Chemistry:Stannane
From HandWiki
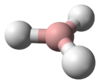
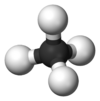
Stannane /ˈstæneɪn/ or tin hydride is an inorganic compound with the chemical formula SnH4. It is a colourless gas that ignites on contact with air. SnH4 is a heavy analogue of methane but much less robust. In contrast to this rarely used species, stannane also refers to a large collection of organotin compounds.
Preparation and reactions
Stannane can be prepared by the reaction of SnCl
4 and lithium aluminium hydride according to the following idealized equation:[1]
- SnCl
4 + Li[AlH
4] → SnH
4 + LiCl + AlCl
3
Stannane decomposes slowly at room temperature to give metallic tin and hydrogen.[1]
- SnH
4 → Sn + 2H
2
Organotin hydrides are more stable, e.g. triphenyltin hydride, also known as triphenylstannane.
Occurrence in industry
The volatility of stannane is potentially relevant to production of microchips.[2]
See also
- Organotin
References
- ↑ 1.0 1.1 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Qerimi, Dren; Herschberg, Andrew C.; Panici, Gianluca; Hays, Parker; Pohlman, Tyler; Ruzic, David N. (2022). "Tin removal by an annular surface wave plasma antenna in an extreme ultraviolet lithography source". Journal of Applied Physics 132 (11). doi:10.1063/5.0094375.
 |