Chemistry:Dichlorine hexoxide
| |||
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Dichlorine hexoxide
| |||
| Other names
Chlorine trioxide; Chloryl perchlorate; Chlorine(V,VII) oxide
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider |
| ||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| Cl 2O 6 | |||
| Molar mass | 166.901 g/mol | ||
| Appearance | red liquid | ||
| Density | 1.65 g/cm3 | ||
| Melting point | 3.5 °C (38.3 °F; 276.6 K) | ||
| Boiling point | 200 °C (392 °F; 473 K) | ||
| Reacts | |||
| Hazards | |||
| Main hazards | oxidizer | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
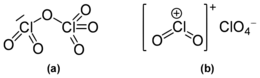
Dichlorine hexoxide is the chemical compound with the molecular formula Cl
2O
6 or O
2Cl–O–ClO
3, which is correct for its gaseous state. However, in liquid or solid form, this chlorine oxide ionizes into the dark red ionic compound chloryl perchlorate or dioxochloronium(V) perchlorate [ClO
2]+
[ClO
4]−
, which may be thought of as the mixed anhydride of chloric and perchloric acids. This compound is a notable perchlorating agent.[1]
Molecular structure
It was originally reported to exist as the monomeric chlorine trioxide ClO
3 in gas phase,[2] but was later shown to remain an oxygen-bridged dimer after evaporation and until thermal decomposition into chlorine perchlorate, Cl
2O
4, and oxygen.[3] The compound ClO
3 was then rediscovered.[4]
It is a dark red fuming liquid at room temperature that crystallizes as a red ionic compound, chloryl perchlorate, [ClO
2]+
[ClO
4]−
. The red color shows the presence of chloryl ions. Thus, chlorine's formal oxidation state in this compound remains a mixture of chlorine(V) and chlorine(VII) both in the gas phase and when condensed; however by breaking one oxygen-chlorine bond some electron density does shifts towards the chlorine(VII).[citation needed]
Properties
Cl
2O
6 is diamagnetic and is a very strong oxidizing agent. Although stable at room temperature, it explodes violently on contact with organic compounds[5] It is a strong dehydrating agent:
- Cl
2O
6 + H
2O → HClO
4 + HClO
3
Many reactions involving Cl
2O
6 reflect its ionic structure, [ClO
2]+
[ClO
4]−
, including the following:[6]
- NO
2F + Cl
2O
6 → [NO
2]+
ClO−
4 + ClO
2F - NO + Cl
2O
6 → [NO]+
ClO−
4 + ClO
2 - 2 V
2O
5 + 12 Cl
2O
6 → 4 VO(ClO
4)
3 + 12 ClO
2 + 3 O
2 - SnCl
4 + 6 Cl
2O
6 → [ClO
2]
2[Sn(ClO
4)
6] + 4 ClO
2 + 2 Cl
2
It reacts with gold to produce the chloryl salt [ClO
2]+
[Au(ClO
4)
4]−
:[7]
- 2 Au + 6 Cl
2O
6 → 2 [ClO
2]+
[Au(ClO
4)
4]−
+ Cl
2
Several other transition metal perchlorate complexes are prepared using dichlorine hexoxide.
Nevertheless, it can also react as a source of the ClO
3 radical:[citation needed]
- 2 AsF
5 + Cl
2O
6 → 2 ClO
3AsF
5
Synthesis
References
- ↑ Jean-Louis Pascal; Frédéric Favier (1998). "Inorganic perchlorato complexes" (in en). Coordination Chemistry Reviews 178-180 (1): 865–902. doi:10.1016/S0010-8545(98)00102-7.
- ↑ C. F. Goodeve, F. A. Todd (1933). "Chlorine Hexoxide and Chlorine Trioxide". Nature 132 (3335): 514–515. doi:10.1038/132514b0. Bibcode: 1933Natur.132..514G.
- ↑ Lopez, Maria; Juan E. Sicre (1990). "Physicochemical properties of chlorine oxides. 1. Composition, ultraviolet spectrum, and kinetics of the thermolysis of gaseous dichlorine hexoxide". J. Phys. Chem. 94 (9): 3860–3863. doi:10.1021/j100372a094.
- ↑ Grothe, Hinrich; Willner, Helge (1994). "Chlorine Trioxide: Spectroscopic Properties, Molecular Structure, and Photochemical Behavior". Angew. Chem. Int. Ed. 33 (14): 1482–1484. doi:10.1002/anie.199414821.
- ↑ Mary Eagleson (1994). Concise encyclopedia chemistry. Walter de Gruyter. p. 215. ISBN 3-11-011451-8. https://archive.org/details/conciseencyclope00eagl.
- ↑ Harry Julius Emeléus, Alan George Sharpe (1963). Advances in Inorganic Chemistry and Radiochemistry. Academic Press. p. 65. ISBN 0-12-023605-2. https://books.google.com/books?id=pRXIwIV-hB8C.
- ↑ Cunin, Frédérique; Catherine Deudon; Frédéric Favier; Bernard Mula; Jean Louis Pascal (2002). "First Anhydrous Gold Perchlorato Complex: ClO2Au(ClO4)4. Synthesis and Molecular and Crystal Structure Analysis". Inorganic Chemistry 41 (16): 4173–4178. doi:10.1021/ic020161z. PMID 12160405.
 |