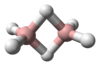Chemistry:1-Butyne
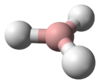
1-Butyne is an organic compound with the formula CH
3CH
2C≡CH. It is a terminal alkyne. The compound is a common terminal alkyne substrate in diverse studies of catalysis. It is a colorless combustible gas.[1] In 2017, 3.9 million pounds (1,700 long tons) was produced in the USA.[2]
1-Butyne participates in reactions typical for terminal alkynes, such as alkyne metathesis,[3] hydrogenation, condensation with formaldehyde. Based on its heat of combustion, it is slightly more stable than its isomer 2-butyne.[4]
The combustion of 1-Butyne produces propargyl radicals, a pre-cursor to soot and polycyclic aromatic hydrocarbons, as the propargyl radicals can form basic aromatic rings, making butyne's fuel usage a concern for emissions.[5]
1-Butyne is in unsaturated C4 petroleum cuts, and has to be separated out in industrial hydrorefining to make 1-butene, which is used to make low density polyethylene and polybutene. Distillation is impractical due to similar boiling points, so 1-butyne is removed by catalytic hydrogenation.[6] Usually the catalyst is palladium, operated with liquid hydrocarbon and hydrogen gas at 20-60°C and pressures up to 10 bar.[7]
See also
References
- ↑ Lide, David R. (2008). CRC Handbook of Chemistry and Physics, 89th Edition. CRC Press. pp. 3–84. ISBN 978-0-8493-0488-0.
- ↑ EPA, US. "US EPA: Chemical Data Reporting: 1-Butyne" (in en). https://chemview.epa.gov/chemview/.
- ↑ Zhang, Wei; Kraft, Stefan; Moore, Jeffrey S. (2004). "Highly Active Trialkoxymolybdenum(VI) Alkylidyne Catalysts Synthesized by a Reductive Recycle Strategy". Journal of the American Chemical Society 126 (1): 329–335. doi:10.1021/ja0379868. PMID 14709099.
- ↑ Prosen, E.J.; Maron, F.W.; Rossini, F.D. (1951). "Heats of combustion, formation, and insomerization of ten C4 hydrocarbons". Journal of Research of the National Bureau of Standards 46 (2): 106. doi:10.6028/jres.046.015.
- ↑ Lu, Haitao; Dong, Shijun; Liu, Fuqiang; Nagaraja, Shashank S.; Lindblade, Nathan; Turner, Mattias A.; Mathieu, Olivier; Petersen, Eric L. et al. (1 January 2023). "A wide-range experimental and kinetic modeling study of the pyrolysis and oxidation of 1-butyne". Proceedings of the Combustion Institute 39 (1): 355–364. doi:10.1016/j.proci.2022.09.044. https://www.sciencedirect.com/science/article/pii/S1540748922004357. Retrieved 2 April 2025.
- ↑ García Colli, G.; Alves, J. A.; Martínez, O. M.; Barreto, G. F. (1 August 2019). "Application of a catalytic membrane reactor to the selective hydrogenation of 1-Butyne". Chemical Engineering and Processing - Process Intensification 142: 107518. doi:10.1016/j.cep.2019.04.018.
- ↑ Alves, J. A.; Bressa, S. P.; Martínez, O. M.; Barreto, G. F. (15 January 2007). "Kinetic study of the liquid-phase hydrogenation of 1-butyne over a commercial palladium/alumina catalyst". Chemical Engineering Journal 125 (3): 131–138. doi:10.1016/j.cej.2006.08.006. https://www.sciencedirect.com/science/article/pii/S1385894706003226.
External links
 |