Chemistry:Latamoxef
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Intramuscular, intravenous |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | 35 to 50% |
| Metabolism | Nil |
| Elimination half-life | 2 hours |
| Excretion | Mostly renal, unchanged; also biliary |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
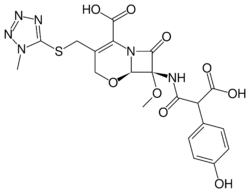
| Formula | C20H20N6O9S |
| Molar mass | 520.47 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 117 to 122 °C (243 to 252 °F) (dec.) |
| |
| |
| | |
Latamoxef (or moxalactam) is an oxacephem antibiotic usually grouped with the cephalosporins. In oxacephems such as latamoxef, the sulfur atom of the cephalosporin core is replaced with an oxygen atom.
Latamoxef has been associated with prolonged bleeding time, and several cases of coagulopathy, some fatal, were reported during the 1980s.[1][2] Latamoxef is no longer available in the United States. As with other cephalosporins with a methylthiotetrazole side chain, latamoxef causes a disulfiram reaction when mixed with alcohol. Additionally, the methylthiotetrazole side chain inhibits γ-carboxylation of glutamic acid; this can interfere with the actions of vitamin K.[citation needed]
It has been described as a third-generation cephalosporin.[3]
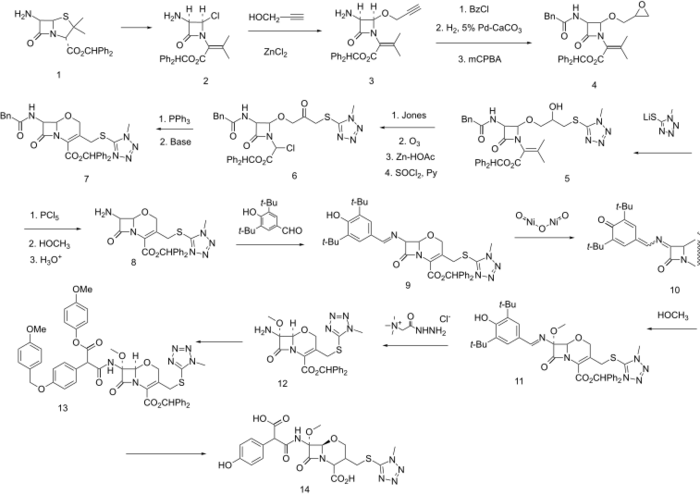
Synthesis
Oxa-substituted third generation cephalosporin antibiotic (oxacephalosporin).
References
- ↑ "Prolonged bleeding times and bleeding diathesis associated with moxalactam administration". JAMA 249 (1): 69–71. January 1983. doi:10.1001/jama.249.1.69. PMID 6217353.
- ↑ "Enhanced bleeding with cefoxitin or moxalactam. Statistical analysis within a defined population of 1493 patients". Archives of Internal Medicine 146 (11): 2159–2164. November 1986. doi:10.1001/archinte.146.11.2159. PMID 3778044.
- ↑ "The hematologic effects of latamoxef sodium when used as a prophylaxis during surgical treatment". Surgery, Gynecology & Obstetrics 164 (6): 525–529. June 1987. PMID 3296254.
- ↑ Nagata W, Narisada M, "1-Oxadethiacephalosporine, Verfahren zu Arzneimittel [1-oxadethiacephalosporins, procedure for medicinal products]", DE patent 2713370, published 1977-09-29
- ↑ Nagata W, Narisada M, US patent 4138486, issued 1979, assigned to Shionogi
- ↑ Synthetic studies on beta-lactam antibiotics. Part 101. Synthesis of <nowiki>7beta-[2-carboxy-2-(4-hydroxyphenyl)acetamido]-7alpha-methoxy-3-journal = Journal of Medicinal Chemistry. 22. July 1979. pp. 757–759. doi:10.1021/jm00193a001. PMID 448673.
- ↑ "Discovery and development of Moxalactam (6059-S): the chemistry and biology of 1-oxacephems". Medicinal Research Reviews 1 (3): 217–248. 1981. doi:10.1002/med.2610010302. PMID 6213825.
- ↑ "Synthetic Studies on b-Lactam Antibiotics. Part 5. A Synthesis of 7b-Acylamino-3-methyl-1-oxadethia-3-cephem-4-carboxylic Acids". Heterocycles 7 (2): 839. 1977. doi:10.3987/S-1977-02-0839.
 |
