Chemistry:Isosorbide dinitrate
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Isordil, others[1] |
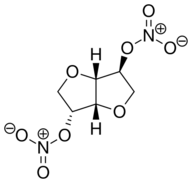
| Other names | ISDN; (3R,3aS,6S,6aS)-hexahydrofuro[3,2-b]furan-3,6-diyl dinitrate |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682348 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 10–90%, average 25% |
| Metabolism | Liver |
| Elimination half-life | 1 hour |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C6H8N2O8 |
| Molar mass | 236.136 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Isosorbide dinitrate is a medication used for heart failure, esophageal spasms, and to treat and prevent chest pain from not enough blood flow to the heart.[1] It has been found to be particularly useful in heart failure due to systolic dysfunction together with hydralazine.[2][1] It is taken by mouth or under the tongue.[1]
Common side effects include headache, lightheadedness with standing, and blurred vision.[1] Severe side effects include low blood pressure.[1] It is unclear if use in pregnancy is safe for the baby.[1] It should not be used together with PDE5 Inhibitors.[1] Isosorbide dinitrate is in the nitrate family of medications and works by dilating blood vessels.[1]
Isosorbide dinitrate was first written about in 1939.[3] It is on the World Health Organization's List of Essential Medicines.[4] Isosorbide dinitrate is available as a generic medication.[1][5] A long-acting form exists.[1] In 2020, it was the 299th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[6][7]
Medical uses
It is used for angina, in addition to other medications for congestive heart failure, and for esophageal spasms.[1] It is available as an oral tablet both in extended release and slow release. The onset of action for Isosorbide Dinitrate is thirty minutes and the onset of action for oral extended release is 12–24 hours.
Long-acting nitrates can be more useful as they are generally more effective and stable in the short term.
Side effects
Tolerance
After long-term use for treating chronic conditions, tolerance may develop in patients, reducing its effectiveness. The mechanisms of nitrate tolerance have been thoroughly investigated in the last 30 years and several hypotheses have been proposed. These include:
- Impaired biotransformation of isosorbide dinitrate to its active principle NO (or a NO-related species)
- Neurohormonal activation, causing sympathetic activation and release of vasoconstrictors such as endothelin and angiotensin II which counteract the vasodilation induced by isosorbide dinitrate
- Plasma volume expansion
- The oxidative stress hypothesis[8]
The last hypothesis might represent a unifying hypothesis, and an isosorbide dinitrate-induced inappropriate production of oxygen free radicals might induce a number of abnormalities which include the ones described above. Furthermore, nitrate tolerance is shown to be associated with vascular abnormalities which have the potential to worsen patients prognosis:[9] these include endothelial and autonomic dysfunction.[10]
Other side effects
In the short run, isosorbide dinitrate can cause severe headaches, necessitating analgesic administration for relief of pain, as well as severe hypotension, and, in certain cases, bradycardia.
Rarely occurring are allergic reactions (rash; hives; itching; difficulty breathing; tightness in the chest; swelling of the mouth, face, lips, or tongue); fainting; fast or slow heartbeat; nausea; new or worsening chest pain; vomiting.
Mechanism of action
Similar to other nitrites and organic nitrates, isosorbide dinitrate is converted to nitric oxide (NO), an active intermediate compound which activates the enzyme guanylate cyclase (atrial natriuretic peptide receptor A). This stimulates the synthesis of cyclic guanosine 3',5'-monophosphate (cGMP) which then activates a series of protein kinase-dependent phosphorylations in the smooth muscle cells, eventually resulting in the dephosphorylation of the myosin light chain of the smooth muscle fiber. The subsequent sequestration of calcium ions results in the relaxation of the smooth muscle cells and vasodilation.[11]
Society and culture
Isosorbide dinitrate is sold in the US under the brand names Dilatrate-SR by Schwarz and Isordil by Valeant, according to FDA Orange Book. It is sold under the trade name Isoket in the United Kingdom, Argentina, and Hong Kong. It is also a component of BiDil.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 "Isosorbide Dinitrate/Mononitrate". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/isosorbide-dinitrate-mononitrate.html.
- ↑ "Pharmacologic management of heart failure caused by systolic dysfunction". American Family Physician 77 (7): 957–964. April 2008. PMID 18441861.
- ↑ (in en) Analogue-based Drug Discovery. John Wiley & Sons. 2006. p. 454. ISBN 9783527607495. https://books.google.com/books?id=FjKfqkaKkAAC&pg=PA454.
- ↑ World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Competitive Generic Therapy Approvals". 29 June 2023. https://www.fda.gov/drugs/generic-drugs/competitive-generic-therapy-approvals.
- ↑ "The Top 300 of 2020". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Isosorbide Dinitrate - Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/IsosorbideDinitrate.
- ↑ "Evidence for enhanced vascular superoxide anion production in nitrate tolerance. A novel mechanism underlying tolerance and cross-tolerance". The Journal of Clinical Investigation 95 (1): 187–94. January 1995. doi:10.1172/JCI117637. PMID 7814613.
- ↑ "Long-term nitrate use may be deleterious in ischemic heart disease: A study using the databases from two large-scale postinfarction studies. Multicenter Myocardial Ischemia Research Group". American Heart Journal 138 (3 Pt 1): 577–85. September 1999. doi:10.1016/s0002-8703(99)70163-8. PMID 10467211.
- ↑ "Nitrate-induced toxicity and preconditioning: a rationale for reconsidering the use of these drugs". Journal of the American College of Cardiology 52 (4): 251–4. July 2008. doi:10.1016/j.jacc.2008.04.019. PMID 18634978.
- ↑ Pharmacology (8th ed.). Elsevier. 2016. pp. 261. ISBN 978-0-7020-5362-7.
External links
- "Isosorbide dinitrate". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/isosorbide%20dinitrate.
 |
