Biology:Saccharopine dehydrogenase
In molecular biology, the protein domain Saccharopine dehydrogenase (SDH), also named Saccharopine reductase, is an enzyme involved in the metabolism of the amino acid lysine, via an intermediate substance called saccharopine. The Saccharopine dehydrogenase enzyme can be classified under EC 1.5.1.7, EC 1.5.1.8, EC 1.5.1.9, and EC 1.5.1.10. It has an important function in lysine metabolism and catalyses a reaction in the alpha-Aminoadipic acid pathway. This pathway is unique to fungal organisms therefore, this molecule could be useful in the search for new antibiotics. This protein family also includes saccharopine dehydrogenase and homospermidine synthase. It is found in prokaryotes, eukaryotes and archaea.
Function
Simplistically, SDH uses NAD+ as an oxidant to catalyse the reversible pyridine nucleotide dependent oxidative deamination of the substrate, Saccharopine, in order to form the products, lysine and alpha-ketoglutarate. This can be described by the following equation:[1]
- SDH
Saccharopine ⇌ lysine + alpha-ketoglutarate
Saccharopine dehydrogenase EC catalyses the condensation to of l-alpha-aminoadipate-delta-semialdehyde (AASA) with l-glutamate to give an imine, which is reduced by NADPH to give saccharopine.[2] In some organisms this enzyme is found as a bifunctional polypeptide with lysine ketoglutarate reductase (PF).
Homospermidine synthase proteins (EC). Homospermidine synthase (HSS) catalyses the synthesis of the polyamine homospermidine from 2 mol putrescine in an NAD+-dependent reaction.[3]
Structure
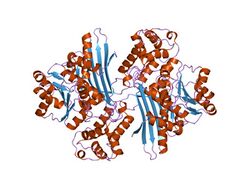
There appears to be two protein domains of similar size. One domain is a Rossmann fold that binds NAD+/NADH, and the other is relatively similar. Both domains contain a six-stranded parallel beta-sheet surrounded by alpha-helices and loops (alpha/beta fold).[4]
Clinical significance
Deficiencies are associated with hyperlysinemia.
References
- ↑ "Supporting role of lysine 13 and glutamate 16 in the acid-base mechanism of saccharopine dehydrogenase from Saccharomyces cerevisiae". Archives of Biochemistry and Biophysics 522 (1): 57–61. June 2012. doi:10.1016/j.abb.2012.03.027. PMID 22521736.
- ↑ "Chemical mechanism of saccharopine reductase from Saccharomyces cerevisiae". Biochemistry 48 (25): 5899–907. June 2009. doi:10.1021/bi900599s. PMID 19449898.
- ↑ "Purification, molecular cloning and expression in Escherichia coli of homospermidine synthase from Rhodopseudomonas viridis". European Journal of Biochemistry 240 (2): 373–9. September 1996. doi:10.1111/j.1432-1033.1996.0373h.x. PMID 8841401.
- ↑ "Crystal structures of ligand-bound saccharopine dehydrogenase from Saccharomyces cerevisiae". Biochemistry 46 (44): 12512–21. November 2007. doi:10.1021/bi701428m. PMID 17939687.
- Saccharopine+Dehydrogenases at the US National Library of Medicine Medical Subject Headings (MeSH)
 |