Chemistry:Chlorine trifluoride oxide
| |
| |
| Names | |
|---|---|
| IUPAC name
trifluoro(oxo)-λ5-chlorane
| |
| Other names
Chlorosyl trifluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
PubChem CID
|
|
| |
| |
| Properties | |
| ClOF 3 | |
| Molar mass | 108.44 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.865 g/cm3 |
| Melting point | −42 °C (−44 °F; 231 K) |
| Boiling point | 29 °C (84 °F; 302 K) |
| Structure | |
| monoclinic | |
| C2/m | |
a = 9.826, b = 12.295, c = 4.901 α = 90°, β = 90.338°, γ = 90°[2]
| |
Lattice volume (V)
|
592.1 |
Formula units (Z)
|
8 |
| Hazards | |
| GHS pictograms |    
|
| GHS Signal word | Danger |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Chlorine oxide trifluoride or chlorine trifluoride oxide is a corrosive colorless liquid molecular compound with formula ClOF
3. It was developed secretly as a rocket fuel oxidiser.
Production
Chlorine oxide trifluoride was originally made at Rocketdyne[3] by treating dichlorine monoxide with fluorine. Other substances that could react with fluorine to make it includes sodium chlorite NaClO
2, and chlorine nitrate Cl–O–NO
2. The first published production method was a reaction of dichlorine monoxide with oxygen difluoride OF
2. Yet other production methods are reactions between ClO
2F or ClO
3F and chlorine fluorides.[4] A safer approach is the use chlorine nitrate with fluorine.
Reactions
As a Lewis base it can lose a fluoride ion to Lewis acids, yielding the difluorooxochloronium(V) cation [ClOF
2]+
.[5] Compounds with this include: [ClOF
2]+
[BF
4]−
, [ClOF
2]+
[PF
6]−
, [ClOF
2]+
[AsF
6]−
, [ClOF
2]+
[SbF
6]−
, [ClOF
2]+
[BiF
6]−
, [ClOF
2]+
[VF
6]−
, [ClOF
2]+
[NbF
6]−
, [ClOF
2]+
[TaF
6]−
, [ClOF
2]+
[UF
6]−
, ([ClOF
2]+
)
2[SiF
6]2−, [ClOF
2]+
[MoOF
5]−
, [ClOF
2]+
[Mo
2O
4F
9]−
,[4] [ClOF
2]+
[PtF
6].[6]
Functioning as a Lewis acid, it can gain a fluoride ion from a strong base to yield a tetrafluorooxochlorate(V) anion ClOF−
4.[7] These include K+
[ClOF
4]−
(potassium tetrafluorooxochlorate(V)), Rb+
[ClOF
4]−
(rubidium tetrafluorooxochlorate(V)), and Cs+
[ClOF
4]−
(caesium tetrafluorooxochlorate(V)). These three salts are white crystalline solids.[4][8] This allows purification of ClOF
3, as at room temperature a solid complex is formed, but this decomposes between 50 and 70 °C. Other likely impurities either will not react with alkali fluoride, or if they do will not easily decompose.[3]
Chlorine trifluoride oxide fluoridates various materials such as chlorine monoxide, chlorine, glass or quartz.[3]
Chlorine trifluoride oxide adds to chlorine fluorosulfate:
- ClOF
3 + 2 ClOSO
2F → S
2O
5F
2 + FClO
2 + 2 ClF.
The reaction also produces SO
2F
2.[3]
Chlorine trifluoride oxide can fluoridate and add oxygen in the same reaction, reacting with molybdenum pentafluoride, silicon tetrafluoride, tetrafluorohydrazine (over 100 °C), HNF
2, and F
2NCOF. From HNF
2 the main result was NF
3O. From MoF
5, the results were MoF
6 and MoOF
4.[3]
It reacts explosively with hydrocarbons.[3] With small amounts of water, ClO
2F is formed along with HF.[3]
Over 280 °C ClOF
3 decomposes to oxygen and chlorine trifluoride.[3]
Properties
The boiling point of chlorine trifluoride oxide is 29 °C.[9]
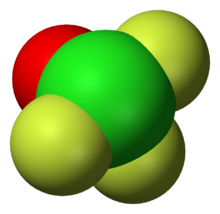
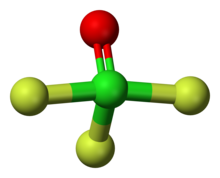
The shape of the molecule is a trigonal bipyramid, with two fluorine atoms at the top and bottom (apex) (Fa) and an electron pair, oxygen and fluorine (Fe) on the equator.[7] The Cl=O bond length is 1.405 Å, Cl-Fe 1.603 Å, other Cl-Fa 1.713 Å, ∠FeClO=109° ∠FaClO=95°, ∠FaClFe=88°. The molecule is polarised, Cl has a +1.76 charge, O has −0.53, equatorial F has −0.31 and apex F has −0.46. The total dipole moment is 1.74 D.[10]
References
- ↑ Urben, Peter (2017) (in en). Bretherick's Handbook of Reactive Chemical Hazards. Elsevier. p. 784. ISBN 9780081010594. https://books.google.com/books?id=rnXUDAAAQBAJ&pg=PA784.
- ↑ Ellern, Arkady; Boatz, Jerry A.; Christe, Karl O.; Drews, Thomas; Seppelt, Konrad (September 2002). "The Crystal Structures of ClF3O, BrF3O, and [NO]+[BrF4O]–". Zeitschrift für anorganische und allgemeine Chemie 628 (9–10): 1991–1999. doi:10.1002/1521-3749(200209)628:9/10<1991::AID-ZAAC1991>3.0.CO;2-1.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 (in en) Advances in Inorganic Chemistry and Radiochemistry. Academic Press. 1976. pp. 331–333. ISBN 9780080578675. https://books.google.com/books?id=EWlBFTxYth4C&pg=PA331.
- ↑ 4.0 4.1 4.2 Holloway, John H.; Laycock, David (1983) (in en). Advances in Inorganic Chemistry. Academic Press. pp. 178–179. ISBN 9780080578767. https://books.google.com/books?id=W4RngLN275wC&pg=PA178.
- ↑ Christe, Karl O.; Curtis, E. C.; Schack, Carl J. (September 1972). "Chlorine trifluoride oxide. VII. Difluorooxychloronium(V) cation, ClF2O+. Vibrational spectrum and force constants". Inorganic Chemistry 11 (9): 2212–2215. doi:10.1021/ic50115a046.
- ↑ 6.0 6.1 6.2 Schack, Carl J.; Lindahl, C. B.; Pilipovich, Donald.; Christe, Karl O. (September 1972). "Chlorine trifluoride oxide. IV. Reaction chemistry". Inorganic Chemistry 11 (9): 2201–2205. doi:10.1021/ic50115a043.
- ↑ 7.0 7.1 Christe, K.O.; Schack, C.J. (1976). Chlorine Oxyfluorides. Advances in Inorganic Chemistry and Radiochemistry. 18. pp. 319–398. doi:10.1016/S0065-2792(08)60033-3. ISBN 9780120236183.
- ↑ Christe, Karl O.; Schack, Carl J.; Pilipovich, Donald.; Christe, Karl O. (September 1972). "Chlorine trifluoride oxide. V. Complex formation with Lewis acids and bases". Inorganic Chemistry 11 (9): 2205–2208. doi:10.1021/ic50115a044.
- ↑ Pilipovich, Donald.; Lindahl, C. B.; Schack, Carl J.; Wilson, R. D.; Christe, Karl O. (September 1972). "Chlorine trifluoride oxide. I. Preparation and properties". Inorganic Chemistry 11 (9): 2189–2192. doi:10.1021/ic50115a040.
- ↑ Oberhammer, Heinz.; Christe, Karl O. (January 1982). "Gas-phase structure of chlorine trifluoride oxide, ClF3O". Inorganic Chemistry 21 (1): 273–275. doi:10.1021/ic00131a050.
 |
