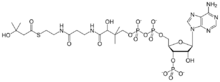
Chemistry:Beta-Hydroxy beta-methylbutyryl-CoA
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
3′-O-Phosphonoadenosine 5′-[(3R)-3-hydroxy-4-{[3-({2-[(3-hydroxy-3-methylbutanoyl)sulfanyl]ethyl}amino)-3-oxopropyl]amino}-2,2-dimethyl-4-oxobutyl dihydrogen diphosphate]
| |
| Systematic IUPAC name
O1-{[(2R,3S,4R,5R)-5-(6-Amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)oxolan-2-yl]methyl} O3-[(3R)-3-hydroxy-4-{[3-({2-[(3-hydroxy-3-methylbutanoyl)sulfanyl]ethyl}amino)-3-oxopropyl]amino}-2,2-dimethyl-4-oxobutyl] dihydrogen diphosphate | |
| Other names
β-hydroxyisovaleryl-CoA
3-hydroxyisovaleryl-CoA 3-hydroxy-3-methylbutyryl-CoA | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| |
| |
| Properties | |
| C26H44N7O18P3S | |
| Molar mass | 867.649946 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
β-Hydroxy β-methylbutyryl-coenzyme A (HMB-CoA), also known as 3-hydroxyisovaleryl-CoA, is a metabolite of L-leucine that is produced in the human body.[1][2] Its immediate precursors are β-hydroxy β-methylbutyric acid (HMB) and β-methylcrotonoyl-CoA (MC-CoA). It can be metabolized into HMB, MC-CoA, and HMG-CoA in humans.
Metabolic pathway
Notes
- ↑ This reaction is catalyzed by an unknown thioesterase enzyme.[3][4]
References
- ↑ 1.0 1.1 1.2 "International Society of Sports Nutrition Position Stand: beta-hydroxy-beta-methylbutyrate (HMB)". Journal of the International Society of Sports Nutrition 10 (1): 6. February 2013. doi:10.1186/1550-2783-10-6. PMID 23374455.
- ↑ 2.0 2.1 2.2 Nutrient Metabolism: Structures, Functions, and Genes (2nd ed.). Academic Press. May 2015. pp. 385–388. ISBN 978-0-12-387784-0. https://books.google.com/books?id=aTQTAAAAQBAJ&printsec=frontcover#v=onepage. Retrieved 6 June 2016. "Energy fuel: Eventually, most Leu is broken down, providing about 6.0kcal/g. About 60% of ingested Leu is oxidized within a few hours ... Ketogenesis: A significant proportion (40% of an ingested dose) is converted into acetyl-CoA and thereby contributes to the synthesis of ketones, steroids, fatty acids, and other compounds"
Figure 8.57: Metabolism of L-leucine - ↑ "KEGG Reaction: R10759". Kanehisa Laboratories. http://www.genome.jp/dbget-bin/www_bget?rn:R10759. Retrieved 24 June 2016.
- ↑ "Urinary excretion of 3-hydroxyisovaleric acid and 3-hydroxyisovaleryl carnitine increases in response to a leucine challenge in marginally biotin-deficient humans". The Journal of Nutrition 141 (11): 1925–1930. November 2011. doi:10.3945/jn.111.146126. PMID 21918059. "Metabolic impairment diverts methylcrotonyl CoA to 3-hydroxyisovaleryl CoA in a reaction catalyzed by enoyl-CoA hydratase (22, 23). 3-Hydroxyisovaleryl CoA accumulation can inhibit cellular respiration either directly or via effects on the ratios of acyl CoA:free CoA if further metabolism and detoxification of 3-hydroxyisovaleryl CoA does not occur (22). The transfer to carnitine by 4 carnitine acyl-CoA transferases distributed in subcellular compartments likely serves as an important reservoir for acyl moieties (39–41). 3-Hydroxyisovaleryl CoA is likely detoxified by carnitine acetyltransferase producing 3HIA-carnitine, which is transported across the inner mitochondrial membrane (and hence effectively out of the mitochondria) via carnitine-acylcarnitine translocase (39). 3HIA-carnitine is thought to be either directly deacylated by a hydrolase to 3HIA or to undergo a second CoA exchange to again form 3-hydroxyisovaleryl CoA followed by release of 3HIA and free CoA by a thioesterase.".