Chemistry:Liquid carbon dioxide

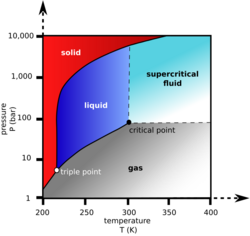
Liquid carbon dioxide is the liquid form of carbon dioxide (CO2). At normal atmospheric pressure, carbon dioxide can only exist as a gas or solid, and is ordinarily found as a trace gas in Earth's atmosphere. Its liquid state can exist at pressures above 5.1 atm (5.2 bar; 75 psi), between the temperatures of its triple point, −56.6 °C (−69.9 °F) and its critical point, 31.1 °C (88.0 °F).[1] Solid CO2, known as dry ice, occurs at low temperatures, and has commercial applications. Dry ice sublimes above −78.5 °C (−109.3 °F) at atmospheric pressure—that is, it transitions directly from solid to gas without an intermediate liquid stage. The uses and applications of liquid carbon dioxide include extracting virgin olive oil from olive paste, in fire extinguishers, and as a coolant. With supercritical carbon dioxide, liquid carbon dioxide is used for decaffeinating coffee.[2]
Properties

Liquid carbon dioxide is a type of liquid which is formed from highly compressed and cooled gaseous carbon dioxide. It does not form under atmospheric conditions. It only exists when the pressure is above 5.1 atm and the temperature is under 31.1 °C (88.0 °F) (temperature of critical point) and above −56.6 °C (−69.9 °F) (temperature of triple point). The chemical symbol remains the same as gaseous carbon dioxide (CO2).[3] It is transparent and odorless, and its density is 1101 kg/m3 when the liquid is at full saturation at −37 °C (−35 °F),[4] decreasing to half that near its critical point.
The solubility of water in liquid carbon dioxide is measured in a range of temperatures, ranging from −29 °C (−20 °F) to 22.6 °C (72.7 °F). At this temperature, the pressure is measured in a range from 15 to 60 atmospheres. The solubility turned out to be very low: from 0.02 to 0.10 %.[5]
Uses

Uses of liquid carbon dioxide include the preservation of food, in fire extinguishers, and in commercial food processes. For food preservation, liquid carbon dioxide is used to refrigerate, preserve, store, and soften. In a fire extinguisher, the CO2 is stored under pressure as a liquid to act as an anti-flammable.[3] The liquid carbon dioxide not only reduces combustion by displacing oxygen, but also cools the burning surface to avoid further damage. Solvent extraction using compressed liquid CO2 can be used in industrial processes such as removing caffeine from coffee[3] or improving the yield of olive oil production.[6]
Liquid carbon dioxide is tested as a means of CO2 transportation for underground or subsea storage purposes. Due to its high density as a liquid, it is much more feasible to ship than as a gas. [7]
CO2 is also used in large-scale heat pumps for district heating, replacing less-environmentally-friendly refrigerants. The CO2 changes phases between liquid and gaseous in the process.[8][9]
See also
Other chemical compounds and elements are commonly used for commercial and research purposes in their liquid state:
References
- ↑ "How to Make Liquid CO2" (in en). https://sciencing.com/make-liquid-co-5192428.html.
- ↑ Zabot, Giovani L. (January 2020). "Chapter 11 - Decaffeination using supercritical carbon dioxide" (in en). Green Sustainable Process for Chemical and Environmental Engineering and Science. Elsevier. pp. 255–278. doi:10.1016/B978-0-12-817388-6.00011-8. ISBN 9780128173886. https://www.sciencedirect.com/science/article/pii/B9780128173886000118. Retrieved 2022-01-01.
- ↑ 3.0 3.1 3.2 "What is Liquid CO2 and What Can It Be Useful For? | Co2 Gas Blog". 2017-10-08. https://www.co2gas.co.uk/2017/10/08/liquid-co2-can-uses/.
- ↑ "Carbon Dioxide - Thermophysical Properties". 2017. https://www.engineeringtoolbox.com/CO2-carbon-dioxide-properties-d_2017.html.
- ↑ Stone, Hosmer W. (December 1943). "Solubility of Water in Liquid Carbon Dioxide". Industrial & Engineering Chemistry 35 (12): 1284–1286. doi:10.1021/ie50408a015. ISSN 0019-7866.
- ↑ Romano, Raffaele; Manzo, Nadia; Montefusco, Immacolata; Romano, Annalisa; Santini, Antonello (2014-03-12). "Liquid Carbon Dioxide Use in the Extraction of Extra Virgin Olive Oil From Olive Paste". Journal of Food Research 3 (4): 119. doi:10.5539/jfr.v3n4p119. ISSN 1927-0895.
- ↑ "Visit the North Sea oilfield where CO2 is pumped under the seabed". 30 January 2026. https://www.bbc.com/news/articles/cq5y7dd284do.
- ↑ Gabrielli, Paolo; Sansavini, Giovanni; Singh, Siddhant; Garcia, Luis Sanz; Jacquemoud, Emmanuel; Jenny, Philipp (1 October 2022). "Off-Design Modeling and Operational Optimization of Trans-Critical Carbon Dioxide Heat Pumps". Journal of Engineering for Gas Turbines and Power 144 (10). doi:10.1115/1.4055233.
- ↑ "MAN Energy Solutions to Supply World’s Largest Air-to-Water Heat Pump for Helsinki’s District Heating". 28 August 2024. https://www.man-es.com/company/press-releases/press-details/2024/08/28/man-energy-solutions-to-supply-world-s-largest-air-to-water-heat-pump-for-helsinki-s-district-heating.
 |
