Chemistry:Sodium metaborate
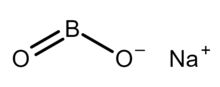 Sodium metaborate monomer
| |
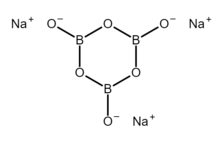 Sodium metaborate trimer
| |
| Names | |
|---|---|
| IUPAC name
Sodium metaborate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| NaBO 2 | |
| Molar mass | 65.80 g·mol−1 |
| Appearance | Colorless crystals |
| Odor | Odorless |
| Density | 2.464 g/cm3 (anhydrous)[1] |
| Melting point | 966 °C (1,771 °F; 1,239 K) |
| Boiling point | 1,434[2] °C (2,613 °F; 1,707 K) |
| 16.4 g/(100 mL) (0 °C) 28.2 g/(100 mL) (25 °C) 125.2 g/(100 mL) (100 °C) | |
| Solubility | insoluble in ether, ethanol |
| Structure | |
| trigonal | |
| Thermochemistry | |
Heat capacity (C)
|
65.94 J/(mol·K) |
Std molar
entropy (S |
73.39 J/(mol·K) |
Std enthalpy of
formation (ΔfH⦵298) |
−1059 kJ/mol |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
2330 mg/kg (rat, oral) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Sodium metaborate is a chemical compound of sodium, boron, and oxygen with formula NaBO
2.[3] However, the metaborate ion is trimeric in the anhydrous solid, therefore a more correct formula is Na
3B
3O
6 or (Na+
)
3[B
3O
6]3−. The formula can be written also as Na
2O · B
2O
3 to highlight the relation to the main oxides of sodium and boron.[2] The name is also applied to several hydrates whose formulas can be written NaBO
2 · nH
2O for various values of n.
The anhydrous and hydrates are colorless crystalline solids. The anhydrous form is hygroscopic.[4]
Hydrates and solubility
The following hydrates crystallize from solutions of the proper composition in various temperature ranges:[5]
- tetrahydrate NaBO
2 · 4H2O from −6 to 53.6 °C - dihydrate NaBO
2 · 2H2O from 53.6 °C to 105 °C - hemihydrate NaBO
2 · 0.5H2O from 105 °C to the boiling point.
Early reports of a monohydrate NaBO
2 · H2O have not been confirmed.[5]
Structure
Anhydrous
Solid anhydrous sodium metaborate has the hexagonal crystal system with space group . It actually contains a six-membered rings with the formula [B
3O
6]3−, consisting of alternating boron and oxygen atoms with one negatively charged extra oxygen atom attached to each boron atom.[6][1] All nine atoms lie on a plane.[4] The six oxygen atoms are evenly divided into two distinct structural sites, with different B–O bond lengths: B–O(external) 128.0 pm and B–O(bridge) 143.3 pm.[6] The density is 2.348 ± 0.005 g/cm3.[4] The approximate dimensions of the hexagonal cell are a = 1275 pm, c = 733 pm.[4] However, the true unit cell is rhombohedral and has dimensions: ar= 776 pm, α = 110.6°, Z = 6 (5.98) molecules KB0[4]
Dihydrate
The dihydrate NaBO
2 · 2H2O crystallizes in the triclinic crystal system, but is nearly monoclinic, with both α and γ very close to 90°. The cell parameters are a = 678 pm , b = 1058A pm, c = 588 pm, α = 91.5°, β = 22.5°, γ = 89°, Z = 4, density 1.905 g/cm3. The refractive indices at 25°C and wavelength 589.3 nm are α = 1.439, β = 1.473, γ = 1.484. The dispersion is strong, greater at red than at violet.[7]
The transition temperature between the dihydrate and the hemihydrate is 54 °C. However, the crystalline dihydrate will remain metastable until 106 °C to 110 °C, and change slowly above that temperature.[7]
Vapor
Infrared spectroscopy of the vapor from anhydrous sodium metaborate, heated to between 900 °C and 1400 °C, shows mostly isolated clusters with formula NaBO
2, and some dimers thereof.[8] Electron diffraction studies by Akishin and Spiridonov showed a structure O=B–O–Na with linear anion O=B–O−
and angle B–O–Na of 90-110°. The atomic distances are O=B: 120 pm, B–O: 136 pm,O–Na: 214 pm[9]
Preparation
Sodium metaborate is prepared by the fusion of sodium carbonate and boron oxide B
2O
3[1][4] or borax Na
2B
4O
7. Another way to create the compound is by the fusion of borax with sodium hydroxide at 700 °C:
- B
2O
3 + 2 NaOH → 2 NaBO
2 + H
2O
The boiling point of sodium metaborate (1434 °C) is lower than that of boron oxide (1860 °C) and borax (1575 °C) In fact, while the metaborate boils without change of composition, borax gives off a vapor of sodium metaborate with a small excess of sodium oxide Na
2O.[2]
The anhydrous salt can also be prepared from the tetraborate by heating to 270 °C in vacuum.[10]
Although not performed industrially, hydrolysis of sodium borohydride Na[BH
4] with a suitable catalyst gives sodium metaborate and hydrogen gas:[11]
- Na[BH
4] + 2 H
2O → NaBO
2 + 4 H
2 (ΔH = −217 kJ/mol)
Reactions
With water
When sodium metaborate is dissolved in water, the anion combines with two water molecules to form the tetrahydroxyborate anion [B(OH)
4]−
.[11]
Electrochemical conversion to borax
Electrolysis of a concentrated aqueous solution of 20%[clarification needed] NaBO
2 · 4H2O with an anion exchange membrane and inert anode (such as gold, palladium, or boron-doped diamond) converts the metaborate anion to tetraborate B
4O2−
7, and the sodium salt of the later (borax) precipitates as a white powder.[12]
- 8BO2−
2 → 2B
4O2−
7 + O
2 + 4 e−
Reduction to sodium borohydride
Sodium metaborate can be converted to sodium borohydride by several methods, including the reaction with various reducing agents at high temperatures and pressure,[12] or with magnesium hydride MgH
2 by ball milling at room temperature, followed by extraction of the Na[BH
4] with isopropylamine.[13][10]
- NaBO
2 + 2 MgH
2 → Na[BH
4] + 2 MgO
Another method is the electrolytic reduction of a concentrated sodium metaborate solution,[10] namely
- BO−
2 + 6 H
2O + 8 e−
→ [BH
4]−
+ 8 OH−
However, this method is not efficient since it competes with the reduction of hydroxide:
- 4 OH−
→ 2 H
2O + O
2 + 4 e−
Nanofiltration membranes can effectively separate the borohydride from the metaborate.[11]
Reaction with alcohols
Anhydrous sodium metaborate refluxed with methanol yields the corresponding sodium tetramethoxyborate (melting point: 253-258 °C, CAS number: 18024-69-6[14]):[15]
- Na+
BO−
2 + 4 CH
3OH → Na+
[B(OCH
3)
4]−
+ 2 H
2O
The analogous reaction with ethanol yields the sodium tetraethoxyborate.[15]
Uses
Current and proposed applications of sodium metaborate include:
- Manufacture of borosilicate glasses, which are resistant to uneven or fast heating because of their small coefficient of thermal expansion.
- Composition of herbicides.[16]
- Raising the pH of injected fluids for oil extraction.[17]
See also
References
- ↑ 1.0 1.1 1.2 Fang, Ssu-Mien (1938). "The Crystal Structure of Sodium Metaborate Na3 (B3 O6)". Zeitschrift für Kristallographie - Crystalline Materials 99 (1–6): 1–8. doi:10.1524/zkri.1938.99.1.1.
- ↑ 2.0 2.1 2.2 Cole, Sandford S.; Taylor, Nelson W. (1935). "THE SYSTEM Na2O-B2O3, IV Vapor Pressures of Boric Oxide, Sodium Metaborate, and Sodium Diborate between 1150°C and 1400°C". Journal of the American Ceramic Society 18 (1–12): 82–85. doi:10.1111/j.1151-2916.1935.tb19358.x.
- ↑ "Sodium metaborate" Substance page at the Chemister website. Accessed on 2022-06-28.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 Zachariasen, W. H. (1937). "The Crystal Structure of Potassium Metaborate, K3(B3O6)". The Journal of Chemical Physics 5 (11): 919–922. doi:10.1063/1.1749962.
- ↑ 5.0 5.1 Nies, Nelson P.; Hulbert, Richard W. (1967). "Solubility isotherms in the system sodium oxide-boric oxide-water. Revised solubility-temperature curves of boric acid, borax, sodium pentaborate, and sodium metaborate". Journal of Chemical & Engineering Data 12 (3): 303–313. doi:10.1021/je60034a005.
- ↑ 6.0 6.1 Marezio, M.; Plettinger, H. A.; Zachariasen, W. H. (1963). "The bond lengths in the sodium metaborate structure". Acta Crystallographica 16 (7): 594–595. doi:10.1107/S0365110X63001596. Bibcode: 1963AcCry..16..594M.
- ↑ 7.0 7.1 Krc, John (1951). "Crystallographic Data. 44. Sodium Metaborate Dihydrate". Analytical Chemistry 23 (5): 806. doi:10.1021/ac60053a043.
- ↑ Alfred Büchler and Edward P. Marram (1963). "Gaseous Metaborates. II. Infrared Spectraof Alkali Metaborate Vapors". Journal of Chemical Physics 39: 292. doi:10.1063/1.173424439.
- ↑ Akishin, P. A.; Spiridonov, V. P. (1962). "Electron diffraction study of the structure of LiBO2 and NaBO2 metaborate molecules in the vapor state". Journal of Structural Chemistry 3 (3): 251–253. doi:10.1007/BF01151477.
- ↑ 10.0 10.1 10.2 Kong, Lingyan; Cui, Xinyu; Jin, Huazi; Wu, Jie; Du, Hao; Xiong, Tianying (2009). "Mechanochemical Synthesis of Sodium Borohydride by Recycling Sodium Metaborate". Energy & Fuels 23 (10): 5049–5054. doi:10.1021/ef900619y.
- ↑ 11.0 11.1 11.2 Atiyeh, H.; Davis, B. (2007). "Separation of sodium metaborate from sodium borohydride using nanofiltration membranes for hydrogen storage application". International Journal of Hydrogen Energy 32 (2): 229–236. doi:10.1016/j.ijhydene.2006.06.003. Bibcode: 2007IJHE...32..229A.
- ↑ 12.0 12.1 Park, E.; Ukjeong, S.; Hojung, U.; Kim, S.; Lee, J.; Woonam, S.; Hoonlim, T.; Junpark, Y. et al. (2007). "Recycling of sodium metaborate to borax". International Journal of Hydrogen Energy 32 (14): 2982–2987. doi:10.1016/j.ijhydene.2007.03.029. Bibcode: 2007IJHE...32.2982P.
- ↑ Li, Z.P.; Liu, B.H.; Arai, K.; Morigazaki, N.; Suda, S. (2003). "Protide compounds in hydrogen storage systems". Journal of Alloys and Compounds 356-357: 469–474. doi:10.1016/S0925-8388(02)01241-0.
- ↑ "Product". https://www.sigmaaldrich.com/GB/en/product/enamine/enah65927943?context=bbe.
- ↑ 15.0 15.1 Kemmitt, T.; Gainsford, G.J. (2009). "Regeneration of sodium borohydride from sodium metaborate, and isolation of intermediate compounds". International Journal of Hydrogen Energy 34 (14): 5726–5731. doi:10.1016/j.ijhydene.2009.05.108. Bibcode: 2009IJHE...34.5726K.
- ↑ "BareSpot Monobor-Chlorate". Product safety data sheet at the BareSpot company website.Retrieved 2022-06-28.
- ↑ Chen, Fuzhen; Jiang, Hanqiao; Bai, Xiaohu; Zheng, Wei (2013). "Evaluation the performance of sodium metaborate as a novel alkali in alkali/Surfactant/Polymer flooding". Journal of Industrial and Engineering Chemistry 19 (2): 450–457. doi:10.1016/j.jiec.2012.08.029.
 |
