Chemistry:Isocyanate
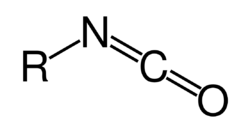
In organic chemistry, isocyanate is the functional group with the formula R–N=C=O. Organic compounds that contain an isocyanate group are referred to as isocyanates. An organic compound with two isocyanate groups is known as a diisocyanate. Diisocyanates are manufactured for the production of polyurethanes, a class of polymers.[1][2][3]
Isocyanates should not be confused with cyanate esters and isocyanides, very different families of compounds. The cyanate (cyanate ester) functional group (R–O–C≡N) is arranged differently from the isocyanate group (R–N=C=O). Isocyanides have the connectivity R–N≡C, lacking the oxygen of the cyanate groups.
Structure and bonding
In terms of bonding, isocyanates are closely related to carbon dioxide (CO2) and carbodiimides (C(NR)2). The C−N=C=O unit that defines isocyanates is planar, and the N=C=O linkage is nearly linear. In phenyl isocyanate, the C=N and C=O distances are respectively 1.195 and 1.173 Å. The C−N=C angle is 134.9° and the N=C=O angle is 173.1°.[4]
Production
Isocyanates are usually produced from amines by phosgenation, i.e. treating with phosgene:
- [math]\ce{ RNH2 + COCl2 -> RNCO + 2 HCl }[/math]
These reactions proceed via the intermediacy of a carbamoyl chloride (RNHC(O)Cl). Owing to the hazardous nature of phosgene, the production of isocyanates requires special precautions.[1]
Another route to isocyanates entails addition of isocyanic acid to alkenes. Complementarily, alkyl isocyanates form by displacement reactions involving alkyl halides and alkali metal cyanates.[5]
Three rearrangement reactions involving nitrenes give isocyanates:
- Schmidt reaction, a reaction where a carboxylic acid is treated with ammonia and hydrazoic acid yielding an isocyanate.
- Curtius rearrangement degradation of an acyl azide to an isocyanate and nitrogen gas.
- Lossen rearrangement, the conversion of a hydroxamic acid to an isocyanate via the formation of an O-acyl, sulfonyl, or phosphoryl intermediate.
Reactivity
With nucleophiles
Isocyanates are electrophiles, and as such they are reactive toward a variety of nucleophiles including alcohols, amines, and even water having a higher reactivity compared to structurally analogous isothiocyanates.[6]
Upon treatment with an alcohol, an isocyanate forms a urethane linkage:
- [math]\ce{ ROH + R'NCO -> ROC(O)N(H)R' }[/math]
where R and R' are alkyl or aryl groups. If a diisocyanate is treated with a compound containing two or more hydroxyl groups, such as a diol or a polyol, polymer chains are formed, which are known as polyurethanes.
File:Polyurethane synthesis.tif
Isocyanates react with water to form carbon dioxide:
- [math]\ce{ RNCO + H2O -> RNH2 + CO2 }[/math]
This reaction is exploited in tandem with the production of polyurethane to give polyurethane foams. The carbon dioxide functions as a blowing agent.[7]
Isocyanates also react with amines to give ureas:
- [math]\ce{ R2NH + R'NCO -> R2NC(O)N(H)R' }[/math]
The addition of an isocyanate to a urea gives a biuret:
- [math]\ce{ R2NC(O)N(H)R' + R''NCO -> R2NC(O)NR'C(O)NHR'' }[/math]
Reaction between a di-isocyanate and a compound containing two or more amine groups produces long polymer chains known as polyureas.
Carbodiimides are produced by the decarboxylation of alkyl and aryl isocyanate using phosphine oxides as a catalyst:[8]
- [math]\ce{ C6H11NCO -> (C6H11N)2C + CO2 }[/math]
Cyclization
Isocyanates also can react with themselves. Aliphatic diisocyanates can trimerise to from substituted isocyanuric acid groups. This can be seen in the formation of polyisocyanurate resins (PIR) which are commonly used as rigid thermal insulation. Isocyanates participate in Diels–Alder reactions, functioning as dienophiles.
Rearrangement reactions
Isocyanates are common intermediates in the synthesis of primary amines via hydrolysis:
- Hofmann rearrangement, a reaction in which a primary amide is treated with a strong oxidizer such as sodium hypobromite[9][10][11] or lead tetraacetate[12] to form an isocyanate intermediate.
Common isocyanates
The global market for diisocyanates in the year 2000 was 4.4 million tonnes, of which 61.3% was methylene diphenyl diisocyanate (MDI), 34.1% was toluene diisocyanate (TDI), 3.4% was the total for hexamethylene diisocyanate (HDI) and isophorone diisocyanate (IPDI), and 1.2% was the total for various others.[13] A monofunctional isocyanate of industrial significance is methyl isocyanate (MIC), which is used in the manufacture of pesticides.
Common applications
MDI is commonly used in the manufacture of rigid foams and surface coating.[1] Polyurethane foam boards are used in construction for insulation. TDI is commonly used in applications where flexible foams are used, such as furniture and bedding. Both MDI and TDI are used in the making of adhesives and sealants due to weather-resistant properties. Isocyanates, both MDI and TDI are widely used in as spraying applications of insulation due to the speed and flexibility of applications. Foams can be sprayed into structures and harden in place or retain some flexibility as required by the application.[14] HDI is commonly utilized in high-performance surface-coating applications, including automotive paints.
Health and safety
The risks of isocyanates was brought to the world's attention with the Bhopal disaster, which caused the death of nearly 4000 people from the accidental release of methyl isocyanate.
LD50s for isocyanates are typically several hundred milligrams per kilogram.[15] Despite this low acute toxicity, an extremely low short-term exposure limit (STEL) of 0.07 mg/m3 is the legal limit for all isocyanates (except methyl isocyanate: 0.02 mg/m3) in the United Kingdom.[16] These limits are set to protect workers from chronic health effects such as occupational asthma, contact dermatitis, or irritation of the respiratory tract.[17]
Since they are used in spraying applications, the properties of their aerosols have attracted attention.[18][19] In the U.S., OSHA conducted a National Emphasis Program on isocyanates starting in 2013 to make employers and workers more aware of the health risks.[20] Polyurethanes have variable curing times, and the presence of free isocyanates in foams vary accordingly.[21]
Both the US National Toxicology Program (NTP) and International Agency for Research on Cancer (IARC) have evaluated TDI as a potential human carcinogen and Group 2B "possibly carcinogenic to humans".[22][23] MDI appears to be relatively safer and is unlikely a human carcinogen.[23] The IARC evaluates MDI as Group 3 "not classifiable as to its carcinogenicity in humans".[24]
All major producers of MDI and TDI are members of the International Isocyanate Institute, which promotes the safe handling of MDI and TDI.
Hazards
Toxicity
Isocyanates can present respiratory hazards as particulates, vapors or aerosols. Autobody shop workers are a very commonly examined population for isocyanate exposure as they are repeatedly exposed when spray painting automobiles[25] and can be exposed when installing truck bed liners.[26][27] Hypersensitivity pneumonitis has slower onset and features chronic inflammation that can be seen on imaging of the lungs. Occupational asthma is a worrisome outcome of respiratory sensitization to isocyanates as it can be acutely fatal.[28] Diagnosis of occupational asthma is generally performed using pulmonary function testing (PFT) and performed by pulmonology or occupational medicine physicians.[29] Occupational asthma is much like asthma in that it causes episodic shortness of breath and wheezing. Both the dose and duration of exposure to isocyanates can lead to respiratory sensitization.[30] Dermal exposures to isocyanates can sensitize an exposed person to respiratory disease.
Dermal exposures can occur via mixing, spraying coatings or applying and spreading coatings manually. Dermal exposures to isocyanates is known to lead to respiratory sensitization.[31] Even when the right personal protective equipment (PPE) is used, exposures can occur to body areas not completely covered.[32] Isocyanates can also permeate improper PPE, necessitating frequent changes of both disposable gloves and suits if they become over exposed.
Flammability
Methyl isocyanate (MIC) is highly flammable.[33] MDI and TDI are much less flammable.[34] Flammability of materials is a consideration in furniture design.[35] The specific flammability hazard is noted on the safety data sheet (SDS) for specific isocyanates.
Hazard controls
Elimination and substitution seeks to eliminate a hazard directly from use in industrial processes. Elimination if possible also has the possibility of eliminating the need for other controls. If unable to make an elimination, substituting a less hazardous isocyanate may also control hazards. Because of the hazards inherent in isocyanates, there is ongoing research for suitable replacements.[36] The EPA has sponsored work on finding suitable replacements for isocyanates in polyurethane coatings.[37]
Engineering controls seek to decrease hazards by creating barriers to hazard exposure. Using the source–pathway–receptor model, an engineering control acts on the pathway to mitigate hazards emanating from the source from reaching the receptor. An automated spraying booth with a separate ventilation system would be an example of engineering controls. Appropriate ventilation is a common engineering control when using isocyanates.[18][38]
Administrative controls are policy or training based controls to decrease hazards. A quarterly training session on recognizing symptoms of occupational asthma or proper respirator use would be examples of administrative controls. Administrative controls can be effective in reducing hazards for which personal protective equipment does not exist, for example, no eating or smoking in work areas can prevent ingestion of hazardous chemicals. Training is required by OSHA[39]
Personal protective equipment (PPE) is the lowest level of hazard control. For isocyanates commonly used, PPE include respirators for inhalation hazards and gloves to minimize absorption of dermal hazards. PPE like respirators are sensitive to fit and require some maintenance periodically. In some autobody paint and clear-coat spraying applications exposure limits exceed the protection factor of half mask respirators, and a full-face mask is required.[25][26] Eye protection is an important component of PPE.[38] Gloves and coveralls are appropriate personal protective equipment for workers.[19][40] Gloves and protective clothing can be effective in reducing dermal exposures, but user resistance can arise due to loss of tactile sensation or increased thermal burden. The material and thickness of gloves are an important component of protection.[41][42]
Industrial hygiene
Exposure assessment is the domain of industrial hygienists. An objective of exposure assessment is to ensure regulatory compliance with occupational exposure limits (OELs) below. OSHA guidelines provide detailed technical guidance on measuring isocyanates by sampling and analytics procedures tailored to specific chemicals. In the case of MDI, sample is by glass-fiber filters at standard air flow rates and liquid chromatography.[43]
Occupational health surveillance is primarily the domain of medical professionals. This can include counseling, respirator fit testing, tracking of biologic exposure using biologic exposure indices (BEI) and PFT results. Biologic monitoring levels for isocyanates exist[44] but may not be commonly used. One example of a monitoring program by the United States Navy relies on pulmonary function testing and screening questionnaires.[45]
The combination of industrial hygiene and medical surveillance can have a significant effect on the incidence of occupational asthma.[46]
Emergency management is a complex process of preparation and should be considered in a setting where a release of bulk chemicals may threaten the well-being of the public. The Bhopal disaster involving release of MIC and resulting in the deaths of thousands of people and affecting hundreds of thousands more. As a result of major industrial incidents like this, public health officials have proposed disaster preparedness programs aimed at assessing hazards, prevention by engineering and coordinated responses.[47] More recently MIC was involved in an explosion at a pesticide manufacturing plant in West Virginia.[48]
Occupational exposure limits
Exposure limits can be expressed as ceiling limits, a maximal value, short-term exposure limits (STEL), a 15-minute exposure limit or an 8-hour time-weighted average limit (TWA). Below is a sampling, not exhaustive, as less common isocyanates also have specific limits within the United States, and in some regions there are limits on total isocyanate, which recognizes some of the uncertainty regarding the safety of mixtures of chemicals as compared to pure chemical exposures. For example, while there is no OEL for HDI, NIOSH has a REL of 5 ppb for an 8-hour TWA and a ceiling limit of 20 ppb, consistent with the recommendations for MDI.[49]
| Organization (region) | Standard | Value |
|---|---|---|
| OSHA (USA) | Ceiling limit | 20 ppb[50] |
| NIOSH (USA) | Recommended exposure limit (REL) – ceiling limit | 20 ppb[51] |
| NIOSH (USA) | Recommended exposure limit (REL) – TWA | 5 ppb[51] |
| ACGIH (USA) | Threshold limit value (TLV) | 5 ppb[52] |
| Safe Work (Australia) | All isocyanates – TWA | 0.02 mg/m3[53] (approximately 2.5 ppb for comparison) |
| Safe Work (Australia) | All isocyanates – STEL | 0.07 mg/m3[53] (approximately 10 ppb for comparison) |
| Heath & Safety Executive (UK) | All isocyanates – TWA | 0.02 mg/m3[54] |
| Heath & Safety Executive (UK) | All isocyanates – STEL | 0.07 mg/m3[54] |
| Organization (region) | Standard | Value |
|---|---|---|
| OSHA (USA) | Ceiling limit | 20 ppb[50] |
| NIOSH (USA) | Recommended exposure limit (REL) | [none][55] |
| ACGIH (USA) | Threshold limit value (TLV) | 5 ppb[52] |
| ACGIH (USA) | Ceiling limit | 20 ppb[52] |
Regulation
United States
The Occupational Safety and Health Administration (OSHA) is the regulatory body covering worker safety. OSHA puts forth permissible exposure limit (PEL) 20 ppb for MDI and detailed technical guidance on exposure assessment.[45]
The National Institutes of Health (NIOSH) is the agency responsible for providing the research and recommendations regarding workplace safety, while OSHA is more of an enforcement body. NIOSH is responsible for producing the science that can result in recommended exposure limits (REL), which can be lower than the PEL. OSHA is tasked with enforcement and defending the enforceable limits (PELs). In 1992, when OSHA reduced the PEL for TDI to the NIOSH REL, the PEL reduction was challenged in court, and the reduction was reversed.[56]
The Environmental Protection Agency (EPA) is also involved in the regulation of isocyanates with regard to the environment and also non-worker persons that might be exposed.[57]
The American Conference of Governmental Industrial Hygienists (ACGIH) is a non-government organization that publishes guidance known as threshold limit values (TLV)[56] for chemicals based research as constant work exposure level without ill-effect[clarify]. The TLV is not an OSHA-enforceable value, unless the PEL is the same.
European Union
The European Chemicals Agency (ECHA) provides regulatory oversight of chemicals used within the European Union.[58] ECHA has been implementing policy aimed at limiting worker exposure through elimination by lower allowable concentrations in products and mandatory worker training, an administrative control.[59] Within the European Union, many nations set their own occupational exposure limits for isocyanates.
International groups
The United Nations , through the World Health Organization (WHO) together with the International Labour Organization (ILO) and United Nations Environment Programme (UNEP), collaborate on the International Programme on Chemical Safety (IPCS) to publish summary documents on chemicals. The IPCS published one such document in 2000 summarizing the status of scientific knowledge on MDI.[60]
The IARC evaluates the hazard data on chemicals and assigns a rating on the risk of carcinogenesis. In the case of TDI, the final evaluation is possibly carcinogenic to humans (Group 2B).[61] For MDI, the final evaluation is not classifiable as to its carcinogenicity to humans (Group 3).[62]
The International Isocyanate Institute is an international industry consortium that seeks promote the safe utilization of isocyanates by promulgating best practices.[63]
See also
References
- ↑ 1.0 1.1 1.2 Christian Six; Frank Richter (2005). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a14_611.
- ↑ Saul Patai, ed (1977). Cyanates and Their Thio Derivatives: Part 1, Volume 1. PATAI'S Chemistry of Functional Groups. doi:10.1002/9780470771525. ISBN 978-0-470-77152-5.
- ↑ Saul Patai, ed (1977). Cyanates and Their Thio Derivatives: Part 2, Volume 2. PATAI'S Chemistry of Functional Groups. doi:10.1002/9780470771532. ISBN 978-0-470-77153-2.
- ↑ Byrn, Marianne P.; Curtis, Carol J.; Hsiou, Yu; Khan, Saeed I.; Sawin, Philip A.; Tendick, S. Kathleen; Terzis, Aris; Strouse, Charles E. (1993). "Porphyrin sponges: conservative of host structure in over 200 porphyrin-based lattice clathrates". Journal of the American Chemical Society (American Chemical Society (ACS)) 115 (21): 9480–9497. doi:10.1021/ja00074a013. ISSN 0002-7863.
- ↑ Reinhard Richter, Henri Ulrich (1977). "Syntheses and Preparative Applications of Isocyanates". in Saul Patai. Cyanates and Their Thio Derivatives: Part 2, Volume 2. PATAI'S Chemistry of Functional Groups. pp. 619–818. doi:10.1002/9780470771532.ch1. ISBN 978-0-470-77153-2.
- ↑ Li, Zhen; Mayer, Robert J.; Ofial, Armin R.; Mayr, Herbert (2020-04-27). "From Carbodiimides to Carbon Dioxide: Quantification of the Electrophilic Reactivities of Heteroallenes". Journal of the American Chemical Society 142 (18): 8383–8402. doi:10.1021/jacs.0c01960. PMID 32338511.
- ↑ Coleman, M.M.; Painter, P. (2019). Fundamentals of Polymer Science: An Introductory Text, Second Edition. CRC Press. p. 39. ISBN 978-1-351-44639-6. https://books.google.com/books?id=VvSDDwAAQBAJ.
- ↑ Campbell, T. W.; Monagle, J. J. (1963). "Diphenylcarbodiimide". Organic Syntheses 43: 31. doi:10.15227/orgsyn.043.0031.
- ↑ http://alpha.chem.umb.edu/chemistry/orgchem/CH20Handout.pdf , Ch20Handout, University of Massachusetts Boston
- ↑ Mann, F. G.; Saunders, B. C. (1960). Practical Organic Chemistry, 4th Ed.. London: Longman. p. 128. ISBN 978-0-582-44407-2. https://www.scribd.com/doc/46973684/Practical-Organic-Chemistry-Frederick-George-Mann.
- ↑ Cohen, Julius (1900). Practical Organic Chemistry 2nd Ed.. London: Macmillan and Co., Limited. p. 72. https://archive.org/details/practicalorgani00cohegoog. "Practical Organic Chemistry Cohen Julius."
- ↑ Baumgarten, Henry; Smith, Howard; Staklis, Andris (1975). "Reactions of amines. XVIII. Oxidative rearrangement of amides with lead tetraacetate". The Journal of Organic Chemistry 40 (24): 3554–3561. doi:10.1021/jo00912a019.
- ↑ Randall, D. (2002). The Polyurethanes Book. Wiley. ISBN 978-0-470-85041-1.
- ↑ US EPA, OCSPP (2015-08-14). "Chemicals and Production of Spray Polyurethane Foam – Why It Matters" (in en). https://www.epa.gov/saferchoice/chemicals-and-production-spray-polyurethane-foam-why-it-matters.
- ↑ Allport D. C., Gilbert, D. S. and Outterside S. M. (eds) (2003). MDI and TDI: safety, health & the environment: a source book and practical guide. Chichester, Wiley.
- ↑ http://www.hse.gov.uk/pUbns/priced/eh40.pdf [bare URL PDF]
- ↑ "Isocyanates – Controlling hazardous substances – Managing occupational health risks in construction". http://www.hse.gov.uk/construction/healthrisks/hazardous-substances/isocyanates.htm.
- ↑ 18.0 18.1 "CDC – Isocyanates – NIOSH Workplace Safety and Health Topic" (in en-us). 2018-11-09. https://www.cdc.gov/niosh/topics/isocyanates/default.html.
- ↑ 19.0 19.1 "Isocyanate Exposure, Reaction and Protection – Quick Tips #233 – Grainger Industrial Supply". https://www.grainger.com/content/qt-233-isocyanate.
- ↑ "OSHA announces new National Emphasis Program for occupational exposure to isocyanates" (in en). Occupational Safety and Health Administration. https://www.osha.gov/news/newsreleases/trade/06252013.
- ↑ Riedlich, C. (2010). "Risk of isocyanate exposure in the construction industry". CPWR – the Center for Construction Research and Training: 1–8. http://www.cpwr.com/sites/default/files/publications/Yale%20CPWR%20Small%20Study%20Final%20Report%206-21-2010.pdf.
- ↑ IXOM. "Safety Data Sheet – TOLUENE DIISOCYANATE (TDI)". http://msds.orica.com/pdf/shess-en-cds-010-000030115101.pdf.
- ↑ 23.0 23.1 "Health Effects of Diisocyanates: Guidance for Medical Personnel". American Chemistry Council. https://polyurethane.americanchemistry.com/Resources-and-Document-Library/Health-Effects-of-Diisocyanates-Guidance-for-Medical-Personnel.pdf.
- ↑ "SAFETY DATA SHEET". Everchem. https://everchem.com/wp-content/uploads/2016/06/Everchem-MDI-CD-Modified-SDS.pdf.
- ↑ 25.0 25.1 Reeb-Whitaker, Carolyn; Whittaker, Stephen G.; Ceballos, Diana M.; Weiland, Elisa C.; Flack, Sheila L.; Fent, Kenneth W.; Thomasen, Jennifer M.; Trelles Gaines, Linda G. et al. (2012). "Airborne Isocyanate Exposures in the Collision Repair Industry and a Comparison to Occupational Exposure Limits". Journal of Occupational and Environmental Hygiene 9 (5): 329–339. doi:10.1080/15459624.2012.672871. PMID 22500941.
- ↑ 26.0 26.1 "Preventing Asthma and Death from MDI Exposure During Spray-on Truck Bed Liner and Related Applications". https://www.cdc.gov/niosh/docs/2006-149/pdfs/2006-149.pdf.
- ↑ Bogaert, Pieter; Tournoy, Kurt G.; Naessens, Thomas; Grooten, Johan (January 2009). "Where Asthma and Hypersensitivity Pneumonitis Meet and Differ". The American Journal of Pathology 174 (1): 3–13. doi:10.2353/ajpath.2009.071151. ISSN 0002-9440. PMID 19074616.
- ↑ Kimber, Ian; Dearman, Rebecca J.; Basketter, David A. (2014-07-25). "Diisocyanates, occupational asthma and IgE antibody: implications for hazard characterization" (in en). Journal of Applied Toxicology 34 (10): 1073–1077. doi:10.1002/jat.3041. ISSN 0260-437X. PMID 25059672.
- ↑ OSHA. "Do You Have Work-Related Asthma? A Guide for YOU and YOUR DOCTOR". https://www.osha.gov/Publications/OSHA3707.pdf.
- ↑ Daniels, Robert D. (2018-02-01). "Occupational asthma risk from exposures to toluene diisocyanate: A review and risk assessment" (in en). American Journal of Industrial Medicine 61 (4): 282–292. doi:10.1002/ajim.22815. ISSN 0271-3586. PMID 29389014.
- ↑ Bello, Dhimiter; Herrick, Christina A.; Smith, Thomas J.; Woskie, Susan R.; Streicher, Robert P.; Cullen, Mark R.; Liu, Youcheng; Redlich, Carrie A. (2006-11-28). "Skin Exposure to Isocyanates: Reasons for Concern" (in en). Environmental Health Perspectives 115 (3): 328–335. doi:10.1289/ehp.9557. ISSN 0091-6765. PMID 17431479.
- ↑ Ceballos, Diana M.; Fent, Kenneth W.; Whittaker, Stephen G.; Gaines, Linda G. T.; Thomasen, Jennifer M.; Flack, Sheila L.; Nylander-French, Leena A.; Yost, Michael G. et al. (2011-08-10). "Survey of Dermal Protection in Washington State Collision Repair Industry" (in en). Journal of Occupational and Environmental Hygiene 8 (9): 551–560. doi:10.1080/15459624.2011.602623. ISSN 1545-9624. PMID 21830873.
- ↑ Pubchem. "Methyl isocyanate" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/methyl_isocyanate#section=Safety-and-Hazards.
- ↑ ISOPA. "Dealing with fires involving MDI and TDI". https://www.isopa.org/media/1613/fs-dealing-with-fires-involving-mditdi-_final_.pdf.
- ↑ Weiss-Hills, Samantha (2018-05-28). "Is Your Couch Poisoning You?" (in en). https://www.architecturaldigest.com/story/flame-retardants-is-your-couch-poisoning-you.
- ↑ "Finding a substitute for methyl isocyanate" (in en). Chemistry World. https://www.chemistryworld.com/news/finding-a-substitute-for-methyl-isocyanate/5600.article.
- ↑ "Final Report | Isocyanate-Free Polyurethane Coatings | Research Project Database | Grantee Research Project | ORD | US EPA" (in en). https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.highlight/abstract/10482/report/F.
- ↑ 38.0 38.1 "Isocyanates – Controlling hazardous substances – Managing occupational health risks in construction" (in en-GB). http://www.hse.gov.uk/construction/healthrisks/hazardous-substances/isocyanates.htm.
- ↑ "Safety and Health Topics | Isocyanates – Additional Resources" (in en-us). Occupational Safety and Health Administration. https://www.osha.gov/SLTC/isocyanates/additionalinformation.html.
- ↑ American Chemistry Council. "Guidance for Selection of Protective Clothing for MDI Users". https://polyurethane.americanchemistry.com/Resources-and-Document-Library/Guidance-for-the-Selection-of-Protective-Clothing-for-MDI-Users.pdf.
- ↑ Ceballos, Diana; Reeb-Whitaker, Carolyn; Glazer, Patricia; Murphy-Robinson, Helen; Yost, Michael (2014-03-28). "Understanding Factors That Influence Protective Glove Use Among Automotive Spray Painters" (in en). Journal of Occupational and Environmental Hygiene 11 (5): 306–313. doi:10.1080/15459624.2013.862592. ISSN 1545-9624. PMID 24215135.
- ↑ "Chemical Resistant Gloves > Painters and Repairers Education Program | Internal Medicine" (in en). Yale School of Medicine. https://medicine.yale.edu/intmed/prep/worker/protection/gloves.aspx.
- ↑ "Sampling and Analytical Methods | Methylene Bisphenyl Isocyanate (MDI) – (Organic Method #047)" (in en-us). Occupational Safety and Health Administration. https://www.osha.gov/dts/sltc/methods/organic/org047/org047.html.
- ↑ Hu, Jimmy; Cantrell, Phillip; Nand, Aklesh (2017-07-29). "Comprehensive Biological Monitoring to Assess Isocyanates and Solvents Exposure in the NSW Australia Motor Vehicle Repair Industry" (in en). Annals of Work Exposures and Health 61 (8): 1015–1023. doi:10.1093/annweh/wxx064. ISSN 2398-7308. PMID 29028250.
- ↑ 45.0 45.1 "MEDICAL SURVEILLANCE PROCEDURES MANUAL AND MEDICAL MATRIX (EDITION 11)". Navy And Marine Corps Public Health Center. https://www.public.navy.mil/surfor/Documents/6260_NMCPHC_TM.pdf.
- ↑ Tarlo, S. M.; Liss, G. M.; Yeung, K. S. (2002-01-01). "Changes in rates and severity of compensation claims for asthma due to diisocyanates: a possible effect of medical surveillance measures" (in en). Occupational and Environmental Medicine 59 (1): 58–62. doi:10.1136/oem.59.1.58. ISSN 1351-0711. PMID 11836470.
- ↑ Rose, Dale A.; Murthy, Shivani; Brooks, Jennifer; Bryant, Jeffrey (2017-09-11). "The Evolution of Public Health Emergency Management as a Field of Practice" (in en). American Journal of Public Health 107 (S2): S126–S133. doi:10.2105/ajph.2017.303947. ISSN 0090-0036. PMID 28892444.
- ↑ "CSB Issues Report on 2008 Bayer CropScience Explosion: Finds Multiple Deficiencies Led to Runaway Chemical Reaction; Recommends State Create Chemical Plant Oversight Regulation" (in en). CSB. https://www.csb.gov/csb-issues-report-on-2008-bayer-cropscience-explosion-finds-multiple-deficiencies-led-to-runaway-chemical-reaction-recommends-state-create-chemical-plant-oversight-regulation/.
- ↑ "CDC – NIOSH Pocket Guide to Chemical Hazards – Hexamethylene diisocyanate". https://www.cdc.gov/niosh/npg/npgd0320.html.
- ↑ 50.0 50.1 "1910.1000 TABLE Z-1 Limits for Air Contaminants" (in en). Occupational Safety and Health Administration. https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.1000TABLEZ1.
- ↑ 51.0 51.1 "Methylene bisphenyl isocyanate". CDC. https://www.cdc.gov/niosh/npg/npgd0413.html.
- ↑ 52.0 52.1 52.2 Allport, D. C.; Gilbert, D. S.; Outterside, S. M. (2003). MDI and TDI: Safety, Health and the Environment. England: John Wiley & Sons, LTD. p. 346. ISBN 978-0-471-95812-3.
- ↑ 53.0 53.1 Safe Work Australia. "Guide to handling isocyanates". https://www.safeworkaustralia.gov.au/system/files/documents/1702/guide-to-handling-isocyanates.pdf.
- ↑ 54.0 54.1 HSE (2018). EH40/2005 Workplace exposure limits. United Kingdom: The Stationery Office. p. 17. ISBN 978-0-7176-6703-1.
- ↑ "Toluene-2,4-diisocyanate". CDC. https://www.cdc.gov/niosh/npg/npgd0621.html.
- ↑ 56.0 56.1 (in en-us) Request for assistance in preventing asthma and death from diisocyanate exposure. 1996-03-01. doi:10.26616/NIOSHPUB96111. https://www.cdc.gov/niosh/docs/96-111/default.html.
- ↑ US EPA, OCSPP, OPPT, EETD (2015-06-06). "Spray Polyurethane Foam (SPF) Insulation and How to Use it More Safely" (in en). https://www.epa.gov/saferchoice/spray-polyurethane-foam-spf-insulation-and-how-use-it-more-safely.
- ↑ "Restriction proposal on diisocyanates and several authorisation applications agreed by RAC and SEAC" (in en-GB). ECHA. https://echa.europa.eu/-/restriction-proposal-on-diisocyanates-and-several-authorisation-applications-agreed-by-rac-and-seac.
- ↑ Vincentz Network GmbH & Co. KG. "Proposed restriction of diisocyanates" (in en-GB). https://www.european-coatings.com/Raw-materials-technologies/Proposed-restriction-of-diisocyanates.
- ↑ Sekizawa J., Greenberg M. M. (2000). "Concise International Chemical Assessment Document 27: DIPHENYLMETHANE DIISOCYANATE (MDI)". https://www.who.int/ipcs/publications/cicad/en/cicad27.pdf?ua=1.
- ↑ "TOLUENE DIISOCYANATES". IARC. 1987. https://monographs.iarc.fr/wp-content/uploads/2018/06/mono71-37.pdf.
- ↑ "4,4′-METHYLENEDIPHENYL DIISOCYANATE AND POLYMERIC 4,4′-METHYLENEDIPHENYL DIISOCYANATE". https://monographs.iarc.fr/wp-content/uploads/2018/06/mono71-47.pdf.
- ↑ "Welcome to the International Isocyanate Institute". http://www.diisocyanates.org/.
External links
- NIOSH Safety and Health Topic: Isocyanates, from the website of the National Institute for Occupational Safety and Health (NIOSH)
- Health and Safety Executive, website of the UK Health and Safety Executive, useful search terms on this site — isocyanates, MVR, asthma
- International Isocyanate Institute | dii International Isocyanate Institute
- [web.archive.org/web/20170611085642/http://www.actsafe.ca/wp-content/uploads/resources/pdf/Isocyanates.pdf Safe Working Procedure for Isocyanate-Containing Products], June 200
- Isocyanates – Measurement Methodology, Exposure and Effects, Swedish National Institute for Working Life Workshop (1999)
- Health and Safety Executive, Guidance Note (EH16) Isocyanates: Toxic Hazards and Precautions (1984)
- The Society of the Plastics Industry – Technical Bulletin AX119 MDI-Based Polyurethane
- Foam Systems: Guidelines for Safe Handling and Disposal (1993)
- An occupational hygiene assessment of the use and control of isocyanates in the UK by Hilary A Cowie et al. HSE Research Report RR311/2005. Prepared by the Institute of Occupational Medicine for the Health and Safety Executive
 |