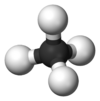
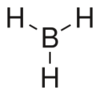
Chemistry:Borane
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Borane[1]
| |||
| Systematic IUPAC name
Borane (substitutive) Trihydridoboron (additive) | |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| 44 | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| BH3 | |||
| Molar mass | 13.83 g·mol−1 | ||
| Appearance | colourless gas | ||
| Conjugate acid | Boronium | ||
| Thermochemistry | |||
Std molar
entropy (S |
187.88 kJ mol−1 K−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
106.69 kJ mol−1 | ||
| Structure | |||
| D3h | |||
| trigonal planar | |||
| 0 D | |||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
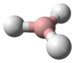
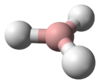
Borane, also known as borine, is an unstable and highly reactive molecule with the chemical formula BH3. The preparation of borane carbonyl, BH3(CO), played an important role in exploring the chemistry of boranes, as it indicated the likely existence of the borane molecule.[2] However, the molecular species BH3 is a very strong Lewis acid. Consequently, it is highly reactive and can only be observed directly as a continuously produced, transitory, product in a flow system or from the reaction of laser ablated atomic boron with hydrogen.[3] It normally dimerizes to diborane in the absence of other chemicals.[4]
Structure and properties
BH3 is a trigonal planar molecule with D3h symmetry. The experimentally determined B–H bond length is 119 pm.[5]
In the absence of other chemical species, it reacts with itself to form diborane. Thus, it is an intermediate in the preparation of diborane according to the reaction:[6]
- BX3 +BH4− → HBX3− + (BH3) (X=F, Cl, Br, I)
- 2 BH3 → B2H6
The standard enthalpy of dimerization of BH3 is estimated to be −170 kJ mol−1.[7] The boron atom in BH3 has 6 valence electrons. Consequently, it is a strong Lewis acid and reacts with any Lewis base ('L' in equation below) to form an adduct:[8]
- BH3 + L → L—BH3
in which the base donates its lone pair, forming a dative covalent bond. Such compounds are thermodynamically stable, but may be easily oxidised in air. Solutions containing borane dimethylsulfide and borane–tetrahydrofuran are commercially available; in tetrahydrofuran a stabilising agent is added to prevent the THF from oxidising the borane.[9] A stability sequence for several common adducts of borane, estimated from spectroscopic and thermochemical data, is as follows:
BH3 has some soft acid characteristics as sulfur donors form more stable complexes than do oxygen donors.[6] Aqueous solutions of BH3 are extremely unstable.[10][11]
- BH3 + 3H
2O → B(OH)3 + 3 H2
Reactions
Molecular BH3 is believed to be a reaction intermediate in the pyrolysis of diborane to produce higher boranes:[6]
- B2H6 ⇌ 2BH3
- BH3 +B2H6 → B3H7 +H2 (rate determining step)
- BH3 + B3H7 ⇌ B4H10
- B2H6 + B3H7 → BH3 + B4H10
- ⇌ B5H11 + H2
Further steps give rise to successively higher boranes, with B10H14 as the most stable end product contaminated with polymeric materials, and a little B20H26.
Borane ammoniate, which is produced by a displacement reaction of other borane adducts, eliminates elemental hydrogen on heating to give borazine (HBNH)3.[12]
Borane adducts are widely used in organic synthesis for hydroboration, where BH3 adds across the C=C bond in alkenes to give trialkylboranes:[13]
- (THF)BH3 + 3 CH2=CHR → B(CH2CH2R)3 + THF
This reaction is regioselective.[14] Other borane derivatives can be used to give even higher regioselectivity.[15] The product trialkylboranes can be converted to useful organic derivatives. With bulky alkenes one can prepare species such as [HBR2]2, which are also useful reagents in more specialised applications. Borane dimethylsulfide which is more stable than borane–tetrahydrofuran may also be used.[16][15]
Hydroboration can be coupled with oxidation to give the hydroboration-oxidation reaction. In this reaction, the boryl group in the generated organoborane is substituted with a hydroxyl group.[17]
As a Lewis acid
Phosphine-boranes, with the formula R3−nHnPBH3, are adducts of organophosphines and borane.
Borane(5) is the dihydrogen complex of borane. Its molecular formula is BH5 or possibly BH3(η2-H2).[18] It is only stable at very low temperatures and its existence is confirmed in very low temperature.[19][20] Borane(5) and methanium (CH5+) are isoelectronic.[21] Its conjugate base is the borohydride anion.
See also
References
- ↑ "Borane". https://pubchem.ncbi.nlm.nih.gov/compound/Borane#section=IUPAC-Name&fullscreen=true.
- ↑ Burg, Anton B.; Schlesinger, H. I. (May 1937). "Hydrides of boron. VII. Evidence of the transitory existence of borine (BH3): Borine carbonyl and borine trimethylammine". Journal of the American Chemical Society 59 (5): 780–787. doi:10.1021/ja01284a002.
- ↑ Tague, Thomas J.; Andrews, Lester (1994). "Reactions of Pulsed-Laser Evaporated Boron Atoms with Hydrogen. Infrared Spectra of Boron Hydride Intermediate Species in Solid Argon". Journal of the American Chemical Society 116 (11): 4970–4976. doi:10.1021/ja00090a048. ISSN 0002-7863.
- ↑ Carey, Francis A.; Sundberg, Richard J. (2007). Advanced Organic Chemistry: Part B: Reactions and Synthesis (5th ed.). New York: Springer. p. 337. ISBN 978-0387683546.
- ↑ Kawaguchi, Kentarou (1992). "Fourier transform infrared spectroscopy of the BH3 ν3 band". The Journal of Chemical Physics 96 (5): 3411–3415. doi:10.1063/1.461942. ISSN 0021-9606. Bibcode: 1992JChPh..96.3411K.
- ↑ 6.0 6.1 6.2 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Page, M.; Adams, G.F.; Binkley, J.S.; Melius, C.F. (1987). "Dimerization energy of borane". J. Phys. Chem. 91 (11): 2675–2678. doi:10.1021/j100295a001.
- ↑ Carey, Francis A.; Sundberg, Richard J. (2007). Advanced Organic Chemistry: Part B: Reactions and Synthesis (5th ed.). New York: Springer. p. 337. ISBN 978-0387683546.
- ↑ Hydrocarbon Chemistry, George A. Olah, Arpad Molner, 2d edition, 2003, Wiley-Blackwell ISBN 978-0471417828
- ↑ Finn, Patricia; Jolly, William L. (August 1972). "Asymmetric cleavage of diborane by water. The structure of diborane dihydrate". Inorganic Chemistry 11 (8): 1941–1944. doi:10.1021/ic50114a043. https://escholarship.org/uc/item/7nb9k0rm.
- ↑ D'Ulivo, Alessandro (May 2010). "Mechanism of generation of volatile species by aqueous boranes". Spectrochimica Acta Part B: Atomic Spectroscopy 65 (5): 360–375. doi:10.1016/j.sab.2010.04.010.
- ↑ Housecroft, C. E.; Sharpe, A. G. (2008). "Chapter 13: The Group 13 Elements". Inorganic Chemistry (3rd ed.). Pearson. p. 336. ISBN 978-0-13-175553-6. https://archive.org/details/inorganicchemist00hous_159.
- ↑ Carey, Francis A.; Sundberg, Richard J. (2007). Advanced Organic Chemistry: Part B: Reactions and Synthesis (5th ed.). New York: Springer. p. 337. ISBN 978-0387683546.
- ↑ Carey, Francis A.; Sundberg, Richard J. (2007). Advanced Organic Chemistry: Part B: Reactions and Synthesis (5th ed.). New York: Springer. p. 338. ISBN 978-0387683546.
- ↑ 15.0 15.1 Burkhardt, Elizabeth R.; Matos, Karl (July 2006). "Boron reagents in process chemistry: Excellent tools for selective reductions". Chemical Reviews 106 (7): 2617–2650. doi:10.1021/cr0406918. PMID 16836295.
- ↑ Kollonitisch, J. (1961). "Reductive Ring Cleavage of Tetrahydrofurans by Diborane". J. Am. Chem. Soc. 83 (6): 1515. doi:10.1021/ja01467a056.
- ↑ Carey, Francis A.; Sundberg, Richard J. (2007). Advanced Organic Chemistry: Part B: Reactions and Synthesis (5th ed.). New York: Springer. p. 344. ISBN 978-0387683546.
- ↑ Szieberth, Dénes; Szpisjak, Tamás; Turczel, Gábor; Könczöl, László (19 August 2014). "The stability of η2-H2 borane complexes – a theoretical investigation". Dalton Transactions 43 (36): 13571–13577. doi:10.1039/C4DT00019F. PMID 25092548.
- ↑ Tague, Thomas J.; Andrews, Lester (1 June 1994). "Reactions of Pulsed-Laser Evaporated Boron Atoms with Hydrogen. Infrared Spectra of Boron Hydride Intermediate Species in Solid Argon". Journal of the American Chemical Society 116 (11): 4970–4976. doi:10.1021/ja00090a048.
- ↑ Schreiner, Peter R.; Schaefer III, Henry F.; Schleyer, Paul von Ragué (1 June 1994). "The structure and stability of BH5. Does correlation make it a stable molecule? Qualitative changes at high levels of theory". The Journal of Chemical Physics 101 (9): 7625. doi:10.1063/1.468496. Bibcode: 1994JChPh.101.7625S.
- ↑ A Life of Magic Chemistry: Autobiographical Reflections Including Post-Nobel Prize Years and the Methanol Economy, 159p
 |