Chemistry:Isoniazid
Isoniazid, also known as isonicotinic acid hydrazide (INH), is an antibiotic used for the treatment of tuberculosis. For active tuberculosis, it is often used together with rifampicin, pyrazinamide, and either streptomycin or ethambutol. It may also be used for atypical types of mycobacteria, such as M. avium, M. kansasii, and M. xenopi. It is usually taken by mouth, but may be used by injection into muscle.
Isoniazid is a prodrug that, when activated by catalase-peroxidase KatG, generates adducts and radicals that inhibits the formation of the mycobacterial cell wall. Side effects in those treated with isoniazid include vitamin B6 deficiency, liver toxicity, peripheral neuropathy, and a reduction in blood cell production. Mutations in the ahpC, inhA, kasA, katG, genes of M. tuberculosis may result in isoniazid resistance.
Although first synthesized in 1912, the anti-tuberculosis activity of isoniazid was not discovered until the 1940s. It is on the World Health Organization's List of Essential Medicines and is available as a generic medication.
Medical uses
Tuberculosis
The primary use of isoniazid is as first-line treatment for latent and active infection of Mycobacterium tuberculosis, the causative agent of tuberculosis (TB).[1] In persons with isoniazid-resistant Mycobacterium tuberculosis infection, drug regimens based on isoniazid have a high rate of failure.[2]
In the United States, the indications for isoniazid use approved by the U.S. Food and Drug Administration (FDA) include:.[1]
- Both active and latent TB
- Intravenous drug users with Mantoux test above 10 mm
- HIV-positive people with Mantoux test above 5 mm
- Those who had close contact with TB patient and have Mantoux test above 5 mm
- As a preventative measure for those with pulmonary silicosis or fibrotic pulmonary lesions from recovered TB
Isoniazid can be used alone or in combination with Rifampin for treatment of latent tuberculosis, or as part of a four-drug regimen..[1] The drug regimen typically requires daily or weekly oral administration for a period of three to nine months, often under Directly Observed Therapy (DOT) supervision.[3]
Non-tuberculous mycobacteria
Isoniazid may also be used off-label to treat nontuberculous mycobacterial pulmonary disease..[1] Isoniazid was widely used in the treatment of Mycobacterium avium complex as part of a regimen including rifampicin and ethambutol.[4] Evidence suggests that isoniazid prevents mycolic acid synthesis in M. avium complex as in M. tuberculosis[5] and although this is not bactericidal to M. avium complex, it greatly potentiates the effect of rifampicin.[6]
Special populations
It is recommended that women with active tuberculosis who are pregnant or breastfeeding take isoniazid. Preventive therapy should be delayed until after giving birth.[7] Nursing mothers excrete a relatively low and non-toxic concentration of INH in breast milk, and their babies are at low risk for side effects. Both pregnant women and infants being breastfed by mothers taking INH should take vitamin B6 in its pyridoxine form to minimize the risk of peripheral nerve damage.[8] Vitamin B6 is used to prevent isoniazid-induced B6 deficiency and neuropathy in people with a risk factor, such as pregnancy, lactation, HIV infection, alcoholism, diabetes, kidney failure, or malnutrition.[9]
People with liver dysfunction are at a higher risk for hepatitis caused by INH, and may need a lower dose.[7] Levels of liver enzymes in the bloodstream should be frequently checked in daily alcohol drinkers, pregnant women, IV drug users, people over 35, and those who have chronic liver disease, severe kidney dysfunction, peripheral neuropathy, or HIV infection since they are more likely to develop hepatitis from INH.[7][10]
History
Synthesis

After Friedrich Raschig developed a method to synthesize hydrazine, Hans Meyer and his doctoral student at the German University in Prague Josef Mally researched hydrazides of pyridinecarboxylic acids. By reacting ethyl isonicotinate with hydrazine hydrate, they obtained a compound which, after recrystallization, possessed a melting point of 163°C.[11] Despite its publication in 1912, the compound's pharmaceutical properties were not investigated for decades.
In the 1940s, French physicians discovered that nicotinamide had some activity against tubercle bacilli in vitro and in infected guinea pigs.[12][13] At the same time, German chemists led by G. Domagk investigating sulfo drugs at Bayer developed thioacetazone.[14][13][15] After their findings were made public, in 1950 André Girard (chemist) (fr) modified it to the less toxic thiosemicarbazone of nicotinaldehyde[16] while H. H. Fox developed similar isonicotinaldehyde thiosemicarbazone.[17]
Discovery of anti-TB activity
Soon, multiple laboratories discovered anti-TB activity of isoniazid.[13][14] This led three pharmaceutical companies to unsuccessfully attempted to patent the drug at the same time,[18] the most prominent one being Roche in January 1951,[19] which launched its version, Rimifon, in 1952.[20] Additionally, Soviet physicians Anatoliy Kachugin (ru) and Bella Keyfman independently discovered this activity in 1949, but neither published their findings in a peer-reviewed article nor applied for an inventor's certificate.[21][22]
The drug was first tested at Many Farms, a Navajo community in Arizona, due to the Navajo reservation's tuberculosis problem and because the population had not previously been treated with streptomycin, the main tuberculosis treatment at the time.[23] The research was led by Walsh McDermott, an infectious disease researcher with an interest in public health, who had previously taken isoniazid to treat his own tuberculosis.[24]
Isoniazid and a related drug, iproniazid, were among the first drugs to be referred to as antidepressants.[25] Psychiatric use stopped in 1961 following reports of hepatotoxicity. Use against tuberculosis continued, as isoniazid's effectiveness against the disease outweighs its risks.[26]
Elucidation of mechanism of action
Seminal studies that uncovered the mechanism of action for isoniazid were largely performed in M. smegmatis, a model for the slow-growing M. tuberculosis.[27]: 1, 7 In 1992, Stewart Cole and colleagues discovered that isoniazid was active in resistant M. smegmatis only when KatG, a catalase-peroxidase, was expressed;[27]: 7 [28] KatG is now understood to be critical for the metabolism of the prodrug isoniazid into its active forms..[1][27]: 7
At the Albert Einstein College of Medicine, William R. Jacobs Jr. and coworkers discovered that inhA—which they also found to encode an NADH-specific enoyl-acyl carrier protein reductase—as isoniazid's primary target[29][27]: 7 The isoniazid-NAD adduct was also shown to bind and inhibit InhA, the protein product of inhA.[30]: 37
Modern usage
As part of standard TB chemotherapy, isoniazid is now typically administered alongside at least three other drugs—ethambutol, pyrazinamide, and rifampin—for six to nine months.[30]: 36
Isoniazid is also included on the World Health Organization's List of Essential Medicines.[31] The World Health Organization classifies isoniazid as critically important for human medicine.[32]
Adverse effects
Side effects
Up to 20% of people taking isoniazid experience peripheral neuropathy when taking daily doses of 6 mg/kg of body weight or higher.[33] Gastrointestinal reactions include nausea and vomiting.[34] Aplastic anemia, thrombocytopenia, and agranulocytosis due to lack of production of red blood cells, platelets, and white blood cells by the bone marrow respectively, can also occur. Hypersensitivity reactions are common and can present with a maculopapular rash and fever.[34] Gynecomastia may also occur.[3]
Isoniazid inhibits pyridoxine phosphokinase, which is responsible for maintaining the active form of vitamin B6, pyridoxal 5′-phosphate (P5P). Isoniazid is also associated with increased excretion of pyridoxine. Altogether, this means that isoniazid can cause pyridoxine (vitamin B6) deficiency, leading to peripheral neuropathy and (rarely) sideroblastic anemia via insufficient P5P provided to δ-aminolevulinic acid synthase.[9] It is recommended that isoniazid be taken with pyridoxine in persons at risk of peripheral neuropathy as well as those who have already developed peripheral neuropathy.[35]
Asymptomatic elevation of serum liver enzyme concentrations occurs in 10% to 20% of people taking INH, and liver enzyme concentrations usually return to normal even when treatment is continued.[36] Isoniazid has a boxed warning for severe and sometimes fatal hepatitis, which is age-dependent at a rate of 0.3% in people 21 to 35 years old and over 2% in those over age 50.[34][37] Symptoms suggestive of liver toxicity include nausea, vomiting, abdominal pain, dark urine, right upper quadrant pain, and loss of appetite. Black and Hispanic women are at higher risk for isoniazid-induced hepatotoxicity.[34] When it happens, isoniazid-induced liver toxicity has been shown to occur in 50% of patients within the first 2 months of therapy.[38]
Some recommend that liver function should be monitored carefully in all people receiving it,[7] but others recommend monitoring only in certain populations.[36][39][40] Headache, poor concentration, weight gain, poor memory, insomnia, and depression have all been associated with isoniazid use.[41][42] All patients and healthcare workers should be aware of these serious side effects, especially if suicidal ideation or behavior are suspected.[41][43][44]
Drug interactions
Isoniazid decreases the metabolism of carbamazepine, slowing down its clearance from the body. People taking carbamazepine should have their carbamazepine levels monitored and, if necessary, have their dose adjusted accordingly.[45] Isoniazid can also increase the amount of phenytoin in the body. The doses of phenytoin may need to be adjusted when given with isoniazid.[46][47] Isoniazid may increase the plasma levels of theophylline. There are some cases of theophylline slowing down isoniazid elimination. Both theophylline and isoniazid levels should be monitored.[48] Valproate levels may increase when taken with isoniazid. Valproate levels should be monitored and its dose adjusted if necessary.[46]
People taking isoniazid and acetaminophen are at risk of acetaminophen toxicity. Isoniazid is thought to induce a liver enzyme which causes a larger amount of acetaminophen to be metabolized to a toxic form.[49][50]
It is possible that isoniazid may decrease the serum levels of ketoconazole after long-term treatment. This is seen with the simultaneous use of rifampin, isoniazid, and ketoconazole.[51]
Mechanism of action
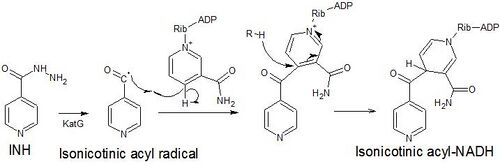
Isoniazid is a prodrug that inhibits the formation of the mycobacterial cell wall. Isoniazid must be activated by KatG, a bacterial catalase-peroxidase enzyme in Mycobacterium tuberculosis.[52] KatG catalyzes the formation of the isonicotinic acyl radical, which spontaneously couples with NADH to form the nicotinoyl-NAD adduct. This complex binds tightly to the enoyl-acyl carrier protein reductase InhA, thereby blocking the natural enoyl-AcpM substrate and the action of fatty acid synthase. This process inhibits the synthesis of mycolic acids, which are required components of the mycobacterial cell wall. A range of radicals are produced by KatG activation of isoniazid, including nitric oxide,[53] which has also been shown to be important in the action of another antimycobacterial prodrug pretomanid.[54]
Isoniazid is bactericidal to rapidly dividing mycobacteria, but is bacteriostatic if the mycobacteria are slow-growing.[55] It inhibits the cytochrome P450 system and hence acts as a source of free radicals.[56]
Isoniazid is a mild non-selective monoamine oxidase inhibitor (MAO-I).[57] It inhibits diamine oxidase more strongly. These two actions are possible explanations for its antidepressant action[58] as well as its ability to cause mania.[26]
Metabolism
Isoniazid reaches therapeutic concentrations in serum, cerebrospinal fluid, and within caseous granulomas. It is metabolized in the liver via acetylation into acetylhydrazine. Two forms of the enzyme are responsible for acetylation, so some patients metabolize the drug more quickly than others. Hence, the half-life is bimodal, with "slow acetylators" and "fast acetylators". A graph of number of people versus time shows peaks at one and three hours. The height of the peaks depends on the ethnicities of the people being tested. The metabolites are excreted in the urine. Doses do not usually have to be adjusted in case of renal failure.
Overdose
Isoniazid causes seizure on overdose due to a depletion of pyridoxal 5′-phosphate (P5P) preventing glutamic acid decarboxylase from making gamma aminobutyric acid (GABA). Ordinary pyridoxine is an effective antidote of this mechanism of toxicity.[59]
Preparation
Isoniazid is an isonicotinic acid derivative. It is manufactured using 4-cyanopyridine and hydrazine hydrate.[60] In another method, isoniazid was claimed to have been made from citric acid starting material.[61]
It can in theory be made from methyl isonicotinate, which is labelled a semiochemical.
Brand names
Brand names for isoniazid include Hydra, Hyzyd, Isovit, Laniazid, Nydrazid, Rimifon, and Stanozide.[62]
Other uses
Chromatography
Isonicotinic acid hydrazide is also used in chromatography to differentiate between various degrees of conjugation in organic compounds barring the ketone functional group.[63]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "Isoniazid". StatPearls [Internet].. Treasure Island (FL): StatPearls Publishing. 16 February 2024. https://www.ncbi.nlm.nih.gov/books/NBK557617/. Retrieved June 22, 2025.
- ↑ "Treatment of isoniazid-resistant tuberculosis with first-line drugs: a systematic review and meta-analysis". The Lancet. Infectious Diseases 17 (2): 223–234. February 2017. doi:10.1016/S1473-3099(16)30407-8. PMID 27865891.
- ↑ 3.0 3.1 Medical-surgical nursing: assessment and management of clinical problems (Ninth ed.). St. Louis, Missouri: Elsevier/Mosby. 5 December 2013. ISBN 978-0-323-10089-2. OCLC 228373703.
- ↑ *"First randomised trial of treatments for pulmonary disease caused by M avium intracellulare, M malmoense, and M xenopi in HIV negative patients: rifampicin, ethambutol and isoniazid versus rifampicin and ethambutol". Thorax 56 (3): 167–172. March 2001. doi:10.1136/thorax.56.3.167. PMID 11182006.
- ↑ "Mechanisms involved in the intrinsic isoniazid resistance of Mycobacterium avium". Molecular Microbiology 27 (6): 1223–1233. March 1998. doi:10.1046/j.1365-2958.1998.00774.x. PMID 9570407.
- ↑ "The pharmacokinetics and pharmacodynamics of pulmonary Mycobacterium avium complex disease treatment". American Journal of Respiratory and Critical Care Medicine 186 (6): 559–565. September 2012. doi:10.1164/rccm.201204-0682OC. PMID 22744719.
- ↑ 7.0 7.1 7.2 7.3 "Isoniazid tablet". 18 October 2018. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=1553312b-ed76-421c-a055-2579bdcf366c.
- ↑ "Drug treatment for tuberculosis during pregnancy: safety considerations". Drug Safety 24 (7): 553–565. 2001. doi:10.2165/00002018-200124070-00006. PMID 11444726.
- ↑ 9.0 9.1 "[Isoniazid induced neuropathy: consider prevention]". Revue des Maladies Respiratoires 23 (2 Pt 1): 157–160. April 2006. doi:10.1016/S0761-8425(06)71480-2. PMID 16788441.
- ↑ "An official ATS statement: hepatotoxicity of antituberculosis therapy". American Journal of Respiratory and Critical Care Medicine 174 (8): 935–952. October 2006. doi:10.1164/rccm.200510-1666ST. PMID 17021358.
- ↑ "Über Hydrazinderivate der Pyridincarbonsäuren" (in de). Monatshefte für Chemie und Verwandte Teile Anderer Wissenschaften 33 (4): 393–414. 1912. doi:10.1007/BF01517946. ISSN 1434-4475. https://link.springer.com/article/10.1007/BF01517946.
- ↑ "Qui a Découvert L'efficacité Des Nouveaux Médicaments Contre La Tuberculose?" (in fr). 1952-05-12. https://www.lemonde.fr/archives/article/1952/05/12/qui-a-decouvert-l-efficacite-des-nouveaux-medicaments-contre-la-tuberculose_1991993_1819218.html.
- ↑ 13.0 13.1 13.2 "Tuberculosis Drug Development: History and Evolution of the Mechanism-Based Paradigm". Cold Spring Harbor Perspectives in Medicine 5 (8). April 2015. doi:10.1101/cshperspect.a021147. PMID 25877396.
- ↑ 14.0 14.1 "The Story of INH". The Journal of Infectious Diseases 119 (6): 678–683. June 1969. doi:10.1093/infdis/119.6.678. PMID 4893894.
- ↑ (in en) Gerhard Domagk: The First Man to Triumph Over Infectious Diseases. LIT Verlag Münster. 2004. ISBN 978-3-8258-6164-3. https://books.google.com/books?id=sg1QAwAAQBAJ.
- ↑ Girard A, "Thiosemicarbazone de l'aldéhyde nicotinique et son procédé de préparation", FR patent 1055796, published 1954-02-22, issued 1950-08-17, assigned to Laboratoires Français de Chimiothérapie
- ↑ Herbert FH, US patent 2676178, issued 20 April 1954
- ↑ "Fourth-generation fluoroquinolones in tuberculosis". Lancet (London, England) 373 (9670): 1148–1149. April 2009. doi:10.1016/S0140-6736(09)60559-6. PMID 19345815.
- ↑ Herbert FH, "1-Isonicotinyl-2-aldosylhydrazine", US patent 2685580, issued 3 August 1954
- ↑ "History". Roche USA. http://www.rocheusa.com/about/history.html.
- ↑ "Забвение". Журнальный зал. 2003. https://magazines.gorky.media/neva/2003/3/zabvenie.html.
- ↑ "Метод Качугина" (in ru). Радио Свобода (Radio Liberty). 2011-05-24. https://www.svoboda.org/a/24196133.html.
- ↑ "The health care experiments at Many Farms: the Navajo, tuberculosis, and the limits of modern medicine, 1952-1962". Bulletin of the History of Medicine 76 (4): 749–790. 2002. doi:10.1353/bhm.2002.0186. PMID 12446978.
- ↑ "Walsh McDermott". Biographical Memoirs. 59. National Academies Press. 1990. pp. 282–307. doi:10.17226/1652. ISBN 978-0-309-04198-0. https://www.nap.edu/read/1652/chapter/15#283.
- ↑ "The creation of the concept of an antidepressant: an historical analysis". Social Science & Medicine 66 (11): 2346–2355. June 2008. doi:10.1016/j.socscimed.2008.01.047. PMID 18321627. https://discovery.ucl.ac.uk/id/eprint/10193706/.
- ↑ 26.0 26.1 "Isoniazid-induced mania and the history of antidepressant drugs: Case report and literature review". Bipolar Disorders 25 (1): 84–87. February 2023. doi:10.1111/bdi.13272. PMID 36380697.
- ↑ 27.0 27.1 27.2 27.3 "Mycobacterium smegmatis: The Vanguard of Mycobacterial Research". Journal of Bacteriology 205 (1): e0033722. January 2023. doi:10.1128/jb.00337-22. PMID 36598232.
- ↑ "The catalase-peroxidase gene and isoniazid resistance of Mycobacterium tuberculosis". Nature 358 (6387): 591–593. August 1992. doi:10.1038/358591a0. PMID 1501713. Bibcode: 1992Natur.358..591Z.
- ↑ "inhA, a gene encoding a target for isoniazid and ethionamide in Mycobacterium tuberculosis". Science (New York, N.Y.) 263 (5144): 227–230. 1994. doi:10.1126/science.8284673. PMID 8284673. Bibcode: 1994Sci...263..227B.
- ↑ 30.0 30.1 "The Mechanism of Isoniazid Killing: Clarity Through the Scope of Genetics". Annual Review of Microbiology 61: 35–50. 2007. doi:10.1146/annurev.micro.61.111606.122346. PMID 18035606.
- ↑ World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ Critically important antimicrobials for human medicine (6th revision ed.). Geneva: World Health Organization. 2019. ISBN 978-92-4-151552-8.
- ↑ Applied Therapeutics. Lippincott Williams & Wilkins. February 12, 2013. ISBN 978-1-60913-713-7.
- ↑ 34.0 34.1 34.2 34.3 "Isoniazid (package insert)". 30 March 2023. https://medlibrary.org/lib/rx/meds/isoniazid-10/.
- ↑ "Treatment for Drug-Susceptible Tuberculosis Disease" (in en-us). 17 April 2025. https://www.cdc.gov/tb/hcp/treatment/tuberculosis-disease.html.
- ↑ 36.0 36.1 "Latent Tuberculosis Infection: A Guide for Primary Health Care Providers". Center for Disease Control. https://www.cdc.gov/tb/publications/ltbi/treatment.htm.
- ↑ Katzung & Trevor's Pharmacology: examination & board review (10th ed.). New York: McGraw-Hill Medical, Lange. 2013. p. 417.
- ↑ "Isoniazid: An overview". UpToDate. 2015. http://www.uptodate.com/contents/isoniazid-an-overview.
- ↑ Treatment of Tuberculosis – Guidelines (4th ed.). World Health Organization. 2010. ISBN 978-92-4-154783-3. http://apps.who.int/iris/bitstream/10665/44165/1/9789241547833_eng.pdf?ua=1&ua=1. Retrieved 25 March 2016.
- ↑ "Chemotherapy and management of tuberculosis in the United Kingdom: recommendations 1998.". Thorax 53 (7): 536–548. July 1998. doi:10.1136/thx.53.7.536. PMID 9797751.
- ↑ 41.0 41.1 "Isoniazid-induced psychosis". The Annals of Pharmacotherapy 32 (9): 889–891. September 1998. doi:10.1345/aph.17377. PMID 9762376.
- ↑ "Acute Psychosis Secondary to Isoniazid in a Patient with Pulmonary Tuberculosis". Bengal Physician Journal 10 (2): 73–74. 4 August 2023. doi:10.5005/jp-journals-10070-8010.
- ↑ "Suicidal psychosis secondary to isoniazid". Pediatric Emergency Care 18 (1): 25–27. February 2002. doi:10.1097/00006565-200202000-00008. PMID 11862134.
- ↑ "Isoniazid-associated psychosis: case report and review of the literature". The Annals of Pharmacotherapy 27 (2): 167–170. February 1993. doi:10.1177/106002809302700205. PMID 8439690.
- ↑ "Interaction between carbamazepine and antituberculosis agents". Chest 99 (6): 1554. June 1991. doi:10.1378/chest.99.6.1554a. PMID 2036861.
- ↑ 46.0 46.1 "Interaction between isoniazid and valproate: a case of valproate overdosage". European Journal of Clinical Pharmacology 40 (2): 197–198. 1991. doi:10.1007/BF00280078. PMID 2065702.
- ↑ "Treatment of tuberculosis and tuberculosis infection in adults and children. American Thoracic Society and The Centers for Disease Control and Prevention". American Journal of Respiratory and Critical Care Medicine 149 (5): 1359–1374. May 1994. doi:10.1164/ajrccm.149.5.8173779. PMID 8173779.
- ↑ "Interaction between isoniazid and theophylline". European Journal of Respiratory Diseases 70 (2): 110–116. February 1987. PMID 3817069.
- ↑ "Severe acetaminophen toxicity in a patient receiving isoniazid". Annals of Internal Medicine 113 (10): 799–800. November 1990. doi:10.7326/0003-4819-113-10-799. PMID 2240884.
- ↑ "Isoniazid potentiation of acetaminophen hepatotoxicity in the rat and 4-methylpyrazole inhibition of it". Research Communications in Chemical Pathology and Pharmacology 69 (1): 115–118. July 1990. PMID 2218067.
- ↑ "Ketoconazole and fluconazole drug interactions". Archives of Internal Medicine 153 (17): 1970–1976. September 1993. doi:10.1001/archinte.153.17.1970. PMID 8357281.
- ↑ "An oxyferrous heme/protein-based radical intermediate is catalytically competent in the catalase reaction of Mycobacterium tuberculosis catalase-peroxidase (KatG)". The Journal of Biological Chemistry 284 (11): 7017–7029. March 2009. doi:10.1074/jbc.M808106200. PMID 19139099.
- ↑ "Nitric oxide generated from isoniazid activation by KatG: source of nitric oxide and activity against Mycobacterium tuberculosis". Antimicrobial Agents and Chemotherapy 48 (8): 3006–3009. August 2004. doi:10.1128/AAC.48.8.3006-3009.2004. PMID 15273113.
- ↑ "PA-824 kills nonreplicating Mycobacterium tuberculosis by intracellular NO release". Science (New York, N.Y.) 322 (5906): 1392–1395. November 2008. doi:10.1126/science.1164571. PMID 19039139. Bibcode: 2008Sci...322.1392S.
- ↑ "Biphasic kill curve of isoniazid reveals the presence of drug-tolerant, not drug-resistant, Mycobacterium tuberculosis in the guinea pig". The Journal of Infectious Diseases 200 (7): 1136–1143. October 2009. doi:10.1086/605605. PMID 19686043.
- ↑ Pharmacology. 864 (4th ed.). Lippincott Williams & Wilkins. 2006. ISBN 978-0-7817-4118-7.
- ↑ "Isoniazid and antidepressants: is there cause for concern?". International Clinical Psychopharmacology 9 (2): 123–125. 1994. doi:10.1097/00004850-199400920-00009. PMID 8056994.
- ↑ The Psychopharmacologists. 2. A Hodder Arnold Publication. 1998. pp. 132–134. ISBN 978-1-86036-010-7.
- ↑ "Pyridoxine for the treatment of isoniazid-induced seizures in intentional ingestions: The experience of a national poison center". The American Journal of Emergency Medicine 36 (10): 1775–1778. October 2018. doi:10.1016/j.ajem.2018.01.085. PMID 29397257.
- ↑ Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Norwich, NY: Elsevier Science. 2008. pp. 1968–1970. ISBN 978-0-8155-1526-5. https://books.google.com/books?id=bRX8MwEACAAJ&q=9780815515265.
- ↑ "Synthesis of isoniazid from citric acid". Journal of the American Pharmaceutical Association 45 (7): 478–480. July 1956. doi:10.1002/jps.3030450714. PMID 13345683.
- ↑ "Drugs@FDA". United States Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/daf/.
- ↑ "Über Hydrazinderivate der Pyridincarbonsäuren" (in de). Monatshefte für Chemie und Verwandte Teile Anderer Wissenschaften 33 (4): 393–414. 1912-04-01. doi:10.1007/bf01517946. ISSN 1434-4475.
 |