Chemistry:Fluorine azide
| |||
| Names | |||
|---|---|---|---|
| Other names
triazadienyl fluoride
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| FN 3 | |||
| Molar mass | 61.019 g/mol | ||
| Appearance | Yellow-green gas | ||
| Melting point | −139 °C (−218 °F; 134 K) | ||
| Boiling point | −30 °C (−22 °F; 243 K) | ||
| Explosive data | |||
| Shock sensitivity | Extreme | ||
| Friction sensitivity | Extreme | ||
| Hazards | |||
| Main hazards | Extremely sensitive explosive | ||
| NFPA 704 (fire diamond) | |||
| Related compounds | |||
Other cations
|
Hydrazoic acid Chlorine azide Bromine azide Iodine azide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
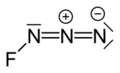
Fluorine azide or triazadienyl fluoride is a yellow green gas composed of nitrogen and fluorine with formula FN
3.[1] Its properties resemble those of ClN
3, BrN
3, and IN
3.[2] The bond between the fluorine atom and the nitrogen is very weak, leading to this substance being very unstable and prone to explosion.[3] Calculations show the F–N–N angle to be around 102° with a straight line of 3 nitrogen atoms.[4]
The gas boils at –30° and melts at –139 °C.[5]
It was first made by John F. Haller in 1942.[6]
Reactions
Fluorine azide can be made by reacting hydrazoic acid or sodium azide, with fluorine gas.[5][7]
- HN
3 + F
2 → N
3F + HF - NaN
3 + F
2 → N
3F + NaF
Fluorine azide decomposes without explosion at normal temperatures to make dinitrogen difluoride:
- 2 FN
3 → N
2F
2 + 2 N
2.[1]
At higher temperatures such as 1000 °C fluorine azide breaks up into nitrogen monofluoride radical:[7]
- FN
3 → NF + N
2
The FN itself dimerizes on cooling.
- 2 NF → N
2F
2
Solid or liquid FN
3 can explode, releasing a large amount of energy. A thin film burns at the rate of 1.6 km/s.[8] Due to the explosion hazard, only very small quantities of this substance should be handled at a time.[9]
FN
3 adducts can be formed with the Lewis acids boron trifluoride (BF
3) and arsenic pentafluoride (AsF
5) at -196 °C. These molecules bond with the first nitrogen atom from the fluorine.[10]
Properties
Spectroscopy
| Parameter | Value[9] | Unit |
| A | 48131.448 | MHz |
| B | 5713.266 | MHz |
| C | 5095.276 | MHz |
| μa | 1.1 | |
| μb | 0.7 |
Shape
Distances between atoms are F–N 0.1444 nm, FN=NN 0.1253 nm and FNN=N 0.1132 nm.[9]
Physical
FN
3 has a density of 1.3 g/cm3.[11]
FN
3 adsorbs on to solid surfaces of potassium fluoride, but not onto lithium fluoride or sodium fluoride. This property was being investigated so that FN
3 could boost the energy of solid propellants.[11]
The ultraviolet photoelectric spectrum shows ionisation peaks at 11.01, 13,72, 15.6, 15.9, 16.67, 18.2, and 19.7 eV. Respectively these are assigned to the orbitals: π, nN or nF, nF, πF, nN or σ, π and σ.[3]
References
- ↑ 1.0 1.1 Gipstein, Edward; John F. Haller (1966). "Absorption Spectrum of Fluorine Azide". Applied Spectroscopy 20 (6): 417–418. doi:10.1366/000370266774386470. ISSN 0003-7028. Bibcode: 1966ApSpe..20..417G.
- ↑ Saxena, P. B. (2007-01-01). Chemistry of Interhalogen Compounds. Discovery Publishing House. p. 96. ISBN 9788183562430. https://books.google.com/books?id=nvatWdX1ZWcC&pg=PA96. Retrieved 16 June 2014.
- ↑ 3.0 3.1 Rademacher, Paul; Andreas J. Bittner; Gabriele Schatte; Helge Willner (1988). "Photoelectron Spectrum and Electronic Structure of Triazadienyl Fluoride, N3F". Chemische Berichte 121 (3): 555–557. doi:10.1002/cber.19881210325. ISSN 0009-2940.
- ↑ Peters, Nancy J. S.; Leland C. Allen; Raymond A. Firestone (1988). "Fluorine azide and fluorine nitrate: structure and bonding". Inorganic Chemistry 27 (4): 755–758. doi:10.1021/ic00277a035. ISSN 0020-1669.
- ↑ 5.0 5.1 Gholivand, Khodayar; Gabriele Schatte; Helge Willner (1987). "Properties of triazadienyl fluoride, N3F". Inorganic Chemistry 26 (13): 2137–2140. doi:10.1021/ic00260a025. ISSN 0020-1669.
- ↑ Lowe, Derek (21 October 2008). "Things I Won't Work With: Triazadienyl Fluoride.". In the Pipeline. https://www.science.org/content/blog-post/things-i-won-t-work-triazadienyl-fluoride.
- ↑ 7.0 7.1 Benard, D. J.; B. K. Winker; T. A. Seder; R. H. Cohn (1989). "Production of nitrogen monofluoride (a1Δ) by dissociation of fluorine azide". The Journal of Physical Chemistry 93 (12): 4790–4796. doi:10.1021/j100349a022. ISSN 0022-3654.
- ↑ Seder, T.A.; D.J. Benard (1991). "The decomposition of condensed phase fluorine azide". Combustion and Flame 85 (3–4): 353–362. doi:10.1016/0010-2180(91)90139-3. ISSN 0010-2180. Bibcode: 1991CoFl...85..353S.
- ↑ 9.0 9.1 9.2 Christen, Dines.; H. G. Mack; G. Schatte; H. Willner (1988). "Structure of triazadienyl fluoride, FN3, by microwave, infrared, and ab initio methods". Journal of the American Chemical Society 110 (3): 707–712. doi:10.1021/ja00211a007. ISSN 0002-7863. Bibcode: 1988JAChS.110..707C.
- ↑ Schatte, G.; H. Willner (1991). "Die Wechselwirkung von N3F mit Lewis-Säuren und HF. N3F als möglicher Vorläufer für die Synthese von N3+-Salzen = The interaction of N3F with Lewis acids and HF•N3F as possible precursor for the synthesis of N3+ salts" (in de). Zeitschrift für Naturforschung B 46 (4): 483–489. doi:10.1515/znb-1991-0410. ISSN 0932-0776.
- ↑ 11.0 11.1 Brener, Nathan E.; Kestner, Neil R.; Callaway, Joseph (December 1990). Theoretical Studies of Highly Energetic CBES Materials: Final Report for the Period 2 March 1987 to 31 May 1987. Louisiana State University, Department of Physics and Astronomy. pp. 21–27. http://apps.dtic.mil/dtic/tr/fulltext/u2/a231340.pdf. Retrieved 25 June 2014.
External links
Salts and covalent derivatives of the azide ion
| |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HN3 | He | ||||||||||||||||||
| LiN3 | Be(N3)2 | B(N3)3 | CH3N3, C(N3)4 |
N(N3)3,H2N—N3 | O | FN3 | Ne | ||||||||||||
| NaN3 | Mg(N3)2 | Al(N3)3 | Si(N3)4 | P | SO2(N3)2 | ClN3 | Ar | ||||||||||||
| KN3 | Ca(N3)2 | Sc(N3)3 | Ti(N3)4 | VO(N3)3 | Cr(N3)3, CrO2(N3)2 |
Mn(N3)2 | Fe(N3)3 | Co(N3)2, Co(N3)3 |
Ni(N3)2 | CuN3, Cu(N3)2 |
Zn(N3)2 | Ga(N3)3 | Ge | As | Se(N3)4 | BrN3 | Kr | ||
| RbN3 | Sr(N3)2 | Y | Zr(N3)4 | Nb | Mo | Tc | Ru(N3)63− | Rh(N3)63− | Pd(N3)2 | AgN3 | Cd(N3)2 | In | Sn | Sb | Te | IN3 | Xe(N3)2 | ||
| CsN3 | Ba(N3)2 | Hf | Ta | W | Re | Os | Ir(N3)63− | Pt(N3)62− | Au(N3)4− | Hg2(N3)2, Hg(N3)2 |
TlN3 | Pb(N3)2 | Bi(N3)3 |
Po | At | Rn | |||
| Fr | Ra(N3)2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||
| ↓ | |||||||||||||||||||
| La | Ce(N3)3, Ce(N3)4 |
Pr | Nd | Pm | Sm | Eu | Gd(N3)3 | Tb | Dy | Ho | Er | Tm | Yb | Lu | |||||
| Ac | Th | Pa | UO2(N3)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||||
 |