Chemistry:1-Heptene
From HandWiki
1-Heptene is an organic compound with the formula CH
2=CH(CH
2)
4CH
3. It is the terminal "linear" C7 alkene. A colorless volatile liquid, it is produced as one of myriad products by the Fischer-Tropsch Process. It is used to prepare 1-octene, a common comonomer.[1] It is classified as higher olefin, or alkene with the formula C7H14.
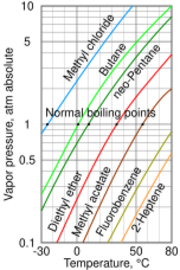
Four isomers of 1-heptene are known: cis- and trans-2-heptene as well as cis- and trans-3-heptene.
Further reading
- Carl Schaschke, 2014, A Dictionary of Chemical Engineering, Oxford University Press.
- G. I. Nikishin, Yu. N. Ogibin & L. Kh. Rakhmatullina, 1975, ‘Peroxydisulfate-initiated reactions of 1-heptene with acetic and propionic acids ’, Bulletin of the Academy of Sciences of the USSR, Division of chemical science, volume 23, pages1479–1483 * Yu. D. Shenin, T. V. Kotenko & A. N. Egorenkova, Nystatin. IV. 1969, The heptaene component of samples of nystatin-nursimicin , Pharmaceutical Chemistry Journal volume 3, pages 631–634 * Nicholas E. Leadbeater, Cynthia B. McGowan, 2013, Experiment 2: Second-Order Elimination Reaction Preparation of Heptene from 2-Bromoheptane , Laboratory Experiments Using Microwave Heating, chapter 3 * E. S. Mortikov, M. I. Rozengart & B. A. Kazanskii, 1968, Dehydrocyclization of n-heptenes under conditions of a pulsed system and in the usual flow-type setup , Bulletin of the Academy of Sciences of the USSR, Division of chemical science volume 17, pages95–98(1968)
References
- ↑ Schmidt, Roland; Griesbaum, Karl; Behr, Arno; Biedenkapp, Dieter; Voges, Heinz-Werner; Garbe, Dorothea; Paetz, Christian; Collin, Gerd et al. (2014). "Hydrocarbons". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–74. doi:10.1002/14356007.a13_227.pub3. ISBN 978-3-527-30673-2.
 |