Chemistry:7-Dehydrocholesterol
| |
| |
| Names | |
|---|---|
| IUPAC name
Cholesta-5,7-dien-3β-ol
| |
| Systematic IUPAC name
(1R,3aR,7S,9aR,9bS,11aR)-9a,11a-Dimethyl-1-[(2R)-6-methylheptan-2-yl]-2,3,3a,6,7,8,9,9a,9b,10,11,11a-dodecahydro-1H-cyclopenta[a]phenanthren-7-ol | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| MeSH | 7-dehydrocholesterol |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C27H44O | |
| Molar mass | 384.638 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
7-Dehydrocholesterol (7-DHC) is a zoosterol that functions in the serum as a cholesterol precursor, and is photochemically converted to vitamin D3 in the skin, therefore functioning as provitamin-D3. The presence of this compound in human skin enables humans to manufacture vitamin D3 (cholecalciferol). Upon exposure to ultraviolet UV-B rays in the sun light, 7-DHC is converted into vitamin D3 via previtamin D3 as an intermediate isomer. It is also found in the milk of several mammalian species.[1][2] Lanolin, a waxy substance that is naturally secreted by wool-bearing mammals, contains 7-DHC which is converted into vitamin D by sunlight and then ingested during grooming as a nutrient. In insects 7-dehydrocholesterol is a precursor for the hormone ecdysone, required for reaching adulthood.[3] It was discovered by Nobel-laureate organic chemist Adolf Windaus.
Biosynthesis
It is synthesized from lathosterol by the enzyme lathosterol oxidase (lathosterol 5-desaturase). This is the next-to-last step of cholesterol biosynthesis.[4] Defective synthesis results in the human inherited disorder lathosterolosis resembling Smith–Lemli–Opitz syndrome.[4] Mice where this gene has been deleted lose the ability to increase vitamin D3 in the blood following UV exposure of the skin.[5]
Location
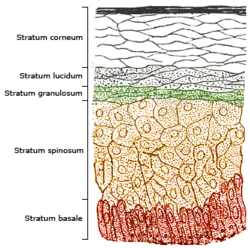
The skin consists of two primary layers: an inner layer, the dermis, comprising largely connective tissue, and an outer, thinner epidermis. The thickness of the epidermis ranges from 0.08 mm to greater than 0.6 mm (from 0.003 to 0.024 inches).[6] The epidermis comprises five strata; from outer to inner, they are the stratum corneum, stratum lucidum, stratum granulosum, stratum spinosum, and stratum basale. The highest concentrations of 7-dehydrocholesterol are found in the epidermal layer of skin—specifically in the stratum basale and stratum spinosum.[7] The production of pre-vitamin D3 is, therefore, greatest in these two layers.
Radiation
Synthesis of pre-vitamin D3 in the skin involves UVB radiation, which effectively penetrates only the epidermal layers of skin. 7-Dehydrocholesterol absorbs UV light most effectively at wavelengths between 295 and 300 nm and, thus, the production of vitamin D3 will occur primarily at those wavelengths.[8] The two most important factors that govern the generation of pre-vitamin D3 are the quantity (intensity) and quality (appropriate wavelength) of the UVB irradiation reaching the 7-dehydrocholesterol deep in the stratum basale and stratum spinosum.[7] Light-emitting diodes (LEDs) can be used to produce the radiation.[9]
Another important consideration is the quantity of 7-dehydrocholesterol present in the skin. Under normal circumstances, ample quantities of 7-dehydrocholesterol (about 25–50 μg/cm2 of skin) are available in the stratum spinosum and stratum basale of human skin to meet the body's vitamin D requirements. 7-DHC insufficiency has been proposed as an alternate cause for Vitamin D deficiency.[10]
Sources
7-DHC can be produced by animals and plants via different pathways. It is not produced by fungi in significant amounts. It is made by some algae, but the pathway is poorly understood.[11] It can also be produced by some bacteria.[10]
Industrially, 7-DHC generally comes from lanolin, and is used to produce vitamin D3 by UV exposure.[12] Lichen (Cladonia rangiferina) is used to produce vegan D3.[13][14]
7-DHC is used for vitamin D3 synthesis via lanosterol in land animals, via cycloartenol in plants, and in algae together with another provitamin D ergosterol for D2. In fungi solely ergosterol is used for synthesis of D2 via lanosterol.[15]
Interactive pathway map
See also
References
- ↑ "7-dehydrocholesterol". The American Heritage Stedman's Medical Dictionary. Houghton Mifflin Company. 21 January 2007.
- ↑ "7-dehydrocholesterol". Answers.com. http://www.answers.com/topic/7-dehydrocholesterol.
- ↑ "Thanks to one gene, this fly needs a cactus to escape Neverland". Not Exactly Rocket Science. 2012. http://blogs.discovermagazine.com/notrocketscience/2012/09/28/fly-cactus-neverland-gene/.
- ↑ 4.0 4.1 "Lathosterolosis: an inborn error of human and murine cholesterol synthesis due to lathosterol 5-desaturase deficiency". Human Molecular Genetics 12 (13): 1631–41. July 2003. doi:10.1093/hmg/ddg172. PMID 12812989.
- ↑ "Attenuation of UVR-induced vitamin D3 synthesis in a mouse model deleted for keratinocyte lathosterol 5-desaturase". The Journal of Steroid Biochemistry and Molecular Biology 171: 187–194. July 2017. doi:10.1016/j.jsbmb.2017.03.017. PMID 28330720. https://research.birmingham.ac.uk/portal/en/publications/attenuation-of-uvrinduced-vitamin-d3-synthesis-in-a-mouse-model-deleted-for-keratinocyte-lathosterol-5desaturase(11f96b2b-f9ff-4ea7-b10d-25547481754c).html.
- ↑ Human Anatomy. Pearson/Benjamin-Cummings Publishers. 2006. p. 89. ISBN 0-8053-7211-3.
- ↑ 7.0 7.1 "Sunlight, season, skin pigmentation, vitamin D, and 25-hydroxyvitamin D: integral components of the vitamin D endocrine system". The American Journal of Clinical Nutrition 67 (6): 1108–1110. June 1998. doi:10.1093/ajcn/67.6.1108. PMID 9625080.
- ↑ "Spectral character of sunlight modulates photosynthesis of previtamin D3 and its photoisomers in human skin". Science 216 (4549): 1001–3. May 1982. doi:10.1126/science.6281884. PMID 6281884.
- ↑ "Ultraviolet B Light Emitting Diodes (LEDs) Are More Efficient and Effective in Producing Vitamin D3 in Human Skin Compared to Natural Sunlight". Scientific Reports 7 (1): 11489. September 2017. doi:10.1038/s41598-017-11362-2. PMID 28904394.
- ↑ 10.0 10.1 "Provitamin D3 modulation through prebiotics supplementation: simulation based assessment". Scientific Reports 9 (1): 19267. December 2019. doi:10.1038/s41598-019-55699-2. PMID 31848400. Bibcode: 2019NatSR...919267G.
- ↑ "Vitamin D in plants: a review of occurrence, analysis, and biosynthesis". Frontiers in Plant Science 4: 136. 2013. doi:10.3389/fpls.2013.00136. PMID 23717318.
- ↑ "The vitamin D epidemic and its health consequences". The Journal of Nutrition 135 (11): 2739S–2748S. November 2005. doi:10.1093/jn/135.11.2739S. PMID 16251641. "[Vitamin D3] is produced commercially by extracting 7-dehydrocholesterol from wool fat, followed by UVB irradiation and purification [...] [Vitamin D2] is commercially made by irradiating and then purifying the ergosterol extracted from yeast".
- ↑ "Vitamin D" (in en). https://www.vegansociety.com/resources/nutrition-and-health/nutrients/vitamin-d.
- ↑ Gangwar, Gourvendra (1 July 2023). "Formulation of Lichen Based Pill a Natural Source of Vitamin D3 with a High Absorption Rate by Ambrosiya Neo-Medicine Pvt. Ltd". International Journal of Biomedical Investigation: 1. https://openj.edwiserinternational.com/index.php/ijbi/article/download/102/101.
- ↑ Göring, Horst (November 2018). "Vitamin D in Nature: A Product of Synthesis and/or Degradation of Cell Membrane Components". Biokhimiya (Moscow) 83 (11): 1350–1357. doi:10.1134/S0006297918110056. PMID 30482146. https://www.researchgate.net/publication/328961025. Retrieved December 2, 2023.
 |
