Chemistry:Dihydrofolic acid
From HandWiki
| |
| |
| Names | |
|---|---|
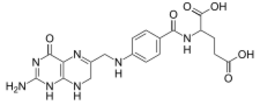
| IUPAC name
N-(4-{[(2-amino-4-oxo-1,4,7,8-tetrahydropteridin-6-yl)methyl]amino}benzoyl)-L-glutamic acid
| |
| Other names
H2folate, DH
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| MeSH | dihydrofolate |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C19H21N7O6 | |
| Molar mass | 443.414 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
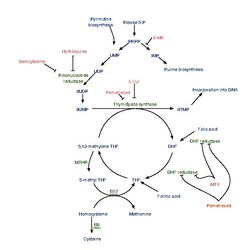
Dihydrofolic acid (conjugate base dihydrofolate) (DHF) is a folic acid (vitamin B9) derivative which is converted to tetrahydrofolic acid by dihydrofolate reductase.[1] Since tetrahydrofolate is needed to make both purines and pyrimidines, which are building blocks of DNA and RNA, dihydrofolate reductase is targeted by various drugs to prevent nucleic acid synthesis.
Interactive pathway map
Further reading
- Gangjee, Aleem; Jain, Hiteshkumar D.; Kurup, Sonali (2007). "Recent Advances in Classical and Non-Classical Antifolates as Antitumor and Antiopportunistic Infection Agents: Part I". Anti-Cancer Agents in Medicinal Chemistry 7 (5): 524–542. doi:10.2174/187152007781668724. PMID 17896913.
References
- ↑ "Dissociation constants for dihydrofolic acid and dihydrobiopterin and implications for mechanistic models for dihydrofolate reductase". Biochemistry 29 (19): 4554–60. May 1990. doi:10.1021/bi00471a008. PMID 2372539.
 |