative state, thraustochytrids measure 4 to 20 μm in diameter and are globose or subglobose in shape. They have a multi-layered cell wall made of sulphated galactose.[59] The singular sporangium of thraustochytrids is typically ovular or spherical in shape, and varies across genus.[63] In the Botryochytrium genus, for example, the shape of the zoosporangium was compared to a grape.[63] Thraustochytrids have biflagellate zoospores with heterokont flagella typical of other Stramenopiles. On the posterior end, the whiplash is short, and on the anterior end, a long tinsel flagellum protrudes.[61]
Ultrastructure
Within the granular cytoplasm lies single dictyosomes, centrioles, endoplasmic reticulum, mitochondria, and lipid bodies in some cases. Thraustochytrids contain many mitochondria, which are polymorphic and have tubular cristae.[61] Made of sulphated polysaccharides, the cell wall of thraustochytrids are multilamellate and non-cellulosic.[18][61] The cell wall is derived from the dictyosome cisternae during thallus development, where circular scales (vesicles) form on the basal membrane to merge. In thraustochytrids, the cell wall is rich in galactose and xylose.[18]
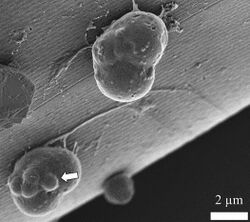
Characteristic of thraustochytrids is their ectoplasmic net—which is an extension of the plasma membrane—emerging from the bothrosome (also known as the sagenogenetosome, or SAG).[19] The cytoplasmic net is unilateral, motile, and resembles fine fibres when viewed under a scanning electron micrograph.[64][63] Depending on the genus, they may be branched or unbranched, and are thought to originate from a single trunk or organelle.[18][1][65] Ectoplasmic nets have the capacity to excrete hydrolytic enzymes (cellulases, amylases, lipases, phosphatases, and/or proteases) to digest organic material in the water, thus assuming the role of decomposition.[66][1][3][4][5] In lab settings, the endoplasmic net of thraustochytrids has been shown the ability to penetrate the sporopollenin of pine pollen, which comprises a plymer that is highly resistant to microbial degradation.[67][68] This experimental process is called pollen-baiting.[67][68] Beyond decomposition, ectoplasmic nets also participate in providing adhesive function, as well as assimilation of digested organic material (absorption).[69]
Life cycle
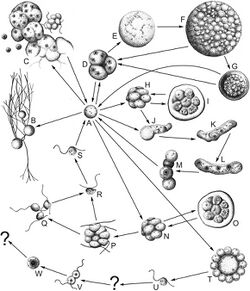
The life cycle of thraustochytrids is generally complicated, differing from genus to genus, and typically consisting of multiple stages of cell types such as zoosporangia, multinucleated cells, mononucleated cells, and amoeboid cells.[24][70][25]
Asexual reproduction
All thraustochytrids undergo a main vegetative life cycle, beginning as a mononucleated cell that undergoes nuclear division to become multinucleated and maturing into a sporangia which release zoospores to begin the cycle again.[10] Branching off of the main vegetative life cycle, additional paths can be taken based on the strain.[10] Traustochytrids undergo cell division in two main ways: through a zoosporangium or through successive bipartition.[61] These methods can occur in the same species and at different stages of the lifecycle. When cell division occurs through the formation of a zoosporangium, the nuclei divide within a single cell to create a multinucleate cell which becomes a zoosporangium following progressive cleavage.[61] When cells divide through successive bipartition, the cell divides immediately after nuclear division, either by invagination of the plasma membrane or fusion with internal vesicular membranes.[61] Thraustochytrids undergo open mitosis, meaning that the nuclear membrane breaks down during cell division, and then reforms following nuclear division.[71]
Amoeboid loop
Certain strains of thraustochytrid are able to enter an amoeboid loop from multiple vegetative life cycle stages, gaining an advantage of being able to move slowly across surfaces as either mononucleated or multinucleated amoeboid cells.[24] Ulkenia, Schizochytrium, Hondaea, and Aurantiochytrium can undergo binary division to form a cluster of mononucleated cells which can then turn into amoeboid cells and enter an amoeboid loop.[61][72] The amoeboid loop can also be entered from mononucleated cells directly turning into mononucleated amoeboid cells or multinucleated cells and sporangia directly turning into multinucleated amoeboid cells.[61] Strains in the amoeboid loop eventually have to re-enter the main vegetative life cycle in order to produce zoosporese.[25]
Sexual reproduction
Although the details of sexual reproduction are poorly understood, vegetative cells are thought to be diploid and undergo meiosis to form a sporangium, which releases gametes.[25] While syngamy has been observed in Aurantiochytrium acetophilum, the fate of the zygote is relatively unknown, however, it is suspected that they enter the vegetative cycle as a mononucleated cell.[25]
Taxonomy
Thraustochytrids were first reported by J.P. Sparrow in 1934.[73] Like other Labyrinthulomycetes, they were classified as fungi due to their ectoplasmic nets and ability to produce zoospores.[74] However, the morphological plasticity of thraustochytrids prevents them from being accurately classified based on their appearance. It was not until 1973 that Sparrow reclassified them as oomycetes, indicating that they were stramenopiles and not fungi.[61] In the years that followed, scientists began to perform concurrent morphological and molecular genetics studies to further explore the placement of thraustochytrids. Using ribosomal RNA as a phylogenetic marker, Cavalier-smith et al. provided strong molecular evidence that indicated thraustochytrids were not closely related to fungi or oomycetes.[75] Other studies supported these findings by highlighting morphological similarities between thraustochytrids and other labyrinthulomycetes.[18][20][23] While the phylogeny of thraustochytrids is still relatively unresolved, they have been clearly defined taxonomically. Thraustochytrida is one of two orders in the class Labyrinthulea and the nomenclature in this group is highly variable due to its history of being considered fungi.[61]
Ecology
Distribution
Thraustochytrids have been found in various habitats such as tropical coasts in the Indian Ocean, Pacific Ocean, and the Northern Arabian sea;[70][76][77] temperate and cold waters in Australia, Argentina, the Mediterranean Sea, and the North sea;[78] and subantarctic, antarctic,[79] and subarctic waters.[80] Overall, thraustochytrids are widespread in marine waters.[81][82][83] They can be found all the way through the photic, euphotic, and aphotic zones.[81][82][83] The ideal salinity range for this protist is ~20‰-30‰; however they are euryhaline and can survive at a salinities as low as 12‰.[84][85][86][87][88][89] Since they require a specific concentration of salt to survive, they are categorized as halophilic protists.[90]
Living conditions
Additionally, they require sodium to live and this cannot be substituted by potassium.[1] They are found at a higher frequency in systems that have large amounts of detritus along with decaying plant material.[1] Areas of note include mangroves, salt marshes, and river output zones.[1] Thraustochytrids gain a significant amount of nutrients for growth from any form of decaying organic matter and, as a result, can thrive in areas with elevated pollution or rich in organic material.[91] They can be found on materials either indigenous (autochthonous) or that has been transported there (allochthonous).[87][66] They are not commonly found on living organisms, and if they are, it is sporadic and in low concentrations.[92] The reasoning for this is suspected to be due to plants being able to release antimicrobial compounds to prevent them from being colonized by microorganisms.[93] In the early stages of decomposition, there are low observed numbers of thraustochytrids as there are still materials that inhibit growth on the organism.[93]
As decomposition progresses, thraustochytrids rapidly populate the substrate.[94][95] In studies involving mangroves, the thraustochytrids on the leaves would produce the enzymes cellulase, amylase, xylanase, proteases, and pectinases, which suggest that they can play a role in the chemical processes taking place.[3] There have been cases of thraustochytrids being cultured from algal surfaces but only in low numbers.[93] Notably, in a case of culturing thraustochytrids on the brown alga Fucus serratus, they were found to be in low numbers potentially due to inhibitory material being secreted by the alga.[93] A 1992 experiment found that thraustochytrids could not be cultured on the green algae Ulva fasciata and Valoniopsis pachynema.[96] These two algae contain high amounts of phenolic compounds, which is believed to be the reason.[96]
Parasitism
There is a lack of concrete evidence regarding parasitic relationships with plants, however studies have found such relationships with invertebrates.[97] In the case of the octopus Eledone cirrhosa, there were found to be ulcerative lesions that could be contagious to other marine organisms.[97] Thraustochytrids could not confidently be determined as the cause of these fatal lesions, with a suggestion being that they came into contact with octopuses after initial infection.[98] Other discoveries of infections similar to what befell Eledone cirrhosa have been noted on oysters, farmed rainbow trout, squid gills, sponges, free living flat worms, cnidarians nudibranchs, and tunicates.[99][100][101][102][103]
Examining cases of parasitic relationships between thraustochytrids and living organisms, the protist can be either the direct cause of disease to the host or an opportunistic parasite.[97][99][100][101][102][103] It is unclear what allows for thraustochytrids to act as a pathogen, however, it appears to be a combination of environmental factors and there being an issue with the host organisms pathogenic defence mechanisms such as being unable to excrete any inhibitory materials.[61]
Other biotic relationships
Studies have found significant amounts of thraustochytrids in the stomach contents and feces of Lytechinus variegatus, a sea urchin.[104] This discovery could be due to either ingestion of detritus, containing thraustochytrids or it could potentially be a regular component of the sea urchin species’ stomach.[104] A species of thraustochytrid, Ulkenia visurgensis have also been found in healthy cnidarians in Indian tidal pools using immunofluorescence.[105] Large amounts of the protist have also been discovered in the feces of the salp Pegea confoederata.[106] These discoveries suggest that there is a relationship between thraustochytrids and the invertebrates mentioned, as well as potentially others in marine environments.[107] Barnacle larvae were also found to survive and grow on substrates where thraustochytrids lived compared to surfaces without, potentially indicating a relationship between the two.[2]
Role in marine food webs
Thraustochytrids play a large role in marine food webs with a significant contribution being in their synthesization of omega-3 polyunsaturated fatty acids (PUFAs): docosahexaenoic acid (DHA), and eicosapentaenoic acid (EPA) which are essential for marine crustaceans.[11][12][13] Their main contributions of these fatty acids to the marine food chain occur in environments where they are able to thrive, usually in areas of high particular detritus in the water column.[11] The PUFAs produced specifically enable growth and reproduction in the crustaceans.[12] Bacteria do not synthesize significant amounts of PUFAs[108] and zooplankton synthesis rates are usually less than 2% of what is required,[67] suggesting that the main source of these fatty acids for them are found further down the food chain and are incorporated into their body from thraustochytrids they feed on.[80] The synthesization of these fatty acids is also important for organisms at higher trophic levels.[109]
Physiology
Since thraustochytrids are obligate heterotrophic protists (non-photosynthetic microalgae), they obtain most of their resources for growth from decaying matter.[10] To act as decomposers, thraustochytrids have evolved to encompass a wide variety of hydrolytic enzymes which include: amylases, proteases, phosphatases, cellulases, lipases, ureases, gelatinase, chitinase, and α-glucosidase. These hydrolytic enzymes are either deposited at the ECM or secreted to the surrounding solution.[1][3][4][6] Ectoplasmic nets have the capacity to excrete hydrolytic enzymes (cellulases, amylases, lipases, phosphatases, and/or proteases) to digest organic material in the water, thus assuming the role of decomposition.[66][1][3][4][5] In lab settings, the endoplasmic net of thraustochytrids has been shown the ability to penetrate the Pine pollen's sporopollenin, which is a highly microbial-resistant polymer.[67][68] This experimental process is called pollen-baiting.[67][68] Beyond decomposition, ectoplasmic nets also participate in providing adhesive function, as well as assimilation of digested organic material (absorption).[69]
Thraustochytrids rely on a wide array of inorganic material for growth such as monopotassium phosphate, sodium chloride, and sodium sulfate. Absence of ions such as potassium can stunt thraustochytrid growth.[33] More specifically, the absence of sodium ions could prevent the uptake of inorganic phosphate that is required for large scale growth conditions.[34] In addition, some species of thraustochytrids can utilize urea as a nitrogen source for growth via a hydrolytic process, which ultimately yields carbon dioxide and ammonia.[6]
In terms of organic carbon sources, thraustochytrids are capable of harnessing organic carbon compounds like maltose, fructose, sucrose, glucose, glycerol, and ethanol for energy expenditure and growth.[35][36][37][38][39] In addition, vitamins such as thiamine, biotin, cobalamin, nicotinic acid, pantothenic acid, and riboflavin are utilized as well.[40][41]
Hong Kong isolates of thraustochytrids species have displayed a wide range of pH for proper growth extending from 4 to 9, however, each individual species exhibited a different range of pH optima. In addition, these Hong Kong isolates tend grow within a temperature range of 20-25 °C, with salinity levels ranging around 7.5-30‰. However, just like the pH ranges, the optima range of temperature and salinity exhibited by each species differed from one another.[110]
Major compounds synthesized
Roughly greater than 65% of fatty acids that constitute thraustochytrids' membranes stem from DHA (22:6) and palmitic acid (16:0).[10] Through unestablished physiological means, thraustochytrids sustained in an environment lacking nitrogen will initiate the synthesis of lipids.[111] It is believed that limitations induced by nitrogen deficiency within the medium can pause cell division, which causes a change in the carbon flux that is used to maintain membrane and protein synthesis and ultimately promotes the production of TAGs.[112] In terms of overall lipid composition, neutral lipids which are mainly constituted of TAGs make up a large portion of glycerolipid distribution relative to polar lipids.[10]
Saturated fatty acids and polyunsaturated fatty acids
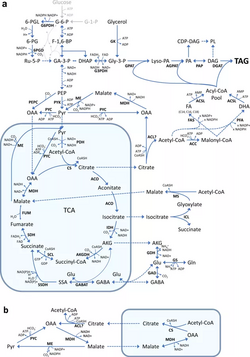
The production of saturated fatty acids and polyunsaturated fatty acids takes place via two pathways which require a type I Fatty Acid Synthase (FAS) construct and a Polyketide Synthase-like (PKS-like) machinery (a.k.a. PUFA synthase), respectively. FAS gives rise to saturated fatty acids that are 16 carbons in length via an aerobic pathway. On the other hand, PUFA synthase gives rise to unsaturated fatty acids that are 20 and 22 carbons in length via an anaerobic pathway. FAS typically produces an abundance of palmitic acid (16:0), while PUFA synthase typically produces an abundance of DHA (22:6).[10][113][114][115][116] It is not certain as to why two different pathways are needed for fatty acid synthesis, but studies have shown that auxotrophic thraustochytrids are a direct result of mutations to the PUFA synthase, thus indicating that the two pathways are not redundant and are independent of one another.[113] It has been reported that if the DH/I domain of PUFA synthase's subunit C is mutated, it will lead to a decrease of greater than 50% to the overall yield of PUFA, thus, indicating its importance to the synthetic pathway and possible exploitation.[48]
Synthesis of DHA
Acetyl-CoA first attaches to KS releasing CoA-SH, then MAT adds a malonyl group to ACP while releasing CoA-SH as a byproduct. KS condenses the activated acetyl with the malonyl group to produce acetoacetyl-CoA, releasing CO2 as a byproduct. KR then reduces acetoacetyl-CoA via NADPH + H+ and the subsequent product is dehydrated via a DH or DH/I dehydratase to produce an acyl chain with a 2-trans double bond (trans-∆2-butenoyl-ACP). The 2-trans double bond may then be reduced via ER utilizing NADPH + H+ (FAS pathway continuation) or isomerized via DH/I leading to a 2,3 or 2,2 trans-cis product (PUFA synthase pathway continuation). The cycle may repeat several times with the addition of two more carbons via either pathway to yield an elongated fatty acid or a precursor to PUFA 22:6 (DHA) through CLF (chain length factor) domain processing.[10][117] It has been reported that the type of DH dehydratase utilized dictates the process towards PUFA 22:6 (DHA) synthesis, which is ultimately determined via the length of an already growing acyl chain.[118]
Synthesis of sterols and carotenoids
To form mevalonate (a precursor to sterols and carotenoids), two acetyl-CoAs combine to form acetoacetyl-CoA, then another acetyl-CoA is added to form HMG-CoA. With the utilization of two NADPH + H+, mevalonate forms. Mevalonate then undergoes three series of reactions with one ATP dedicated to each to form mevalonate-5-PP. Mevalonate-5-PP then loses CO2 and Pi to from ∆3-isopentenyl pyrophosphate, which can isomerize to dimethylallyl pyrophosphate. With the addition of both these components in a head-to-tail condensation, geranyl pyrophosphate can either lead to the synthesis of carotenoids and or sterols via their intermediary product – farnesyl pyrophosphate. Continuing towards sterols, geranyl pyrophosphate with ∆3-isopentenyl pyrophosphate in a head-to-tail condensation will lead to the production of farnesyl pyrophosphate. Farnesyl pyrophosphate then combines with another farnesyl pyrophosphate via utilization of NADPH + H+ to produce squalene (via squalene synthase) – the major precursor to sterol synthesis.[10][117]
Applications
Thraustochytrids produce lots of docosahexaenoic acid (DHA). When cultivated under certain conditions, some thraustochytrids can have their total weight composed of 15-25% DHA.[42] DHA has been reported to have many benefits such as decreasing the onset of depression, having anti-inflammatory properties, decreasing the onset of memory loss via proper neuronal cell development (especially in infants), and many more.[119][26][27][28] Thraustochytrids are also capable of producing carotenoids and sterols, which have been linked to decreasing diseases such as coronary heart disease, cancer, and osteoporosis.[29][30] Furthermore, squalene can be extracted from thraustochytrids, which has benefits linked to activating non-specific immune responses, cancer remedies, UV ionization cell damage reduction, and the capability of acting as an exogenous antioxidant.[31][32]
Potential uses
Thraustochytrids have the potential to overturn exploitation of fish stocks as a new form of sustainable commercial oil producers, while also minimizing losses caused by toxic environmental exposures.[27] This can be accomplished by depriving cells from nitrogen, therefore, triggering the production of DHA.[43] More specifically, a two-stage fermentation protocol could be utilized to accomplish this task. Cells are first grown within a low C:N (high nitrogen content) ratio medium, and then replaced with a high C:N (low nitrogen content) ratio medium, which subsequently prompts an increase in both DHA and FAs yields.[44] If a vast number of 15:0 fatty acids is desired, the low nitrogen medium could be supplied with a surplus of branched amino acids such as valine, isoleucine, leucine.[45]
Squalene (a precursor to sterols) is used to improved human health as a drug delivery system, and a moisturizing agent which is typically derived from shark liver oil.[10][31][32] With an increase in demand for squalene over time, sharks are faced with a major decline in population size. Thus, an incentive for a new form of squalene production was generated, and Aurantiochytrium seems to be a viable solution to this problem.[120][121][122][123]
Scientists have genetically engineered several pathways to increase thraustochytrids’ yield of beneficial products by creating mutants, overexpressing genes, and or introducing knock-ins.[46][47][48] Some experiments had significant results such as producing a 1.5-3-fold increase in fatty acid production, a 2-9 fold increase in astaxanthin production, a 4 fold increase in EPA (20:5) production, and a 2.5-3 fold increase in DHA quantity.[124][125][126][127][128] Recent data has shown that if Aurantiochytrium limacinum SR21 are kept at 50% dissolved oxygen levels within their growth medium, then large increases in both biomass and DHA levels are observed.[129] Other studies and a patent have shown that light could be used to increase biomass, carotenoids, and DHA through either constant or discontinuous illumination using different wavelengths.[85][130][131][132]
Addition of terbinafine hydrochloride and jasmonate to cultures containing certain thraustochytrids strains like Aurantiochytrium mangrovei FB3 and Schizochytrium mangrovei, respectively, have demonstrated an increase in squalene production which could be utilized for sterol synthesis.[120][133] Supplementation of calcium and magnesium ions helped thraustochytrids strains Aurantiochytrium sp. DBTIOC-18 and Schizochytrium sp. DBTIOC-1 to grow under a highly concentrated glycerol medium which typically inhibits growth, and thus gave rise to greater biomass, fatty acid, and DHA production rates.[134]
Extracellular lipases have also been induced in two thraustochytrids strains displaying optimal activity at basic pH, thus giving rise to potential detergent usage.[135] Some scientists have also demonstrated that thraustochytrids could be used to make antigens to fight against influenza and other types of viruses.[136][137] In addition, gene transfers into plant seeds have been successful, thus allowing for the overexpression of PUFA synthase. As a result, these seeds can have their oils isolated and extracted for possible downstream commercial sale.[49][50]
The temperature and or seasons have been reported to alter the fatty acid composition of thraustochytrids isolated from India. Winter isolates depicted a large increase in DHA content which are useful towards nutraceutical applications, while summer isolates depicted a large increase in omega three fatty acids and compounds directly related to biodiesel formulation.[138]
Industrial uses
Companies such as Royal DSM, Alltech, Martek Bioscience, and Ocean Nutrition Canada currently utilize thraustochytrids in some form to produce dietary supplements fit for human and animal consumption.[51][52][53] Food products such as eggs, meat, milk, and baby formula are some of the many examples of products enriched with omega-3 fatty acids derived from thraustochytrids.[52][139][140][141] Many of these products are certified as harmless towards human health by the FDA and European Commission.[142][143][144][145]
Thraustochytrids oils have been used to feed aquaculture such as Atlantic salmon, juveniles of giant grouper, longfin yellowtail, catfish, and salmon parr.[146][147][148][149][150][151][152] It been reported that rotifers are supplemented with polyunsaturated fatty acids using a schizochytrium strain, which are subsequently fed to finfish larvae.[153] Studies have shown that commercial fish supplemented with DHA during the spawning season tend to grow faster and have greater survival rates with reduced abnormalities.[154][155] Astaxanthin, a keto-carotenoid, derived from thraustochytrids have also been used to feed fish, chicken, and turkey, and to even dye food.[10]
Thraustochytrids have also made some break throughs in the biofuel industry. Strains such as Schizochytrium sp. S31 and Schizochytrium mangrovei PQ6 have demonstrated good potential towards the production of certain fuel compounds like biodiesel.[54][55] The Japanese have also developed a new strain of thraustochytrid, Aurantiochytrium 18 W13a, which is capable of producing squalene from sludge waste water. Using ruthenium/cerium oxide catalysis, squalene is then turned into small chain alkanes which can be subsequently used in the production of industrial and commercial fuel.[56][57][58] Scientists have also engineered a new thraustochytrid strain (T18) that can feed on hemi-cellulosic waste generated from feedstocks, and thus produce useful lipids.[156] This was accomplished via overexpressing heterologous xylulose kinase and endogenous xylose isomerase.[156]
A European patent has also demonstrated the capability of oils sourced from thraustochytrids being used towards the creation of thermal insulators.[157]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Raghukumar, Seshagiri (2002-01-01). "Ecology of the marine protists, the Labyrinthulomycetes (Thraustochytrids and Labyrinthulids)" (in en). European Journal of Protistology 38 (2): 127–145. doi:10.1078/0932-4739-00832. ISSN 0932-4739. https://www.sciencedirect.com/science/article/pii/S0932473904700553.
- ↑ 2.0 2.1 Raghukumar, S.; Anil, A. C.; Khandeparker, L.; Patil, J. S. (2000-05-19). "Thraustochytrid protists as a component of marine microbial films". Marine Biology 136 (4): 603–609. doi:10.1007/s002270050720. ISSN 0025-3162. Bibcode: 2000MarBi.136..603R. http://dx.doi.org/10.1007/s002270050720.
- ↑ 3.0 3.1 3.2 3.3 3.4 Raghukumar, S.; Sharma, Sumita; Raghukumar, Chandralata; Sathe-Pathak, Veena; Chandramohan, D. (1994-10-27). "Thraustochytrid and fungal component of marine detritus. IV. Laboratory studies on decomposition of leaves of the mangrove Rhizophora apiculata Blume" (in en). Journal of Experimental Marine Biology and Ecology 183 (1): 113–131. doi:10.1016/0022-0981(94)90160-0. ISSN 0022-0981. https://dx.doi.org/10.1016/0022-0981%2894%2990160-0.
- ↑ 4.0 4.1 4.2 4.3 Nagano, Naoki; Matsui, Shou; Kuramura, Tomoyo; Taoka, Yousuke; Honda, Daiske; Hayashi, Masahiro (2010-05-05). "The Distribution of Extracellular Cellulase Activity in Marine Eukaryotes, Thraustochytrids". Marine Biotechnology 13 (2): 133–136. doi:10.1007/s10126-010-9297-8. ISSN 1436-2228. PMID 20443042. http://dx.doi.org/10.1007/s10126-010-9297-8.
- ↑ 5.0 5.1 5.2 Liu, Ying; Singh, Purnima; Sun, Yuan; Luan, Shengji; Wang, Guangyi (2013-11-24). "Culturable diversity and biochemical features of thraustochytrids from coastal waters of Southern China". Applied Microbiology and Biotechnology 98 (7): 3241–3255. doi:10.1007/s00253-013-5391-y. ISSN 0175-7598. PMID 24270895. http://dx.doi.org/10.1007/s00253-013-5391-y.
- ↑ 6.0 6.1 6.2 TAOKA, Yousuke; NAGANO, Naoki; OKITA, Yuji; IZUMIDA, Hitoshi; SUGIMOTO, Shinichi; HAYASHI, Masahiro (2009-01-23). "Extracellular Enzymes Produced by Marine Eukaryotes, Thraustochytrids". Bioscience, Biotechnology, and Biochemistry 73 (1): 180–182. doi:10.1271/bbb.80416. ISSN 0916-8451. PMID 19129663.
- ↑ Fan, King-Wai; Jiang, Yue; Faan, Yun-Wing; Chen, Feng (2007-03-24). "Lipid Characterization of Mangrove Thraustochytrid − Schizochytrium mangrovei". Journal of Agricultural and Food Chemistry 55 (8): 2906–2910. doi:10.1021/jf070058y. ISSN 0021-8561. PMID 17381126. http://dx.doi.org/10.1021/jf070058y.
- ↑ Margulis, Lynn (1990). Handbook of protoctista : the structure, cultivation, habitats, and life histories of the eukaryotic microorganisms and their descendants exclusive of animals, plants, and fungi. Jones and Bartlett. ISBN 0-86720-052-9. OCLC 802792335. http://worldcat.org/oclc/802792335.
- ↑ Fan, King Wai; Chen, Feng (2007), "Production of High-Value Products by Marine Microalgae Thraustochytrids", Bioprocessing for Value-Added Products from Renewable Resources (Elsevier): pp. 293–323, doi:10.1016/b978-044452114-9/50012-8, ISBN 9780444521149, http://dx.doi.org/10.1016/b978-044452114-9/50012-8, retrieved 2022-03-29
- ↑ 10.00 10.01 10.02 10.03 10.04 10.05 10.06 10.07 10.08 10.09 10.10 10.11 10.12 Morabito, Christian; Bournaud, Caroline; Maës, Cécile; Schuler, Martin; Aiese Cigliano, Riccardo; Dellero, Younès; Maréchal, Eric; Amato, Alberto et al. (2019). "The lipid metabolism in thraustochytrids" (in en). Progress in Lipid Research 76: 101007. doi:10.1016/j.plipres.2019.101007. PMID 31499096.
- ↑ 11.0 11.1 11.2 Lewis, Tom E.; Nichols, Peter D.; McMeekin, Thomas A. (1999). "The Biotechnological Potential of Thraustochytrids". Marine Biotechnology 1 (6): 580–587. doi:10.1007/pl00011813. ISSN 1436-2228. PMID 10612683. Bibcode: 1999MarBt...1..580L. http://dx.doi.org/10.1007/pl00011813.
- ↑ 12.0 12.1 12.2 Harrison, K.E. (1990). "The role of nutrition in maturation, reproduction and embryonic development of decapod crustaceans: A review". Journal of Shellfish Research 9: 1–28.
- ↑ 13.0 13.1 Veloza, Adriana J.; Chu, Fu-Lin E.; Tang, Kam W. (2005-11-18). "Trophic modification of essential fatty acids by heterotrophic protists and its effects on the fatty acid composition of the copepod Acartia tonsa". Marine Biology 148 (4): 779–788. doi:10.1007/s00227-005-0123-1. ISSN 0025-3162. http://dx.doi.org/10.1007/s00227-005-0123-1.
- ↑ 14.0 14.1 Lowenstam, Heinz A.; Weiner, Stephen (1989-07-27), "Protoctista", On Biomineralization (Oxford University Press), doi:10.1093/oso/9780195049770.003.0006, ISBN 978-0-19-504977-0, http://dx.doi.org/10.1093/oso/9780195049770.003.0006, retrieved 2022-03-27
- ↑ 15.0 15.1 AMON, JAMES P.; PERKINS, FRANK O. (1968). "Structure of Labyrinthula sp. Zoospores*". The Journal of Protozoology 15 (3): 543–546. doi:10.1111/j.1550-7408.1968.tb02172.x. ISSN 0022-3921. http://dx.doi.org/10.1111/j.1550-7408.1968.tb02172.x.
- ↑ 16.0 16.1 Kazama, Frederick Y. (1974). "The ultrastructure of nuclear division inThraustochytrium sp.". Protoplasma 82 (3): 155–175. doi:10.1007/bf01276306. ISSN 0033-183X. http://dx.doi.org/10.1007/bf01276306.
- ↑ 17.0 17.1 17.2 Perkins, E.J. (1974), "The Marine Environment", Biology of Plant Litter Decomposition (Elsevier): pp. 683–721, doi:10.1016/b978-0-12-215002-9.50020-4, ISBN 9780122150029, http://dx.doi.org/10.1016/b978-0-12-215002-9.50020-4, retrieved 2022-03-27
- ↑ 18.0 18.1 18.2 18.3 18.4 18.5 MOSS, STEPHEN T. (1985). "An ultrastructural study of taxonomically significant characters of the Thraustochytriales and the Labyrinthulales". Botanical Journal of the Linnean Society 91 (1–2): 329–357. doi:10.1111/j.1095-8339.1985.tb01154.x. ISSN 0024-4074. http://dx.doi.org/10.1111/j.1095-8339.1985.tb01154.x.
- ↑ 19.0 19.1 19.2 Perkins, Frank O. (1972). "The ultrastructure of holdfasts, ?rhizoids?, and ?slime tracks? in thraustochytriaceous fungi and Labyrinthula spp.". Archiv für Mikrobiologie 84 (2): 95–118. doi:10.1007/bf00412431. ISSN 0302-8933. PMID 4559400. http://dx.doi.org/10.1007/bf00412431.
- ↑ 20.0 20.1 20.2 Perkins, Frank O. (1973-02-01). "Observations of thraustochytriaceous (Phycomycetes) and labyrinthulid (Rhizopodea) ectoplasmic nets on natural and artificial substrates—an electron microscope study". Canadian Journal of Botany 51 (2): 485–491. doi:10.1139/b73-057. ISSN 0008-4026. http://dx.doi.org/10.1139/b73-057.
- ↑ 21.0 21.1 Porter, David (1972). "Cell division in the marine slime mold,Labyrinthula sp., and the role of the bothrosome in extracellular membrane production". Protoplasma 74 (4): 427–448. doi:10.1007/bf01281960. ISSN 0033-183X. http://dx.doi.org/10.1007/bf01281960.
- ↑ 22.0 22.1 Bennett, Reuel M.; Honda, D.; Beakes, Gordon W.; Thines, Marco (2017), "Labyrinthulomycota", Handbook of the Protists (Cham: Springer International Publishing): pp. 507–542, doi:10.1007/978-3-319-28149-0_25, ISBN 978-3-319-28147-6, http://dx.doi.org/10.1007/978-3-319-28149-0_25, retrieved 2022-03-27
- ↑ 23.0 23.1 23.2 Alderman, D. J.; Harrison, J. L.; Bremer, G. B.; Jones, E. B. G. (1974). "Taxonomic revisions in the marine biflagellate fungi: The ultrastructural evidence". Marine Biology 25 (4): 345–357. doi:10.1007/bf00404978. ISSN 0025-3162. Bibcode: 1974MarBi..25..345A. http://dx.doi.org/10.1007/bf00404978.
- ↑ 24.0 24.1 24.2 Honda, Daiske; Yokochi, Toshihiro; Nakahara, Toro; Erata, Mayumi; Higashihara, Takanori (1998-04-01). "Schizochytrium limacinum sp. nov., a new thraustochytrid from a mangrove area in the west Pacific Ocean" (in en). Mycological Research 102 (4): 439–448. doi:10.1017/S0953756297005170. ISSN 0953-7562. https://www.sciencedirect.com/science/article/pii/S0953756208608977.
- ↑ 25.0 25.1 25.2 25.3 25.4 Ganuza, Eneko; Yang, Shanshan; Amezquita, Magdalena; Giraldo-Silva, Ana; Andersen, Robert A. (2019-04-01). "Genomics, Biology and Phylogeny Aurantiochytrium acetophilum sp. nov. (Thraustrochytriaceae), Including First Evidence of Sexual Reproduction" (in en). Protist 170 (2): 209–232. doi:10.1016/j.protis.2019.02.004. ISSN 1434-4610. PMID 31100647.
- ↑ 26.0 26.1 Innis, Sheila M. (2008-10-27). "Dietary omega 3 fatty acids and the developing brain" (in en). Brain Research. Brain, Genes and Nutrition 1237: 35–43. doi:10.1016/j.brainres.2008.08.078. ISSN 0006-8993. PMID 18789910. https://www.sciencedirect.com/science/article/pii/S0006899308021033.
- ↑ 27.0 27.1 27.2 Byreddy, Avinesh R. (2016). "Thraustochytrids as an alternative source of omega-3 fatty acids, carotenoids and enzymes". Lipid Technology 28 (3–4): 68–70. doi:10.1002/lite.201600019. ISSN 0956-666X. https://doi.org/10.1002/lite.201600019.
- ↑ 28.0 28.1 Calder, Philip C. (2015). "Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance" (in en). Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 1851 (4): 469–484. doi:10.1016/j.bbalip.2014.08.010. PMID 25149823. https://linkinghub.elsevier.com/retrieve/pii/S1388198114001656.
- ↑ 29.0 29.1 Gyles, Collin L.; Carlberg, Jared G.; Gustafson, Jennifer; Davlut, David A. A.; Jones, Peter J. H. (2010-10-07). "Economic valuation of the potential health benefits from foods enriched with plant sterols in Canada" (in en). Food & Nutrition Research 54: 5113. doi:10.3402/fnr.v54i0.5113. ISSN 1654-661X. PMID 20941328. PMC 2952539. https://foodandnutritionresearch.net/index.php/fnr/article/view/430.
- ↑ 30.0 30.1 Rao, A; Rao, L (2007). "Carotenoids and human health" (in en). Pharmacological Research 55 (3): 207–216. doi:10.1016/j.phrs.2007.01.012. PMID 17349800. https://linkinghub.elsevier.com/retrieve/pii/S1043661807000357.
- ↑ 31.0 31.1 31.2 Kelly, G. S. (1999). "Squalene and its potential clinical uses". Alternative Medicine Review 4 (1): 29–36. ISSN 1089-5159. PMID 9988781. https://pubmed.ncbi.nlm.nih.gov/9988781.
- ↑ 32.0 32.1 32.2 Reddy, L. Harivardhan; Couvreur, Patrick (2009-12-17). "Squalene: A natural triterpene for use in disease management and therapy" (in en). Advanced Drug Delivery Reviews. 2009 Editors' Collection 61 (15): 1412–1426. doi:10.1016/j.addr.2009.09.005. ISSN 0169-409X. PMID 19804806. https://www.sciencedirect.com/science/article/pii/S0169409X09002841.
- ↑ 33.0 33.1 Guenther, Bahnweg (1979). "Studies on the physiology of Thraustochytriales I. Growth requirements and nitrogen nutrition of Thraustochytrium spp., Schizochytrium sp., Japonochytrium sp., Ulkenia spp., and Labyrinthuloides spp.". Veroff Inst Meeresforsch Bremerhaven 17: 245–268.
- ↑ 34.0 34.1 Siegenthaler, Paul A.; Belsky, Melvin M.; Goldstein, Solomon; Menna, Maria (1967). "Phosphate Uptake in an Obligately Marine Fungus II. Role of Culture Conditions, Energy Sources, and Inhibitors" (in en). Journal of Bacteriology 93 (4): 1281–1288. doi:10.1128/jb.93.4.1281-1288.1967. ISSN 0021-9193. PMID 4166323.
- ↑ 35.0 35.1 Lee Chang, Kim Jye; Nichols, Carol Mancuso; Blackburn, Susan I.; Dunstan, Graeme A.; Koutoulis, Anthony; Nichols, Peter D. (2014). "Comparison of Thraustochytrids Aurantiochytrium sp., Schizochytrium sp., Thraustochytrium sp., and Ulkenia sp. for Production of Biodiesel, Long-Chain Omega-3 Oils, and Exopolysaccharide" (in en). Marine Biotechnology 16 (4): 396–411. doi:10.1007/s10126-014-9560-5. ISSN 1436-2228. PMID 24463839. Bibcode: 2014MarBt..16..396L. http://link.springer.com/10.1007/s10126-014-9560-5.
- ↑ 36.0 36.1 Scott, Spencer D.; Armenta, Roberto E.; Berryman, Kevin T.; Norman, Andrew W. (2011). "Use of raw glycerol to produce oil rich in polyunsaturated fatty acids by a thraustochytrid" (in en). Enzyme and Microbial Technology 48 (3): 267–272. doi:10.1016/j.enzmictec.2010.11.008. PMID 22112910. https://linkinghub.elsevier.com/retrieve/pii/S0141022910002590.
- ↑ 37.0 37.1 Quilodrán, B.; Hinzpeter, I.; Quiroz, A.; Shene, C. (2009). "Evaluation of liquid residues from beer and potato processing for the production of docosahexaenoic acid (C22:6n-3, DHA) by native thraustochytrid strains" (in en). World Journal of Microbiology and Biotechnology 25 (12): 2121–2128. doi:10.1007/s11274-009-0115-2. ISSN 0959-3993. http://link.springer.com/10.1007/s11274-009-0115-2.
- ↑ 38.0 38.1 Shene, Carolina; Leyton, Allison; Rubilar, Mónica; Pinelo, Manuel; Acevedo, Francisca; Morales, Eduardo (2013-05-31). "Production of lipids and docosahexasaenoic acid (DHA) by a native Thraustochytrium strain". European Journal of Lipid Science and Technology 115 (8): 890–900. doi:10.1002/ejlt.201200417. ISSN 1438-7697. https://doi.org/10.1002/ejlt.201200417.
- ↑ 39.0 39.1 Xie, Yunxuan; Wang, Guangyi (2015). "Mechanisms of fatty acid synthesis in marine fungus-like protists" (in en). Applied Microbiology and Biotechnology 99 (20): 8363–8375. doi:10.1007/s00253-015-6920-7. ISSN 0175-7598. PMID 26286514. http://link.springer.com/10.1007/s00253-015-6920-7.
- ↑ 40.0 40.1 Fan, King Wai; Chen, Feng (2007-01-01), Yang, Shang-Tian, ed., "Chapter 11 - Production of High-Value Products by Marine Microalgae Thraustochytrids" (in en), Bioprocessing for Value-Added Products from Renewable Resources (Amsterdam: Elsevier): pp. 293–323, doi:10.1016/b978-044452114-9/50012-8, ISBN 978-0-444-52114-9, https://www.sciencedirect.com/science/article/pii/B9780444521149500128, retrieved 2022-03-16
- ↑ 41.0 41.1 Raghukumar, Seshagiri (2017) (in en-gb). Fungi in Coastal and Oceanic Marine Ecosystems. doi:10.1007/978-3-319-54304-8. ISBN 978-3-319-54303-1. https://link.springer.com/book/10.1007/978-3-319-54304-8.
- ↑ 42.0 42.1 Raghukumar, Seshagiri (2008-12-01). "Thraustochytrid Marine Protists: Production of PUFAs and Other Emerging Technologies" (in en). Marine Biotechnology 10 (6): 631–640. doi:10.1007/s10126-008-9135-4. ISSN 1436-2236. PMID 18712565. Bibcode: 2008MarBt..10..631R. https://doi.org/10.1007/s10126-008-9135-4.
- ↑ 43.0 43.1 Meï, Coline E.; Cussac, Mathilde; Haslam, Richard P.; Beaudoin, Frédéric; Wong, Yung-Sing; Maréchal, Eric; Rébeillé, Fabrice (2017). "C1 Metabolism Inhibition and Nitrogen Deprivation Trigger Triacylglycerol Accumulation in Arabidopsis thaliana Cell Cultures and Highlight a Role of NPC in Phosphatidylcholine-to-Triacylglycerol Pathway". Frontiers in Plant Science 7: 2014. doi:10.3389/fpls.2016.02014. ISSN 1664-462X. PMID 28101097.
- ↑ 44.0 44.1 Rosa, Silvina Mariana; Soria, Marcelo Abel; Vélez, Carlos Guillermo; Galvagno, Miguel Angel (2010-04-01). "Improvement of a two-stage fermentation process for docosahexaenoic acid production by Aurantiochytrium limacinum SR21 applying statistical experimental designs and data analysis" (in en). Bioresource Technology 101 (7): 2367–2374. doi:10.1016/j.biortech.2009.11.056. ISSN 0960-8524. PMID 20015637. https://www.sciencedirect.com/science/article/pii/S0960852409015600.
- ↑ 45.0 45.1 Crown, Scott B.; Marze, Nicholas; Antoniewicz, Maciek R. (2015-12-28). "Catabolism of Branched Chain Amino Acids Contributes Significantly to Synthesis of Odd-Chain and Even-Chain Fatty Acids in 3T3-L1 Adipocytes" (in en). PLOS ONE 10 (12): e0145850. doi:10.1371/journal.pone.0145850. ISSN 1932-6203. PMID 26710334. Bibcode: 2015PLoSO..1045850C.
- ↑ 46.0 46.1 Cui, Gu-Zhen; Ma, Zengxin; Liu, Ya-Jun; Feng, Yingang; Sun, Zhijie; Cheng, Yurong; Song, Xiaojin; Cui, Qiu (2016-11-01). "Overexpression of glucose-6-phosphate dehydrogenase enhanced the polyunsaturated fatty acid composition of Aurantiochytrium sp. SD116" (in en). Algal Research 19: 138–145. doi:10.1016/j.algal.2016.08.005. ISSN 2211-9264. https://www.sciencedirect.com/science/article/pii/S2211926416302636.
- ↑ 47.0 47.1 Liu, Zhu; Zang, Xiaonan; Cao, Xuexue; Wang, Zhendong; Liu, Chang; Sun, Deguang; Guo, Yalin; Zhang, Feng et al. (2018-12-11). "Cloning of the pks3 gene of Aurantiochytrium limacinum and functional study of the 3-ketoacyl-ACP reductase and dehydratase enzyme domains" (in en). PLOS ONE 13 (12): e0208853. doi:10.1371/journal.pone.0208853. ISSN 1932-6203. PMID 30533058. Bibcode: 2018PLoSO..1308853L.
- ↑ 48.0 48.1 48.2 Li, Zhipeng; Chen, Xi; Li, Jun; Meng, Tong; Wang, Lingwei; Chen, Zhen; Shi, Yanyan; Ling, Xueping et al. (2018-12-01). "Functions of PKS Genes in Lipid Synthesis of Schizochytrium sp. by Gene Disruption and Metabolomics Analysis" (in en). Marine Biotechnology 20 (6): 792–802. doi:10.1007/s10126-018-9849-x. ISSN 1436-2236. PMID 30136198. Bibcode: 2018MarBt..20..792L. https://doi.org/10.1007/s10126-018-9849-x.
- ↑ 49.0 49.1 Abbadi, Amine; Domergue, Freéderic; Bauer, Jörg; Napier, Johnathan A.; Welti, Ruth; Zähringer, Ulrich; Cirpus, Petra; Heinz, Ernst (2004-10-01). "Biosynthesis of Very-Long-Chain Polyunsaturated Fatty Acids in Transgenic Oilseeds: Constraints on Their Accumulationw⃞" (in en). The Plant Cell 16 (10): 2734–2748. doi:10.1105/tpc.104.026070. ISSN 1532-298X. PMID 15377762.
- ↑ 50.0 50.1 Qi, Baoxiu; Fraser, Tom; Mugford, Sam; Dobson, Gary; Sayanova, Olga; Butler, Justine; Napier, Johnathan A.; Stobart, A. Keith et al. (2004). "Production of very long chain polyunsaturated omega-3 and omega-6 fatty acids in plants" (in en). Nature Biotechnology 22 (6): 739–745. doi:10.1038/nbt972. ISSN 1546-1696. PMID 15146198. https://www.nature.com/articles/nbt972.
- ↑ 51.0 51.1 "Pets" (in en). https://www.dsm.com/anh/species/pets.html.
- ↑ 52.0 52.1 52.2 "Nutritional Lipids in Maternal and Infant Health | DSM Human Nutrition & Health" (in en). https://www.dsm.com/human-nutrition/en/products/nutritional-lipids/nutritional-lipids-in-maternal-and-infant-health.html.
- ↑ 53.0 53.1 "Why life's Products?" (in en). https://www.lifesdha.com/en_US/why-life-s-products-.html.
- ↑ 54.0 54.1 Byreddy, Avinesh R.; Gupta, Adarsha; Barrow, Colin J.; Puri, Munish (2015). "Comparison of Cell Disruption Methods for Improving Lipid Extraction from Thraustochytrid Strains" (in en). Marine Drugs 13 (8): 5111–5127. doi:10.3390/md13085111. ISSN 1660-3397. PMID 26270668.
- ↑ 55.0 55.1 Tohoku University (2013). Leading the restoration of Tohoku and the regeneration of Japan Vol.3. Japan: Tohoku University. pp. 1–24. https://www.tohoku.ac.jp/en/about/images/reconstruction_vol3.pdf.
- ↑ 56.0 56.1 Tohoku University. "A new method of converting algal oil to transportation fuels" (in en). https://phys.org/news/2015-06-method-algal-oil-fuels.html.
- ↑ 57.0 57.1 Tohoku University (2021). Leading the Restoration of Tohoku and the Regeneration of Japan Vol.9. Japan: Tohoku University. pp. 1–200.
- ↑ 58.0 58.1 Oya, Shin-ichi; Kanno, Daisuke; Watanabe, Hideo; Tamura, Masazumi; Nakagawa, Yoshinao; Tomishige, Keiichi (2015-06-11). "Catalytic Production of Branched Small Alkanes from Biohydrocarbons". ChemSusChem 8 (15): 2472–2475. doi:10.1002/cssc.201500375. ISSN 1864-5631. PMID 26097221. Bibcode: 2015ChSCh...8.2472O. https://doi.org/10.1002/cssc.201500375.
- ↑ 59.0 59.1 59.2 V., Raghukumar, S. Damare (2009-11-06). Ecological Studies and Molecular Characterization of Thraustochytrids and Aplanochytrids from Oceanic Water Column. OCLC 713050601. http://worldcat.org/oclc/713050601.
- ↑ Raghukumar, Chandralata; Damare, Samir (2014-04-30), "Deep-Sea Fungi", High-Pressure Microbiology (Washington, DC, USA: ASM Press): pp. 265–291, doi:10.1128/9781555815646.ch15, ISBN 9781683671473, http://dx.doi.org/10.1128/9781555815646.ch15, retrieved 2022-03-27
- ↑ 61.00 61.01 61.02 61.03 61.04 61.05 61.06 61.07 61.08 61.09 61.10 61.11 Fossier Marchan, Loris; Lee Chang, Kim J.; Nichols, Peter D.; Mitchell, Wilfrid J.; Polglase, Jane L.; Gutierrez, Tony (2018-01-01). "Taxonomy, ecology and biotechnological applications of thraustochytrids: A review" (in en). Biotechnology Advances 36 (1): 26–46. doi:10.1016/j.biotechadv.2017.09.003. ISSN 0734-9750. PMID 28911809. https://www.sciencedirect.com/science/article/pii/S0734975017301143.
- ↑ Leyland, Ben; Leu, Stefan; Boussiba, Sammy (October 2017). "Are Thraustochytrids algae?". Fungal Biology 121 (10): 835–840. doi:10.1016/j.funbio.2017.07.006. ISSN 1878-6146. PMID 28889907. http://dx.doi.org/10.1016/j.funbio.2017.07.006.
- ↑ 63.0 63.1 63.2 ICAR-Central Marine Fisheries Research Institute, Kochi-682018, Kerala, India; Jaseera, K. V.; Kaladharan, P. (2021-01-15). "An overview of systematics, morphology, biodiversity and potential utilisation of Thraustochytrids". Journal of the Marine Biological Association of India 62 (2): 13–21. doi:10.6024/jmbai.2020.62.2.2136-02. http://mbai.org.in/php/journaldload.php?id=2557&bkid=125.
- ↑ Hamamoto, Yoko; Honda, Daiske (2019-01-09). Ianora, Adrianna. ed. "Nutritional intake of Aplanochytrium (Labyrinthulea, Stramenopiles) from living diatoms revealed by culture experiments suggesting the new prey–predator interactions in the grazing food web of the marine ecosystem" (in en). PLOS ONE 14 (1): e0208941. doi:10.1371/journal.pone.0208941. ISSN 1932-6203. PMID 30625142. Bibcode: 2019PLoSO..1408941H.
- ↑ Sparrow, F. K. (1960). Aquatic Phycomycetes.. Ann Arbor: University of Michigan Press. doi:10.5962/bhl.title.5685. http://dx.doi.org/10.5962/bhl.title.5685.
- ↑ 66.0 66.1 66.2 Kimura, H; Fukuba, T; Naganuma, T (1999). "Biomass of thraustochytrid protoctists in coastal water". Marine Ecology Progress Series 189: 27–33. doi:10.3354/meps189027. ISSN 0171-8630. Bibcode: 1999MEPS..189...27K.
- ↑ 67.0 67.1 67.2 67.3 67.4 Raghukumar, Seshagiri; Damare, Varada S. (2011-01-01). "Increasing evidence for the important role of Labyrinthulomycetes in marine ecosystems". Botanica Marina 54 (1). doi:10.1515/bot.2011.008. ISSN 1437-4323. http://dx.doi.org/10.1515/bot.2011.008.
- ↑ 68.0 68.1 68.2 68.3 Gupta, Adarsha; Singh, Dilip; Byreddy, Avinesh R.; Thyagarajan, Tamilselvi; Sonkar, Shailendra P.; Mathur, Anshu S.; Tuli, Deepak K.; Barrow, Colin J. et al. (2015-12-07). "Exploring omega-3 fatty acids, enzymes and biodiesel producing thraustochytrids from Australian and Indian marine biodiversity". Biotechnology Journal 11 (3): 345–355. doi:10.1002/biot.201500279. ISSN 1860-6768. PMID 26580151.
- ↑ 69.0 69.1 Nakai, Ryosuke; Naganuma, Takeshi (2015), "Diversity and Ecology of Thraustochytrid Protists in the Marine Environment", Marine Protists (Tokyo: Springer Japan): pp. 331–346, doi:10.1007/978-4-431-55130-0_13, ISBN 978-4-431-55129-4, http://dx.doi.org/10.1007/978-4-431-55130-0_13, retrieved 2022-03-27
- ↑ 70.0 70.1 Bongiorni, Lucia; Jain, Ruchi; Raghukumar, Seshagiri; Aggarwal, Ramesh Kumar (2005-11-17). "Thraustochytrium gaertnerium sp. nov.: a New Thraustochytrid Stramenopilan Protist from Mangroves of Goa, India" (in en). Protist 156 (3): 303–315. doi:10.1016/j.protis.2005.05.001. ISSN 1434-4610. PMID 16325543. https://www.sciencedirect.com/science/article/pii/S1434461005000398.
- ↑ Kumar, S. Raghu (1982-07-01). "Fine structure of the thraustochytrid Ulkenia amoeboidea. II. The amoeboid stage and formation of zoospores". Canadian Journal of Botany 60 (7): 1103–1114. doi:10.1139/b82-140. ISSN 0008-4026. http://dx.doi.org/10.1139/b82-140.
- ↑ Dellero, Younès; Rose, Suzanne; Metton, Coralie; Morabito, Christian; Lupette, Josselin; Jouhet, Juliette; Maréchal, Eric; Rébeillé, Fabrice et al. (2018). "Ecophysiology and lipid dynamics of a eukaryotic mangrove decomposer". Environmental Microbiology 20 (8): 3057–3068. doi:10.1111/1462-2920.14346. ISSN 1462-2912. PMID 29968288. Bibcode: 2018EnvMi..20.3057D. https://doi.org/10.1111/1462-2920.14346.
- ↑ SPARROW, F. K. (1936). "Biological Observations on the Marine Fungi of Woods Hole Waters". The Biological Bulletin 70 (2): 236–263. doi:10.2307/1537470. ISSN 0006-3185. http://dx.doi.org/10.2307/1537470.
- ↑ Ellenbogen, Barbara B.; Aaronson, S.; Goldstein, S.; Belsky, M. (1969). "Polyunsaturated fatty acids of aquatic fungi: Possible phylogenetic significance". Comparative Biochemistry and Physiology 29 (2): 805–811. doi:10.1016/0010-406x(69)91631-4. ISSN 0010-406X. http://dx.doi.org/10.1016/0010-406x(69)91631-4.
- ↑ Cavalier-Smith, T.; Allsopp, M. T. E. P.; Chao, E. E. (1994-12-29). "Thraustochytrids are chromists, not Fungi: 18s rRNA signatures of Heterokonta". Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences 346 (1318): 387–397. doi:10.1098/rstb.1994.0156. ISSN 0962-8436. Bibcode: 1994RSPTB.346..387C. http://dx.doi.org/10.1098/rstb.1994.0156.
- ↑ Lee Chang, Kim Jye; Dunstan, Graeme A.; Abell, Guy C. J.; Clementson, Lesley A.; Blackburn, Susan I.; Nichols, Peter D.; Koutoulis, Anthony (2012). "Biodiscovery of new Australian thraustochytrids for production of biodiesel and long-chain omega-3 oils" (in en). Applied Microbiology and Biotechnology 93 (5): 2215–2231. doi:10.1007/s00253-011-3856-4. ISSN 0175-7598. PMID 22252264. http://link.springer.com/10.1007/s00253-011-3856-4.
- ↑ Ramaiah, N.; Raghukumar, S.; Mangesh, G.; Madhupratap, M. (2005). "Seasonal variations in carbon biomass of bacteria, thraustochytrids and microzooplankton in the Northern Arabian Sea" (in en). Deep Sea Research Part II: Topical Studies in Oceanography 52 (14–15): 1910–1921. doi:10.1016/j.dsr2.2005.05.004. Bibcode: 2005DSRII..52.1910R. https://linkinghub.elsevier.com/retrieve/pii/S0967064505001244.
- ↑ Bongiorni, L; Dini, F (2002). "Distribution and abundance of thraustochytrids in different Mediterranean coastal habitats" (in en). Aquatic Microbial Ecology 30: 49–56. doi:10.3354/ame030049. ISSN 0948-3055. http://www.int-res.com/abstracts/ame/v30/n1/p49-56/.
- ↑ Bahnweg, Günther; Sparrow, Frederick K. (1974). "Four New Species of Thraustochytrium from Antarctic Regions, with Notes on the Distribution of Zoosporic Fungi in the Antarctic Marine Ecosystems". American Journal of Botany 61 (7): 754–766. doi:10.1002/j.1537-2197.1974.tb12298.x. ISSN 0002-9122. http://dx.doi.org/10.1002/j.1537-2197.1974.tb12298.x.
- ↑ 80.0 80.1 Naganuma T.; Kimura H.; Karimoto R. (2006). "Abundance of planktonic thraustochytrids and bacteria and the concentration of particulate ATP in the Greenland and Norwegian seas". Polar Bioscience 20: 37–45. https://core.ac.uk/download/pdf/51485705.pdf.
- ↑ 81.0 81.1 Bongiorni, L; Pusceddu, A; Danovaro, R (2005). "Enzymatic activities of epiphytic and benthic thraustochytrids involved in organic matter degradation" (in en). Aquatic Microbial Ecology 41: 299–305. doi:10.3354/ame041299. ISSN 0948-3055. http://www.int-res.com/abstracts/ame/v41/n3/p299-305/.
- ↑ 82.0 82.1 Li, Qian; Wang, Xin; Liu, Xianhua; Jiao, Nianzhi; Wang, Guangyi (2013). "Abundance and Novel Lineages of Thraustochytrids in Hawaiian Waters" (in en). Microbial Ecology 66 (4): 823–830. doi:10.1007/s00248-013-0275-3. ISSN 0095-3628. PMID 23942794. Bibcode: 2013MicEc..66..823L. http://link.springer.com/10.1007/s00248-013-0275-3.
- ↑ 83.0 83.1 Raghukumar, S; Ramaiah, N; Raghukumar, C (2001). "Dynamics of thraustochytrid protists in the water column of the Arabian Sea" (in en). Aquatic Microbial Ecology 24: 175–186. doi:10.3354/ame024175. ISSN 0948-3055. http://www.int-res.com/abstracts/ame/v24/n2/p175-186/.
- ↑ Goldstein, Solomon (1963). "Development and Nutrition of New Species of Thraustochytrium". American Journal of Botany 50 (3): 271–279. doi:10.1002/j.1537-2197.1963.tb12234.x. ISSN 0002-9122. https://doi.org/10.1002/j.1537-2197.1963.tb12234.x.
- ↑ 85.0 85.1 Goldstein, Solomon (1963). "Studies of a New Species of Thraustochytrium That Displays Light Stimulated Growth". Mycologia 55 (6): 799–811. doi:10.2307/3756484. ISSN 0027-5514. https://www.jstor.org/stable/3756484.
- ↑ Goldstein, Solomon (1963). "Morphological variation and nutrition of a new monocentric marine fungus" (in en). Archiv für Mikrobiologie 45 (1): 101–110. doi:10.1007/BF00410299. ISSN 0302-8933. PMID 13948825. http://link.springer.com/10.1007/BF00410299.
- ↑ 87.0 87.1 Goldstein, Solomon; Belsky, Melvin (1964). "Axenic Culture Studies of a New Marine Phycomycete Possessing an Unusual Type of Asexual Reproduction". American Journal of Botany 51 (1): 72–78. doi:10.1002/j.1537-2197.1964.tb06602.x. ISSN 0002-9122. http://dx.doi.org/10.1002/j.1537-2197.1964.tb06602.x.
- ↑ Alderman, D.J.; Gareth Jones, E.B. (1971). "Physiological requirements of two marine phycomycetes, Althornia crouchii and Ostracoblabe implexa" (in en). Transactions of the British Mycological Society 57 (2): 213–IN3. doi:10.1016/S0007-1536(71)80003-7. https://linkinghub.elsevier.com/retrieve/pii/S0007153671800037.
- ↑ Jones E.B.G.; Harrison J.L. (1976). "Physiology of marine Phycomycetes". Recent Advances in Aquatic Mycology: 261–278.
- ↑ Rabinowitz, C.; Douek, J.; Weisz, R.; Shabtay, A.; Rinkevich, B. (December 2006). "Isolation and characterization of four novel thraustochytrid strains from a colonial tunicate" (in en-US). Indian Journal of Geo-Marine Sciences 35 (4). 2006. ISSN 0379-5136. http://nopr.niscair.res.in/handle/123456789/1533.
- ↑ Lyons, M. Maille; Ward, J. Evan; Uhlinger, Kevin R.; Gast, Rebecca J.; Smolowitz, Roxanna (2005). "Lethal marine snow: Pathogen of bivalve mollusc concealed in marine aggregates" (in en). Limnology and Oceanography 50 (6): 1983–1988. doi:10.4319/lo.2005.50.6.1983. Bibcode: 2005LimOc..50.1983L. http://doi.wiley.com/10.4319/lo.2005.50.6.1983.
- ↑ Johnson, T.W. Jr. (1976). "The Phycomycetes: Morphology and taxonomy". Recent Advances in Aquatic Mycology: 193–211.
- ↑ 93.0 93.1 93.2 93.3 Miller, J. David; Gareth Jones, E. B. (1983). "Observations on the Association of Thraustochytrid Marine Fungi with Decaying Seaweed". Botanica Marina 26 (7). doi:10.1515/botm.1983.26.7.345. ISSN 0006-8055. http://dx.doi.org/10.1515/botm.1983.26.7.345.
- ↑ Sathe-Pathak, Veena; Raghukumar, S.; Raghukumar, Chandralata; Sharma, Sumita (September 1993). "Thraustochytrid and fungal component of marine detritus .I - Field studies on decomposition of the brown alga Sargassum cinereum J. Ag." (in en-US). Indian Journal of Geo-Marine Sciences 22 (3). 1993. ISSN 0975-1033. http://nopr.niscair.res.in/handle/123456789/37868.
- ↑ Raghukumar, S; Sathe-Pathak, V; Sharma, S; Raghukumar, C (1995). "Thraustochytrid and fungal component of marine detritus. III. Field studies on decomposition of leaves of the mangrove Rhizophora apiculata". Aquatic Microbial Ecology 9: 117–125. doi:10.3354/ame009117. ISSN 0948-3055.
- ↑ 96.0 96.1 Raghukumar, C.; Nagarkar, S.; Raghukumar, S. (1992) (in en). Association of thraustochytrids and fungi with living marine algae. https://drs.nio.org/xmlui/handle/2264/2948.
- ↑ 97.0 97.1 97.2 Polglase, Jane L. (1980-11-01). "A Preliminary Report on the Thraustochytrid(s) and Labyrinthulid(s) Associated with a Pathological Condition in the Lesser Octopus Eledone cirrhosa" (in de). Botanica Marina 23 (11): 699–706. doi:10.1515/botm-1980-1106. ISSN 1437-4323. https://www.degruyter.com/document/doi/10.1515/botm-1980-1106/html.
- ↑ McLean, Norman; Porter, David (1982). "The Yellow-Spot Disease of Tritonia diomedea Bergh, 1894 (Mollusca: Gastropoda: Nudibranchia): Encapsulation of the Thraustochytriaceous Parasite by Host Amoebocytes". The Journal of Parasitology 68 (2): 243. doi:10.2307/3281182. ISSN 0022-3395. http://dx.doi.org/10.2307/3281182.
- ↑ 99.0 99.1 Jones, Gwyneth M.; O'Dor, Ronald K. (1983). "Ultrastructural Observations on a Thraustochytrid Fungus Parasitic in the Gills of Squid (Illex illecebrosus LeSueur)". The Journal of Parasitology 69 (5): 903. doi:10.2307/3281055. ISSN 0022-3395. http://dx.doi.org/10.2307/3281055.
- ↑ 100.0 100.1 McLean, Norman; Porter, David (1987). "Lesions produced by a thraustochytrid in Tritonia diomedea (mollusca: Gastropoda: Nudibranchia)" (in en). Journal of Invertebrate Pathology 49 (2): 223–225. doi:10.1016/0022-2011(87)90165-0. https://linkinghub.elsevier.com/retrieve/pii/0022201187901650.
- ↑ 101.0 101.1 Polglase, J.L. (1981). "Thraustochytrids as potential pathogens of marine animals". Bulletin of the British Mycological Society 16: 5.
- ↑ 102.0 102.1 Polglase J.L.; Alderman D.J.; Richards R.H.; Moss S.T. (Ed.) (1986). "Aspects of the progress of mycotic infections in marine animals". The Biology of Marine Fungi, Cambridge University Press Archive: 155–164.
- ↑ 103.0 103.1 Schärer, Lukas; Knoflach, Dagmar; Vizoso, Dita B.; Rieger, Gunde; Peintner, Ursula (2007-09-13). "Thraustochytrids as novel parasitic protists of marine free-living flatworms: Thraustochytrium caudivorum sp. nov. parasitizes Macrostomum lignano" (in en). Marine Biology 152 (5): 1095–1104. doi:10.1007/s00227-007-0755-4. ISSN 0025-3162. Bibcode: 2007MarBi.152.1095S. http://link.springer.com/10.1007/s00227-007-0755-4.
- ↑ 104.0 104.1 Wagner-Merner, Diane TeStrake; Duncan, W. R.; Lawrence, J. M. (1980). "Preliminary Comparison of Thraustochytriaceae in the Guts of a Regular and Irregular Echinoid". Botanica Marina 23 (2). doi:10.1515/botm.1980.23.2.95. ISSN 0006-8055. http://dx.doi.org/10.1515/botm.1980.23.2.95.
- ↑ Raghu-Kumar, S. (1988). "Detection of the thraustochytrid protist Ulkania visurgensis in a hydroid, using immunofluorescence". Marine Biology 97 (2): 253–258. doi:10.1007/bf00391310. ISSN 0025-3162. Bibcode: 1988MarBi..97..253R. http://dx.doi.org/10.1007/bf00391310.
- ↑ Raghukumar, S; Raghukumar, C (1999). "Thraustochytrid fungoid protists in faecal pellets of the tunicate Pegea confoederata, their tolerance to deep-sea conditions and implication in degradation processes". Marine Ecology Progress Series 190: 133–140. doi:10.3354/meps190133. ISSN 0171-8630. Bibcode: 1999MEPS..190..133R.
- ↑ Delille, Daniel; Razouls, Suzanne (1994). "Community structures of heterotrophic bacteria of copepod fecal pellets". Journal of Plankton Research 16 (6): 603–615. doi:10.1093/plankt/16.6.603. ISSN 0142-7873. http://dx.doi.org/10.1093/plankt/16.6.603.
- ↑ Zhukova, NV; Kharlamenko, VI (1999). "Sources of essential fatty acids in the marine microbial loop". Aquatic Microbial Ecology 17: 153–157. doi:10.3354/ame017153. ISSN 0948-3055.
- ↑ Kainz, Martin; Arts, Michael T.; Mazumder, Asit (2004). "Essential fatty acids in the planktonic food web and their ecological role for higher trophic levels". Limnology and Oceanography 49 (5): 1784–1793. doi:10.4319/lo.2004.49.5.1784. ISSN 0024-3590. Bibcode: 2004LimOc..49.1784K. http://dx.doi.org/10.4319/lo.2004.49.5.1784.
- ↑ Fan, K. W.; Vrijmoed, L. L. P.; Jones, E. B. G. (2002-02-25). "Physiological Studies of Subtropical Mangrove Thraustochytrids" (in en). Botanica Marina 45 (1): 50–57. doi:10.1515/BOT.2002.006. https://www.degruyter.com/document/doi/10.1515/BOT.2002.006/html.
- ↑ Jakobsen, Anita N.; Aasen, Inga M.; Josefsen, Kjell D.; Strøm, Arne R. (2008-08-01). "Accumulation of docosahexaenoic acid-rich lipid in thraustochytrid Aurantiochytrium sp. strain T66: effects of N and P starvation and O2 limitation" (in en). Applied Microbiology and Biotechnology 80 (2): 297–306. doi:10.1007/s00253-008-1537-8. ISSN 1432-0614. PMID 18560831. https://doi.org/10.1007/s00253-008-1537-8.
- ↑ Simionato, Diana; Block, Maryse A.; La Rocca, Nicoletta; Jouhet, Juliette; Maréchal, Eric; Finazzi, Giovanni; Morosinotto, Tomas (2013). "The Response of Nannochloropsis gaditana to Nitrogen Starvation Includes De Novo Biosynthesis of Triacylglycerols, a Decrease of Chloroplast Galactolipids, and Reorganization of the Photosynthetic Apparatus" (in en). Eukaryotic Cell 12 (5): 665–676. doi:10.1128/EC.00363-12. ISSN 1535-9778. PMID 23457191.
- ↑ 113.0 113.1 Lippmeier, J. Casey; Crawford, Kristine S.; Owen, Carole B.; Rivas, Angie A.; Metz, James G.; Apt, Kirk E. (2009-06-03). "Characterization of Both Polyunsaturated Fatty Acid Biosynthetic Pathways in Schizochytrium sp.". Lipids 44 (7): 621–630. doi:10.1007/s11745-009-3311-9. ISSN 0024-4201. PMID 19495823. https://doi.org/10.1007/s11745-009-3311-9.
- ↑ Hauvermale, A.; Kuner, J.; Rosenzweig, B.; Guerra, D.; Diltz, S.; Metz, J. G. (2006). "Fatty acid production in Schizochytrium sp.: Involvement of a polyunsaturated fatty acid synthase and a type I fatty acid synthase". Lipids 41 (8): 739–747. doi:10.1007/s11745-006-5025-6. ISSN 0024-4201. PMID 17120926. https://doi.org/10.1007/s11745-006-5025-6.
- ↑ Meesapyodsuk, Dauenpen; Qiu, Xiao (2016-10-01). "Biosynthetic mechanism of very long chain polyunsaturated fatty acids in Thraustochytrium sp. 26185 [S"] (in English). Journal of Lipid Research 57 (10): 1854–1864. doi:10.1194/jlr.M070136. ISSN 0022-2275. PMID 27527703. PMC 5036366. https://www.jlr.org/article/S0022-2275(20)35394-3/abstract.
- ↑ Metz, James G.; Roessler, Paul; Facciotti, Daniel; Levering, Charlene; Dittrich, Franziska; Lassner, Michael; Valentine, Ray; Lardizabal, Kathryn et al. (2001-07-13). "Production of Polyunsaturated Fatty Acids by Polyketide Synthases in Both Prokaryotes and Eukaryotes" (in en). Science 293 (5528): 290–293. doi:10.1126/science.1059593. ISSN 0036-8075. PMID 11452122. https://www.science.org/doi/10.1126/science.1059593.
- ↑ 117.0 117.1 Nelson, David; Cox, Michael (2000). Lehninger Principles of Biochemistry. New York: W.H. Freeman. pp. 836–838.
- ↑ Hayashi, Shohei; Satoh, Yasuharu; Ogasawara, Yasushi; Maruyama, Chitose; Hamano, Yoshimitsu; Ujihara, Tetsuro; Dairi, Tohru (2019-02-18). "Control Mechanism for cis Double‐Bond Formation by Polyunsaturated Fatty-Acid Synthases". Angewandte Chemie International Edition 58 (8): 2326–2330. doi:10.1002/anie.201812623. ISSN 1433-7851. PMID 30623559.
- ↑ Simopoulos, Artemis P. (2008). "The Importance of the Omega-6/Omega-3 Fatty Acid Ratio in Cardiovascular Disease and Other Chronic Diseases" (in en). Experimental Biology and Medicine 233 (6): 674–688. doi:10.3181/0711-MR-311. ISSN 1535-3702. PMID 18408140. http://journals.sagepub.com/doi/10.3181/0711-MR-311.
- ↑ 120.0 120.1 Fan, King Wai; Aki, Tsunehiro; Chen, Feng; Jiang, Yue (2010-07-01). "Enhanced production of squalene in the thraustochytrid Aurantiochytrium mangrovei by medium optimization and treatment with terbinafine" (in en). World Journal of Microbiology and Biotechnology 26 (7): 1303–1309. doi:10.1007/s11274-009-0301-2. ISSN 1573-0972. PMID 24026934. https://doi.org/10.1007/s11274-009-0301-2.
- ↑ Zhang, Aiqing; Xie, Yunxuan; He, Yaodong; Wang, Weijun; Sen, Biswarup; Wang, Guangyi (2019-09-01). "Bio-based squalene production by Aurantiochytrium sp. through optimization of culture conditions, and elucidation of the putative biosynthetic pathway genes" (in en). Bioresource Technology 287: 121415. doi:10.1016/j.biortech.2019.121415. ISSN 0960-8524. PMID 31078814. https://www.sciencedirect.com/science/article/pii/S0960852419306455.
- ↑ Nakazawa, Atsushi; Kokubun, Yume; Matsuura, Hiroshi; Yonezawa, Natsuki; Kose, Ryoji; Yoshida, Masaki; Tanabe, Yuuhiko; Kusuda, Emi et al. (2014-02-01). "TLC screening of thraustochytrid strains for squalene production" (in en). Journal of Applied Phycology 26 (1): 29–41. doi:10.1007/s10811-013-0080-x. ISSN 1573-5176. Bibcode: 2014JAPco..26...29N. https://doi.org/10.1007/s10811-013-0080-x.
- ↑ Li, Qian; Chen, Guan-Qun; Fan, King-Wai; Lu, Fu-Ping; Aki, Tsunehiro; Jiang, Yue (2009-05-27). "Screening and Characterization of Squalene-Producing Thraustochytrids from Hong Kong Mangroves". Journal of Agricultural and Food Chemistry 57 (10): 4267–4272. doi:10.1021/jf9003972. ISSN 0021-8561. PMID 19371138. https://doi.org/10.1021/jf9003972.
- ↑ Suen, Yung Lee; Tang, Hongmei; Huang, Junchao; Chen, Feng (2014-12-24). "Enhanced Production of Fatty Acids and Astaxanthin in Aurantiochytrium sp. by the Expression of Vitreoscilla Hemoglobin". Journal of Agricultural and Food Chemistry 62 (51): 12392–12398. doi:10.1021/jf5048578. ISSN 0021-8561. PMID 25420960. https://doi.org/10.1021/jf5048578.
- ↑ Ye, Jingrun; Liu, Mengmeng; He, Mingxia; Ye, Ying; Huang, Junchao (2019). "Illustrating and Enhancing the Biosynthesis of Astaxanthin and Docosahexaenoic Acid in Aurantiochytrium sp. SK4" (in en). Marine Drugs 17 (1): 45. doi:10.3390/md17010045. ISSN 1660-3397. PMID 30634667.
- ↑ Kobayashi, Takumi; Sakaguchi, Keishi; Matsuda, Takanori; Abe, Eriko; Hama, Yoichiro; Hayashi, Masahiro; Honda, Daiske; Okita, Yuji et al. (2011). "Increase of Eicosapentaenoic Acid in Thraustochytrids through Thraustochytrid Ubiquitin Promoter-Driven Expression of a Fatty Acid Δ5 Desaturase Gene" (in en). Applied and Environmental Microbiology 77 (11): 3870–3876. doi:10.1128/AEM.02664-10. ISSN 0099-2240. PMID 21478316. Bibcode: 2011ApEnM..77.3870K.
- ↑ Matsuda, Takanori; Sakaguchi, Keishi; Hamaguchi, Rie; Kobayashi, Takumi; Abe, Eriko; Hama, Yoichiro; Hayashi, Masahiro; Honda, Daiske et al. (2012-06-01). "Analysis of Δ12-fatty acid desaturase function revealed that two distinct pathways are active for the synthesis of PUFAs in T. aureum ATCC 34304" (in English). Journal of Lipid Research 53 (6): 1210–1222. doi:10.1194/jlr.M024935. ISSN 0022-2275. PMID 22368282. PMC 3351828. https://www.jlr.org/article/S0022-2275(20)31714-4/abstract.
- ↑ Li, Zhipeng; Meng, Tong; Ling, Xueping; Li, Jun; Zheng, Chuqiang; Shi, Yanyan; Chen, Zhen; Li, Zhenqi et al. (2018-05-30). "Overexpression of Malonyl-CoA: ACP Transacylase in Schizochytrium sp. to Improve Polyunsaturated Fatty Acid Production". Journal of Agricultural and Food Chemistry 66 (21): 5382–5391. doi:10.1021/acs.jafc.8b01026. ISSN 0021-8561. PMID 29722541. https://doi.org/10.1021/acs.jafc.8b01026.
- ↑ Huang, Ting Yen; Lu, Wen Chang; Chu, I. Ming (2012-11-01). "A fermentation strategy for producing docosahexaenoic acid in Aurantiochytrium limacinum SR21 and increasing C22:6 proportions in total fatty acid" (in en). Bioresource Technology 123: 8–14. doi:10.1016/j.biortech.2012.07.068. ISSN 0960-8524. PMID 22929740. https://www.sciencedirect.com/science/article/pii/S0960852412011157.
- ↑ YAMAOKA, Yukiho; CARMONA, Marvelisa L; OOTA, Shinji (2004-01-01). "Growth and Carotenoid Production of Thraustochytrium sp. CHN-1 Cultured under Superbright Red and Blue Light-emitting Diodes". Bioscience, Biotechnology, and Biochemistry 68 (7): 1594–1597. doi:10.1271/bbb.68.1594. ISSN 0916-8451. PMID 15277770.
- ↑ Chatdumrong, Wassana; Yongmanitchai, Wichien; Limtong, Savitree; Worawattanamateekul, Wanchai. "Optimization of Docosahexaenoic Acid (DHA) Production and Improvement of Astaxanthin Content in a Mutant Schizochytrium limacinum Isolated from Mangrove Forest in Thailand". Kasetsart Journal - Natural Science 41: 324–334. https://www.researchgate.net/publication/266468310.
- ↑ Romari, Khadidja; Adeline Le Monnier & Cyrilinventor4=Cindy Merlet;Julien Pagliardini;Pierre Calleja;Claude Gudin Rols, "Production of astaxanthin and docosahexaenoic acid in mixotrophic mode using Schizochytrium", US patent 9506100, published 2016-11-29, assigned to Fermentalg
- ↑ Yue, Cai-Jun; Jiang, Yue (2009-08-01). "Impact of methyl jasmonate on squalene biosynthesis in microalga Schizochytrium mangrovei" (in en). Process Biochemistry 44 (8): 923–927. doi:10.1016/j.procbio.2009.03.016. ISSN 1359-5113. https://www.sciencedirect.com/science/article/pii/S1359511309000956.
- ↑ Singh, Dilip; Barrow, Colin J.; Puri, Munish; Tuli, Deepak K.; Mathur, Anshu S. (2016-04-01). "Combination of calcium and magnesium ions prevents substrate inhibition and promotes biomass and lipid production in thraustochytrids under higher glycerol concentration" (in en). Algal Research 15: 202–209. doi:10.1016/j.algal.2016.02.024. ISSN 2211-9264. https://www.sciencedirect.com/science/article/pii/S2211926416300637.
- ↑ Kanchana, R.; Muraleedharan, Usha Devi; Raghukumar, Seshagiri (2011-09-01). "Alkaline lipase activity from the marine protists, thraustochytrids" (in en). World Journal of Microbiology and Biotechnology 27 (9): 2125–2131. doi:10.1007/s11274-011-0676-8. ISSN 1573-0972. https://doi.org/10.1007/s11274-011-0676-8.
- ↑ Bayne, Anne-Cécile V.; Boltz, David; Owen, Carole; Betz, Yelena; Maia, Goncalo; Azadi, Parastoo; Archer-Hartmann, Stephanie; Zirkle, Ross et al. (2013-04-23). "Vaccination against Influenza with Recombinant Hemagglutinin Expressed by Schizochytrium sp. Confers Protective Immunity" (in en). PLOS ONE 8 (4): e61790. doi:10.1371/journal.pone.0061790. ISSN 1932-6203. PMID 23626728. Bibcode: 2013PLoSO...861790B.
- ↑ Raghukumar, Seshagiri; Hajib Naraharirao Madhavan & Jambulimgam Malathi, "Extracellular polysaccharides from labyrinthulomycetes with broad-spectrum antiviral activities", WO patent application 2014045191, published 2014-07-17, assigned to Myko Tech Pvt. Ltd.
- ↑ Bagul, Vaishali P.; Annapure, Uday S. (2021-07-15). "Isolation of fast-growing thraustochytrids and seasonal variation on the fatty acid composition of thraustochytrids from mangrove regions of Navi Mumbai, India" (in en). Journal of Environmental Management 290: 112597. doi:10.1016/j.jenvman.2021.112597. ISSN 0301-4797. PMID 33878627. https://www.sciencedirect.com/science/article/pii/S0301479721006599.
- ↑ Ansorena, D.; Astiasarán, I. (2013-01-01), Domínguez, Herminia, ed., "20 - Development of nutraceuticals containing marine algae oils" (in en), Functional Ingredients from Algae for Foods and Nutraceuticals, Woodhead Publishing Series in Food Science, Technology and Nutrition (Woodhead Publishing): pp. 634–657, doi:10.1533/9780857098689.4.634, ISBN 978-0-85709-512-1, https://www.sciencedirect.com/science/article/pii/B9780857095121500205, retrieved 2022-03-17
- ↑ Woods, Vanessa B.; Fearon, Anna M. (2009-12-01). "Dietary sources of unsaturated fatty acids for animals and their transfer into meat, milk and eggs: A review" (in en). Livestock Science 126 (1): 1–20. doi:10.1016/j.livsci.2009.07.002. ISSN 1871-1413. https://www.sciencedirect.com/science/article/pii/S1871141309002674.
- ↑ Valencia, Idoia; Ansorena, Diana; Astiasarán, Iciar (2007-01-01). "Development of dry fermented sausages rich in docosahexaenoic acid with oil from the microalgae Schizochytrium sp.: Influence on nutritional properties, sensorial quality and oxidation stability" (in en). Food Chemistry 104 (3): 1087–1096. doi:10.1016/j.foodchem.2007.01.021. ISSN 0308-8146. https://www.sciencedirect.com/science/article/pii/S0308814607000763.
- ↑ Lonza Ltd. (January 6, 2010). "GRAS Notice for Ulkenla DHA Oil Derived from Ulkenia sp. Microalga". GRAS Notice (GRN) - FDA: 1–166. http://wayback.archive-it.org/7993/20171031044215/https://www.fda.gov/downloads/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/UCM269492.pdf.
- ↑ NutraSource, Inc.. "DETERMINATION OF THE GENERALLY RECOGNIZED AS SAFE (GRAS) STATUS OF DOCOSAHEXAENOIC ACID-RICH OIL AS A FOOD INGREDIENT FOR INFANT FORMULA APPLICATIONS". GRAS Notice (GRN) - FDA: 1–48. https://www.fda.gov/media/113396/download.
- ↑ "FAO.org". https://www.fao.org/faolex/results/details/en/c/LEX-FAOC038101.
- ↑ Martek Biosciences Corporation (2010). "Application for the authorization of DHA and EPA-rich Algal Oil from Schizochytrium sp.". Regulation (EC) 258/97: 1–63. https://acnfp.food.gov.uk/sites/default/files/mnt/drupal_data/sources/files/multimedia/pdfs/dhaoapplicdossier.pdf.
- ↑ Carter, C.G.; Bransden, M.P.; Lewis, T.E.; Nichols, P.D. (2003-10-01). "Potential of Thraustochytrids to Partially Replace FishOil in Atlantic Salmon Feeds" (in en). Marine Biotechnology 5 (5): 480–492. doi:10.1007/s10126-002-0096-8. ISSN 1436-2236. PMID 14730431. Bibcode: 2003MarBt...5..480C. https://doi.org/10.1007/s10126-002-0096-8.
- ↑ García-Ortega, Armando; Kissinger, Karma R.; Trushenski, Jesse T. (2016-02-01). "Evaluation of fish meal and fish oil replacement by soybean protein and algal meal from Schizochytrium limacinum in diets for giant grouper Epinephelus lanceolatus" (in en). Aquaculture 452: 1–8. doi:10.1016/j.aquaculture.2015.10.020. ISSN 0044-8486. https://www.sciencedirect.com/science/article/pii/S004484861530209X.
- ↑ Kissinger, Karma R.; García-Ortega, Armando; Trushenski, Jesse T. (2016-02-01). "Partial fish meal replacement by soy protein concentrate, squid and algal meals in low fish-oil diets containing Schizochytrium limacinum for longfin yellowtail Seriola rivoliana" (in en). Aquaculture 452: 37–44. doi:10.1016/j.aquaculture.2015.10.022. ISSN 0044-8486. https://www.sciencedirect.com/science/article/pii/S0044848615302118.
- ↑ Kousoulaki, K.; Mørkøre, T.; Nengas, I.; Berge, R. K.; Sweetman, J. (2016-01-20). "Microalgae and organic minerals enhance lipid retention efficiency and fillet quality in Atlantic salmon (Salmo salar L.)" (in en). Aquaculture 451: 47–57. doi:10.1016/j.aquaculture.2015.08.027. ISSN 0044-8486. https://www.sciencedirect.com/science/article/pii/S0044848615301460.
- ↑ Sprague, M.; Walton, J.; Campbell, P. J.; Strachan, F.; Dick, J. R.; Bell, J. G. (2015-10-15). "Replacement of fish oil with a DHA-rich algal meal derived from Schizochytrium sp. on the fatty acid and persistent organic pollutant levels in diets and flesh of Atlantic salmon (Salmo salar, L.) post-smolts" (in en). Food Chemistry 185: 413–421. doi:10.1016/j.foodchem.2015.03.150. ISSN 0308-8146. PMID 25952887. https://www.sciencedirect.com/science/article/pii/S0308814615005440.
- ↑ Faukner, Jimmy; Rawles, Steven D.; Proctor, Andrew; Sink, Todd D.; Chen, Ruguang; Philips, Harold; Lochmann, Rebecca T. (2013). "The Effects of Diets Containing Standard Soybean Oil, Soybean Oil Enhanced with Conjugated Linoleic Acids, Menhaden Fish Oil, or an Algal Docosahexaenoic Acid Supplement on Channel Catfish Performance, Body Composition, Sensory Evaluation, and Storage Characteristics". North American Journal of Aquaculture 75 (2): 252–265. doi:10.1080/15222055.2012.713896. ISSN 1522-2055. Bibcode: 2013NAJA...75..252F. https://doi.org/10.1080/15222055.2012.713896.
- ↑ Miller, Matthew R.; Nichols, Peter D.; Carter, Chris G. (2007-10-01). "Replacement of fish oil with thraustochytrid Schizochytrium sp. L oil in Atlantic salmon parr (Salmo salar L) diets" (in en). Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 148 (2): 382–392. doi:10.1016/j.cbpa.2007.05.018. ISSN 1095-6433. PMID 17588797. https://www.sciencedirect.com/science/article/pii/S1095643307010252.
- ↑ Barclay, William; Zeller, Sam (1996). "Nutritional Enhancement of n-3 and n-6 Fatty Acids in Rotifers and Artemia Nauplii by Feeding Spray-dried Schizochytrium sp.". Journal of the World Aquaculture Society 27 (3): 314–322. doi:10.1111/j.1749-7345.1996.tb00614.x. ISSN 0893-8849. https://doi.org/10.1111/j.1749-7345.1996.tb00614.x.
- ↑ Park, Heum Gi; Puvanendran, Velmurugu; Kellett, Anne; Parrish, Christopher C.; Brown, Joseph A. (2006-01-01). "Effect of enriched rotifers on growth, survival, and composition of larval Atlantic cod (Gadus morhua)". ICES Journal of Marine Science 63 (2): 285–295. doi:10.1016/j.icesjms.2005.10.011. ISSN 1054-3139. Bibcode: 2006ICJMS..63..285P.
- ↑ Takeuchi, Toshio (2014-05-01). "Progress on larval and juvenile nutrition to improve the quality and health of seawater fish: a review" (in en). Fisheries Science 80 (3): 389–403. doi:10.1007/s12562-014-0744-8. ISSN 1444-2906. Bibcode: 2014FisSc..80..389T.
- ↑ 156.0 156.1 Merkx-Jacques, Alexandra; Rasmussen, Holly; Muise, Denise M.; Benjamin, Jeremy J. R.; Kottwitz, Haila; Tanner, Kaitlyn; Milway, Michael T.; Purdue, Laura M. et al. (2018-09-17). "Engineering xylose metabolism in thraustochytrid T18". Biotechnology for Biofuels 11 (1): 248. doi:10.1186/s13068-018-1246-1. ISSN 1754-6834. PMID 30237825.
- ↑ Arbenz, Alice; Stephanie Laurichesse & Remi Perrin et al., "Method for manufacturing a polyurethane-modified foam, foam obtained, and uses", EP patent 3275912, published 2019-01-02, assigned to Soprema (SAS)and Centre National de la Recherche Scientifique & Universite de Strasbourg
Wikidata ☰ Q25365893 entry
 |