Chemistry:Picolinic acid
| |
| Names | |
|---|---|
| Preferred IUPAC name
Pyridine-2-carboxylic acid | |
| Other names
Picolinic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C6H5NO2 | |
| Molar mass | 123.111 g·mol−1 |
| Appearance | White solid |
| Density | 1.526 g/cm³ |
| Melting point | 136 to 138 °C (277 to 280 °F; 409 to 411 K) |
| Slightly soluble (0.41%) in water | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
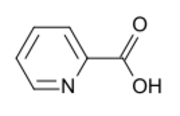
Picolinic acid is an organic compound with the formula NC
5H
4CO
2H. It is a derivative of pyridine with a carboxylic acid (COOH) substituent at the 2-position. It is an isomer of nicotinic acid and isonicotinic acid, which have the carboxyl side chain at the 3- and 4-positions, respectively. It is a white solid although impure samples can appear tan. The compound is soluble in water.
Production
On a commercial scale, picolinic acid is produced by ammoxidation of 2-picoline followed by hydrolysis of the resulting nitrile:
- NC
5H
4CH
3 + 1.5 O
2 + NH
3 → NC
5H
4C≡N + 3 H
2O - NC
5H
4C≡N + 2 H
2O → NC
5H
4CO
2H + NH
3
It is also produced by oxidation of picoline with nitric acid.[1]
- 350px
In the laboratory, picolinic acid is formed from 2-methylpyridine by oxidation with potassium permanganate (KMnO4).[2][3]
Reactions
Hydrogenation of picolinic acid gives piperidine-2-carboxylic acid, a precursor to the drug Mepivacaine.
Picolinic acid is a bidentate chelating agent of elements such as chromium, zinc, manganese, copper, iron, and molybdenum in the human body.[4][5]
It is a substrate in the Mitsunobu reaction. In the Hammick reaction, picolinic acid reacts with ketones to give pyridine-2-carbonols:[6]
- NC
5H
4CO
2H + R
2C=O → NC
5H
4CR
2(OH) + CO
2
Biosynthesis
Picolinic acid is a catabolite of the amino acid tryptophan through the kynurenine pathway.[7][8][9]
The immediate precursor is 2-amino-3-carboxymuconic semialdehyde, which can spontaneously cyclise to quinolinic acid. However, the enzyme aminocarboxymuconate-semialdehyde decarboxylase removes one of its carboxylic acid groups and initially produces 2-aminomuconic semialdehyde.[9]
This intermdiate is chemically unstable and ring-closes to picolinic acid, with loss of water.[9][10]
The function of picolinic acid is unclear, but it has been implicated in a variety of neuroprotective, immunological, and anti-proliferative effects. In addition, it is suggested to assist in the absorption of zinc(II) ions and other divalent or trivalent ions through the small intestine.[11]
Picolinates
Salts of picolinic acid (picolinates) include:
See also
References
- ↑ Shimizu, Shinkichi; Watanabe, Nanao; Kataoka, Toshiaki; Shoji, Takayuki; Abe, Nobuyuki; Morishita, Sinji; Ichimura, Hisao (2007). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a22_399.
- ↑ Singer, Alvin W.; McElvain, S. M. (1940). "Picolinic Acid Hydrochloride". Organic Syntheses 20: 79. doi:10.15227/orgsyn.020.0079.
- ↑ Harold Hart, Leslie E. Craine, David J. Hart, Christopher M. Hadad; Nicole Kindler (2007). 'Organische Chemie 3. Auflage. Weinheim: Wiley-VCH. p. 494. ISBN 978-3-527-31801-8.
- ↑ Lumme, Paavo; Lundgren, Georg; Mark, Wanda; Lundström, Hans; Borch, Gunner; Craig, J. Cymerman (1969). "The Crystal Structure of Zinc Picolinate Tetrahydrate, Zn(C6H4O2N)2(H2O)4". Acta Chemica Scandinavica 23: 3011–3022. doi:10.3891/acta.chem.scand.23-3011.
- ↑ Daugulis, Olafs; Roane, James; Tran, Ly Dieu (2015). "Bidentate, Monoanionic Auxiliary-Directed Functionalization of Carbon–Hydrogen Bonds". Accounts of Chemical Research 48 (4): 1053–1064. doi:10.1021/ar5004626. PMID 25756616.
- ↑ Fuchs, Philip L. (2013-07-29). "Picolinic acid". Catalytic Oxidation Reagents. Wiley Inc.. p. 495ff. ISBN 9781118704844. OCLC 954583821. https://books.google.com/books?id=Cbc2AAAAQBAJ&q=picolinic%20acid.
- ↑ Tan, L. (December 2012). "The kynurenine pathway in neurodegenerative diseases: mechanistic and therapeutic considerations". J Neurol Sci 323 (1–2): 1–8. doi:10.1016/j.jns.2012.08.005. PMID 22939820.
- ↑ Zeng, Ting; Liang, Yanshan; Chen, Jinyao; Cao, Guodong; Yang, Zhu; Zhao, Xingchen; Tian, Jinglin; Xin, Xiong et al. (2021-09-01). "Urinary metabolic characterization with nephrotoxicity for residents under cadmium exposure" (in en). Environment International 154. doi:10.1016/j.envint.2021.106646. ISSN 0160-4120. PMID 34049269. Bibcode: 2021EnInt.15406646Z.
- ↑ 9.0 9.1 9.2 Savitz, J (25 January 2020). "The kynurenine pathway: a finger in every pie.". Molecular Psychiatry 25 (1): 131–147. doi:10.1038/s41380-019-0414-4. PMID 30980044.
- ↑ Ichiyama, Arata; Nakamura, Shigenobu; Kawai, Hitoshi; Honjo, Tasuku; Nishizuka, Yasutomi; Hayaishi, Osamu; Senoh, Siro (1965). "Studies on the Metabolism of the Benzene Ring of Tryptophan in Mammalian Tissues". Journal of Biological Chemistry 240 (2): 740–749. doi:10.1016/S0021-9258(17)45238-0.
- ↑ Evans, Gary (1982). "The Role of Picolinic Acid in Metal Metabolism". Life Chemistry Reports (Harwood Academic Publishers) 1: 57–67. http://naldc.nal.usda.gov/download/46436/PDF. Retrieved 20 March 2015.
 |
