Chemistry:Beloranib
| |
| Names | |
|---|---|
| Preferred IUPAC name
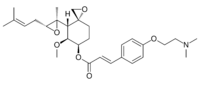
(3R,4S,5S,6R)-5-Methoxy-4-[(2R,3R)-2-methyl-3-(3-methylbut-2-en-1-yl)oxiran-2-yl]-1-oxaspiro[2.5]octan-6-yl (2E)-3-{4-[2-(dimethylamino)ethoxy]phenyl}prop-2-enoate | |
| Other names
CKD-732; ZGN-433
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C29H41NO6 | |
| Molar mass | 499.648 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Beloranib is a former drug candidate for the treatment of obesity. It was discovered by CKD Pharmaceuticals and its clinical development was led by Zafgen.[1] Drug development was halted in 2016 after deaths during clinical trials.[2]
Mechanism of action
Beloranib, an analog of the natural chemical compound fumagillin, is an inhibitor of the enzyme METAP2.[3] It was originally designed as angiogenesis inhibitor for the treatment of cancer.[4] However, once the potential anti-obesity effects of METAP2 inhibition became apparent, the clinical development began to focus on these effects and beloranib has shown positive results in preliminary clinical trials for this indication.[5]
Clinical trials
A Phase I trial was published in 2013,[6] finding a dose that led to weight loss in obese women in comparison to placebo. Results from a Phase II clinical trial for obesity were promising with clinically meaningful weight loss and improvements in cardiometabolic risk factors in the treated group.[7] Zafgen continued with a Phase III trial for Prader–Willi syndrome.[8]
In December 2015, Zafgen halted the Phase III clinical trial of beloranib for Prader–Willi syndrome after a second patient death in order to determine whether the deaths were treatment-related.[9] After discussions with the Food and Drug Administration indicated that the obstacles to gaining approval were insurmountable, product development for beloranib was ended.[2]
References
- ↑ "News Release: Zafgen Secures $33 Million Series C Financing". Zafgen, Inc.. July 7, 2011. http://www.zafgen.com/PDF/zafgen-series-c-070711.pdf.
- ↑ 2.0 2.1 "Zafgen Halts Development of Beloranib, to Cut Jobs by ~34%". nasdaq.com. July 20, 2016. http://www.nasdaq.com/article/zafgen-halts-development-of-beloranib-to-cut-jobs-by-34-cm651992.
- ↑ "Novel inhibitors targeted to methionine aminopeptidase 2 (MetAP2) strongly inhibit the growth of cancers in xenografted nude model". International Journal of Cancer 114 (1): 124–30. March 2005. doi:10.1002/ijc.20687. PMID 15523682.
- ↑ "General pharmacology of CKD-732, a new anticancer agent: effects on central nervous, cardiovascular, and respiratory system". Biological & Pharmaceutical Bulletin 28 (2): 217–23. February 2005. doi:10.1248/bpb.28.217. PMID 15684472.
- ↑ "Zafgen Announces Positive Topline Phase 1b Data for ZGN-433 in Obesity". MedNews. Drugs.com. 5 January 2011. https://www.drugs.com/clinical_trials/zafgen-announces-positive-topline-phase-1b-data-zgn-433-obesity-10955.html.
- ↑ "Ascending dose-controlled trial of beloranib, a novel obesity treatment for safety, tolerability, and weight loss in obese women". Obesity 21 (9): 1782–8. September 2013. doi:10.1002/oby.20356. PMID 23512440.
- ↑ "Efficacy and safety of beloranib for weight loss in obese adults: a randomized controlled trial". Diabetes, Obesity & Metabolism 17 (6): 566–572. June 2015. doi:10.1111/dom.12457. PMID 25732625.
- ↑ "Clinical Trials". http://www.zafgen.com/zafgen/clinical-trials.
- ↑ "UPDATE 4-Zafgen halts obesity drug trial after second patient death". Reuters. 3 December 2015. https://www.reuters.com/article/zafgen-study-idUSL3N13R3OQ20151203.
 |
