Biology:let-7 microRNA precursor
| let-7 microRNA precursor | |
|---|---|
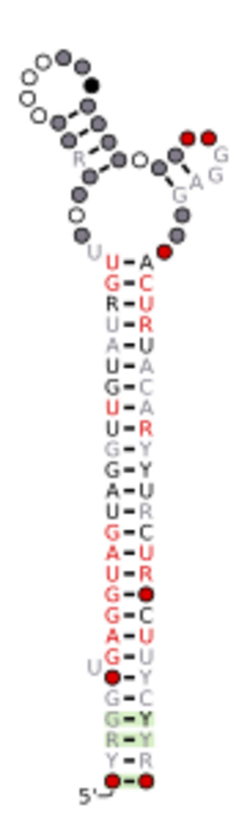 Predicted secondary structure and sequence conservation of let-7 | |
| Identifiers | |
| Symbol | let-7 |
| Rfam | RF00027 |
| miRBase | MI0000001 |
| miRBase family | MIPF0000002 |
| Other data | |
| RNA type | Gene; miRNA |
| Domain(s) | Eukaryota |
| GO | 0035195 0035068 |
| SO | 0001244 |
| PDB structures | PDBe |
The Let-7 microRNA precursor was identified from a study of developmental timing in C. elegans,[1] and was later shown to be part of a much larger class of non-coding RNAs termed microRNAs.[2] miR-98 microRNA precursor from human is a let-7 family member. Let-7 miRNAs have now been predicted or experimentally confirmed in a wide range of species (MIPF0000002[3]). miRNAs are initially transcribed in long transcripts (up to several hundred nucleotides) called primary miRNAs (pri-miRNAs), which are processed in the nucleus by Drosha and Pasha to hairpin structures of about 70 nucleotide. These precursors (pre-miRNAs) are exported to the cytoplasm by exportin5, where they are subsequently processed by the enzyme Dicer to a ~22 nucleotide mature miRNA. The involvement of Dicer in miRNA processing demonstrates a relationship with the phenomenon of RNA interference.
Genomic Locations
In human genome, the cluster let-7a-1/let-7f-1/let-7d is inside the region B at 9q22.3, with the defining marker D9S280-D9S1809. One minimal LOH (loss of heterozygosity) region, between loci D11S1345-D11S1316, contains the cluster miR-125b1/let-7a-2/miR-100. The cluster miR-99a/let-7c/miR-125b-2 is in a 21p11.1 region of HD (homozygous deletions). The cluster let-7g/miR-135-1 is in region 3 at 3p21.1-p21.2.[4]
The let-7 family
The lethal-7 (let-7) gene was first discovered in the nematode as a key developmental regulator and became one of the first two known microRNAs (the other one is lin-4).[5] Soon, let-7 was found in fruit fly, and identified as the first known human miRNA by a BLAST (basic local alignment search tool) research.[6] The mature form of let-7 family members is highly conserved across species.
In C.elegans
In C.elegans, the let-7 family consists of genes encoding nine miRNAs sharing the same seed sequence.[7] Among them, let-7, mir-84, mir-48 and mir-241 are involved in C.elegans heterochronic pathway, sequentially controlling developmental timing of larva transitions.[8] Most animals with loss-of-function let-7 mutation burst through their vulvas and die, and therefore the mutant is lethal (let).[5] The mutants of other let-7 family members have a radio-resistant phenotype in vulval cells, which may be related to their ability to repress RAS.[9]
In Drosophila
There is only one single let-7 gene in the Drosophila genome, which has the identical mature sequence to the one in C.elegans.[10] The role of let-7 has been demonstrated in regulating the timing of neuromuscular junction formation in the abdomen and cell-cycle in the wing.[11] Furthermore, the expression of pri-, pre- and mature let-7 have the same rhythmic pattern with the hormone pulse before each cuticular molt in Drosophila.[12]
In vertebrates
The let-7 family has a lot more members in vertebrates than in C.elegans and Drosophila.[10] The sequences, expression timing, as well as genomic clustering of these miRNAs members are all conserved across species.[13] The direct role of let-7 family in vertebrate development has not been clearly shown as in less complex organisms, yet the expression pattern of let-7 family is indeed temporal during developmental processes.[14] Given that the expression levels of let-7 members are significantly low in human cancers and cancer stem cells,[15] the major function of let-7 genes may be to promote terminal differentiation in development and tumor suppression.
Regulation of expression
Although the levels of mature let-7 members are undetectable in undifferentiated cells, the primary transcripts and the hairpin precursors of let-7 are present in these cells.[16] It indicates that the mature let-7 miRNAs may be regulated in a post-transcriptional manner.
By pluripotency promoting factor LIN28
As one of the genes involved in (but not essential for) induced pluripotent stem (iPS) cell reprogramming,[17] LIN28 expression is reciprocal to that of mature let-7.[18] LIN28 selectively binds the primary and precursor forms of let-7, and inhibits the processing of pri-let-7 to form the hairpin precursor.[19] This binding is facilitated by the conserved loop sequence of primary let-7 family members and RNA-binding domains of LIN28 proteins.[20] Lin-28 uses two zinc knuckle domains to recognize the NGNNG motif in the let-7 precursors,[21] while the Cold-shock domain, connected by a flexible linker, binds to a closed loop in the precursors.[22] On the other hand, let-7 miRNAs in mammals have been shown to regulate LIN28,[23] which implies that let-7 might enhance its own level by repressing LIN28, its negative regulator.[24]
In autoregulatory loop with MYC
Expression of let-7 members is controlled by MYC binding to their promoters. The levels of let-7 have been reported to decrease in models of MYC-mediated tumorigenesis, and to increase when MYC is inhibited by chemicals.[25] In a twist, there are let-7-binding sites in MYC 3' untranslated region(UTR) according to bioinformatic analysis, and let-7 overexpression in cell culture decreased MYC mRNA levels.[26] Therefore, there is a double-negative feedback loop between MYC and let-7. Furthermore, let-7 could lead to IMP1(/insulin-like growth factor II mRNA-binding protein) depletion, which destabilizes MYC mRNA, thus forming an indirect regulatory pathway.[27]
Targets of let-7
Oncogenes: RAS, HMGA2
Let-7 has been demonstrated to be a direct regulator of RAS expression in human cells[28] All the three RAS genes in human, K-, N-, and H-, have the predicted let-7 binding sequences in their 3'UTRs. In lung cancer patient samples, expression of RAS and let-7 showed reciprocal pattern, which has low let-7 and high RAS in cancerous cells, and high let-7 and low RAS in normal cells. Another oncogene, high mobility group A2 (HMGA2), has also been identified as a target of let-7. Let-7 directly inhibits HMGA2 by binding to its 3'UTR.[29] Removal of let-7 binding site by 3'UTR deletion cause overexpression of HMGA2 and formation of tumor.
Cell cycle, proliferation, and apoptosis regulators
Microarray analyses revealed many genes regulating cell cycle and cell proliferation that are responsive to alteration of let-7 levels, including cyclin A2, CDC34, Aurora A and B kinases (STK6 and STK12), E2F5, and CDK8, among others.[28] Subsequent experiments confirmed the direct effects of some of these genes, such as CDC25A and CDK6.[30] Let-7 also inhibits several components of DNA replication machinery, transcription factors, even some tumor suppressor genes and checkpoint regulators.[28] Apoptosis is regulated by let-7 as well, through Casp3, Bcl2, Map3k1 and Cdk5 modulation.[31]
Immunity
Let-7 has been implicated in post-transcriptional control of innate immune responses to pathogenic agents. Macrophages stimulated with live bacteria or purified microbial components down-regulate the expression of several members of the let-7 microRNA family to relieve repression of immune-modulatory cytokines IL-6 and IL-10.[32][33] Let-7 has also been implicated in the negative regulation of TLR4, the major immune receptor of microbial lipopolysaccharide and down-regulation of let-7 both upon microbial and protozoan infection might elevate TLR4 signalling and expression.[34][35] Let-7 has furthermore been reported to regulate the production of cytokine IL-13 by T lymphocytes during allergic airway inflammation thus linking this microRNA to adaptive immunity as well.[36] Down-modulation of let-7 negative regulator Lin28b in human T lymphocytes is believed to accrue during early neonate development to reprogram the immune system towards defense.[37]
Potential clinical use in cancer
Given the prominent phenotype of cell overproliferation and undifferentiation by let-7 loss-of-function in nematodes, and the role of its targets on cell destiny determination, let-7 is closely associated with human cancer and acts as a tumor suppressor.
Diagnosis
Numerous reports have shown that the expression levels of let-7 are frequently low and the chromosomal clusters of let-7 are often deleted in many cancers.[4] Let-7 is expressed at higher levels in more differentiated tumors, which also have lower levels of activated oncogenes such as RAS and HMGA2. Therefore, expression levels of let-7 could be prognostic markers in several cancers associated with differentiation stages.[38] In lung cancer, for example, reduced expression of let-7 is significantly correlated with reduced postoperative survival.[39] The expression of let-7b and let-7g microRNAs are significantly associated with overall survival in 1262 breast cancer patients.[40]
Therapy
Let-7 is also a very attractive potential therapeutic that can prevent tumorigenesis and angiogenesis, typically in cancers that underexpress let-7.[41] Lung cancer, for instance, has several key oncogenic mutations including p53, RAS and MYC, some of which may directly correlate with the reduced expression of let-7, and may be repressed by introduction of let-7.[39] Intranasal administration of let-7 has already been found effective in reducing tumor growth in a transgenic mouse model of lung cancer.[42] Similar restoration of let-7 was also shown to inhibit cell proliferation in breast, colon and hepatic cancers, lymphoma, and uterine leiomyoma.[43]
References
- ↑ Rougvie, AE (2001). "Control of developmental timing in animals". Nature Reviews Genetics 2 (9): 690–701. doi:10.1038/35088566. PMID 11533718.
- ↑ Ambros, V (2001). "microRNAs: tiny regulators with great potential". Cell 107 (7): 823–826. doi:10.1016/S0092-8674(01)00616-X. PMID 11779458.
- ↑ MIPF0000002
- ↑ 4.0 4.1 Calin; Sevignani, C; Dumitru, CD; Hyslop, T; Noch, E; Yendamuri, S; Shimizu, M; Rattan, S et al. (2003). "Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers". PNAS 101 (9): 2999–3004. doi:10.1073/pnas.0307323101. PMID 14973191. Bibcode: 2004PNAS..101.2999C.
- ↑ 5.0 5.1 Reinhart B.J. et al. (2000). "The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans". Nature 403 (6772): 901–906. doi:10.1038/35002607. PMID 10706289. Bibcode: 2000Natur.403..901R.
- ↑ Pasquinelli A.E. (2000). "Conservation of the sequence and temporal expression of let-7 heterochronic regulatory RNA". Nature 408 (6808): 86–89. doi:10.1038/35040556. PMID 11081512. Bibcode: 2000Natur.408...86P.
- ↑ Lim L.P. (2003). "The microRNAs of Caenorhabditis elegans". Genes Dev 17 (8): 991–1008. doi:10.1101/gad.1074403. PMID 12672692.
- ↑ Moss E.G. (2007). "Heterochronic genes and the nature of developmental time". Curr. Biol. 17 (11): R425–R434. doi:10.1016/j.cub.2007.03.043. PMID 17550772.
- ↑ Weidhaas J.B. (2007). "MicroRNAs as potential agents to alter resistance to cytotoxic anticancer therapy". Cancer Res. 67 (23): 11111–11116. doi:10.1158/0008-5472.CAN-07-2858. PMID 18056433.
- ↑ 10.0 10.1 Lagos-Quintana M. et al. (2001). "Identification of novel genes coding for small expressed RNAs". Science 294 (5543): 853–858. doi:10.1126/science.1064921. PMID 11679670. Bibcode: 2001Sci...294..853L.
- ↑ Caygill E.E.; Johnston L.A. (2008). "Temporal Regulation of Metamorphic Processes in Drosophila by the let-7 and miR-125 Heterochronic MicroRNAs". Curr. Biol. 18 (13): 943–950. doi:10.1016/j.cub.2008.06.020. PMID 18571409.
- ↑ Thummel C.S. (2001). "Molecular mechanisms of developmental timing in C. elegans and Drosophila". Dev. Cell 1 (4): 453–465. doi:10.1016/S1534-5807(01)00060-0. PMID 11703937.
- ↑ Rodriguez A. (2004). "Identification of Mammalian microRNA Host Genes and Transcription Units". Genome Res. 14 (10A): 1902–1910. doi:10.1101/gr.2722704. PMID 15364901.
- ↑ Kloosterman W.P.; Plasterk R.H. (2006). "The diverse functions of microRNAs in animal development and disease". Dev. Cell 11 (4): 441–450. doi:10.1016/j.devcel.2006.09.009. PMID 17011485.
- ↑ Esquela-Kerscher A.; Slack F.J. (2006). "Oncomirs – microRNAs with a role in cancer". Nature Reviews Cancer 6 (4): 259–269. doi:10.1038/nrc1840. PMID 16557279.
- ↑ Thomson J.M. (2006). "Extensive post-transcriptional regulation of microRNAs and its implications for cancer". Genes Dev. 20 (16): 2202–2207. doi:10.1101/gad.1444406. PMID 16882971.
- ↑ Yu J. et al. (2007). "Induced pluripotent stem cell lines derived from human somatic cells". Science 318 (5858): 1917–1920. doi:10.1126/science.1151526. PMID 18029452. Bibcode: 2007Sci...318.1917Y.
- ↑ Viswanathan S.R. et al. (2008). "Selective blockade of microRNA processing by Lin-28". Science 320 (5872): 97–100. doi:10.1126/science.1154040. PMID 18292307. Bibcode: 2008Sci...320...97V.
- ↑ Newman M.A. (2008). "Lin-28 interaction with the let-7 precursor loop mediates regulated microRNA processing". RNA 14 (8): 1539–49. doi:10.1261/rna.1155108. PMID 18566191.
- ↑ Piskounova E. (2008). "Determinants of microRNA processing inhibition by the developmentally regulated RNA-binding protein Lin28". J. Biol. Chem. 283 (31): 21310–21314. doi:10.1074/jbc.C800108200. PMID 18550544.
- ↑ Loughlin, Fionna E; Gebert, Luca F R; Towbin, Harry; Brunschweiger, Andreas; Hall, Jonathan; Allain, Frédéric H-T (11 December 2011). "Structural basis of pre-let-7 miRNA recognition by the zinc knuckles of pluripotency factor Lin28". Nature Structural & Molecular Biology 19 (1): 84–89. doi:10.1038/nsmb.2202. PMID 22157959.
- ↑ Nam, Yunsun; Chen, Casandra; Gregory, Richard I.; Chou, James J.; Sliz, Piotr (November 2011). "Molecular Basis for Interaction of let-7 MicroRNAs with Lin28". Cell 147 (5): 1080–1091. doi:10.1016/j.cell.2011.10.020. PMID 22078496.
- ↑ Moss E.G.; Tang L. (2003). "Conservation of the heterochronic regulator Lin-28, its developmental expression and microRNA complementary sites". Dev. Biol. 258 (2): 432–442. doi:10.1016/S0012-1606(03)00126-X. PMID 12798299.
- ↑ Ali, P. S.; Ghoshdastider, U; Hoffmann, J; Brutschy, B; Filipek, S (2012). "Recognition of the let-7g miRNA precursor by human Lin28B". FEBS Letters 586 (22): 3986–90. doi:10.1016/j.febslet.2012.09.034. PMID 23063642.
- ↑ Chang T.C. (2007). "Widespread microRNA repression by Myc contributes to tumorigenesis". Nat. Genet. 40 (1): 43–50. doi:10.1038/ng.2007.30. PMID 18066065.
- ↑ Koscianska E. et al. (2007). "Prediction and preliminary validation of oncogene regulation by miRNAs". BMC Mol. Biol. 8: 79. doi:10.1186/1471-2199-8-79. PMID 17877811.
- ↑ Ioannidis P. (2005). "CRD-BP/IMP1 expression characterizes cord blood CD34+ stem cells and affects c-myc and IGF-II expression in MCF-7 cancer cells". J. Biol. Chem. 280 (20): 20086–20093. doi:10.1074/jbc.M410036200. PMID 15769738.
- ↑ 28.0 28.1 28.2 "RAS is regulated by the let-7 microRNA family". Cell 120 (5): 635–47. 2005. doi:10.1016/j.cell.2005.01.014. PMID 15766527.
- ↑ Mayr C. et al. (2007). "Disrupting the Pairing Between let-7 and Hmga2 Enhances Oncogenic Transformation". Science 315 (5818): 1576–1579. doi:10.1126/science.1137999. PMID 17322030. Bibcode: 2007Sci...315.1576M.
- ↑ Johnson C.D. (2007). "The let-7 microRNA represses cell proliferation pathways in human cells". Cancer Res. 67 (16): 7713–7722. doi:10.1158/0008-5472.CAN-07-1083. PMID 17699775.
- ↑ He YJ, Guo L, D ZH. (2009) Let-7 and mir-24 in uvb-induced apoptosis [Chinese]. Zhonghua Fang She Yi Xue Yu Fang Hu Za Zhi. 29, 234–6.
- ↑ Schulte LN (2011). "Analysis of the host miRNA response to Salmonella uncovers the control of major cytokines by the let-7 family". The EMBO Journal 30 (10): 1977–1989. doi:10.1038/emboj.2011.94. PMID 21468030.
- ↑ Liu Y (2011). "MicroRNA-98 negatively regulates IL-10 production and endotoxin tolerance in macrophages after LPS stimulation". FEBS Letters 585 (12): 1963–1968. doi:10.1016/j.febslet.2011.05.029. PMID 21609717.
- ↑ Hu G (2009). "MicroRNA-98 and let-7 Confer Cholangiocyte Expression of Cytokine-Inducible Src Homology 2-Containing Protein in Response to Microbial Challenge". The Journal of Immunology 183 (3): 1617–1624. doi:10.4049/jimmunol.0804362. PMID 19592657.
- ↑ Androulidaki A (2009). "Akt1 controls macrophage response to LPS by regulating microRNAs". Immunity 31 (2): 220–231. doi:10.1016/j.immuni.2009.06.024. PMID 19699171.
- ↑ Kumar M (2011). "Let-7 microRNA-mediated regulation of IL-13 and allergic airway inflammation". The Journal of Allergy and Clinical Immunology 128 (5): 1077–1085. doi:10.1016/j.jaci.2011.04.034. PMID 21616524.
- ↑ Yuan J et al. (2012). "Lin28b Reprograms Adult Bone Marrow Hematopoietic Progenitors to Mediate Fetal-Like Lymphopoiesis". Science 335 (6073): 1195–12000. doi:10.1126/science.1216557. PMID 22345399. Bibcode: 2012Sci...335.1195Y.
- ↑ Shell S; Park SM; Radjabi AR et al. (2007). "Let-7 expression defines two differentiation stages of cancer". Proc Natl Acad Sci U S A 104 (27): 11400–5. doi:10.1073/pnas.0704372104. PMID 17600087. Bibcode: 2007PNAS..10411400S.
- ↑ 39.0 39.1 Takamizawa JExpression error: Unrecognized word "etal". (2004). "Reduced expression of the let-7 micrornas in human lung cancers in association with shortened postoperative survival". Cancer Res. 64 (11): 3753–6. doi:10.1158/0008-5472.CAN-04-0637. PMID 15172979.
- ↑ Lánczky, András; Nagy, Ádám; Bottai, Giulia; Munkácsy, Gyöngyi; Szabó, András; Santarpia, Libero; Győrffy, Balázs (2016-12-01). "miRpower: a web-tool to validate survival-associated miRNAs utilizing expression data from 2178 breast cancer patients". Breast Cancer Research and Treatment 160 (3): 439–446. doi:10.1007/s10549-016-4013-7. ISSN 1573-7217. PMID 27744485.
- ↑ "Role of Dicer and Drosha for endothelial microrna expression and angiogenesis". Circ Res 101 (1): 59–68. 2007. doi:10.1161/CIRCRESAHA.107.153916. PMID 17540974.
- ↑ EsquelaExpression error: Unrecognized word "etal". (2008). "The let-7 microrna reduces tumor growth in mouse models of lung cancer". Cell Cycle 7 (6): 759–64. doi:10.4161/cc.7.6.5834. PMID 18344688.
- ↑ Barh D.; Malhotra R.; Ravi B.; Sindhurani P. (2010). "MicroRNA let-7: an emerging next-generation cancer therapeutic". Current Oncology 17 (1): 70–80. doi:10.3747/co.v17i1.356. PMID 20179807.
Further reading
- "Collagen regulation of let-7 in pancreatic cancer involves TGF-β1-mediated membrane type 1-matrix metalloproteinase expression". Oncogene 30 (8): 1002–1008. 2010. doi:10.1038/onc.2010.485. PMID 21057545.
- "Double negative feedback loop between reprogramming factor LIN28 and microRNA let-7 regulates aldehyde dehydrogenase 1-positive cancer stem cells". Cancer Res 70 (22): 9463–9472. 2010. doi:10.1158/0008-5472.CAN-10-2388. PMID 21045151.
- Wölfl, Stefan, ed (2010). "Let-7 MicroRNA Family Is Selectively Secreted into the Extracellular Environment via Exosomes in a Metastatic Gastric Cancer Cell Line". PLOS ONE 5 (10): e13247. doi:10.1371/journal.pone.0013247. PMID 20949044. Bibcode: 2010PLoSO...513247O.
- "Ascl1a regulates Müller glia dedifferentiation and retina regeneration via a Lin-28-dependent, let-7 miRNA signaling pathway". Nat Cell Biol 12 (11): 1101–7. 2010. doi:10.1038/ncb2115. PMID 20935637.
- "Role of KRAS let-7 LCS6 SNP in metastatic colorectal cancer patients". Ann Oncol 22 (1): 234–5. 2010. doi:10.1093/annonc/mdq472. PMID 20926546.
- "Hormonal regulation of Drosophila microRNA let-7 and miR-125 that target innate immunity". Fly (Austin) 4 (4): 306–11. 2010. doi:10.4161/fly.4.4.13008. PMID 20798594.
- "A Yin-Yang Balancing Act of the Lin28/Let-7 Link in Tumorigenesis". J Hepatol 53 (5): 974–5. 2010. doi:10.1016/j.jhep.2010.07.001. PMID 20739081.
- "Review Article: let-7 and miR-17-92: Small-sized major players in lung cancer development". Cancer Sci 102 (1): 9–17. 2010. doi:10.1111/j.1349-7006.2010.01707.x. PMID 20735434.
- "Regulation of opioid tolerance by let-7 family microRNA targeting the μ opioid receptor". J Neurosci 30 (30): 10251–8. 2010. doi:10.1523/JNEUROSCI.2419-10.2010. PMID 20668208.
- "NMR structure of the let-7 miRNA interacting with the site LCS1 of lin-41 mRNA from Caenorhabditis elegans". Nucleic Acids Res 38 (21): 7814–21. 2010. doi:10.1093/nar/gkq640. PMID 20660479.
- "Epigenetic Down-Regulation of the Tumor Suppressor Gene PRDM1/Blimp-1 in Diffuse Large B Cell Lymphomas : A Potential Role of the MicroRNA Let-7". Am J Pathol 177 (3): 1470–9. 2010. doi:10.2353/ajpath.2010.091291. PMID 20651244.
- "Proinflammatory Role for let-7 MicroRNAS in Experimental Asthma". J Biol Chem 285 (39): 30139–49. 2010. doi:10.1074/jbc.M110.145698. PMID 20630862.
- "Lin-28: an early embryonic sentinel that blocks Let-7 biogenesis". Int J Biochem Cell Biol 42 (8): 1330–3. 2010. doi:10.1016/j.biocel.2009.02.023. PMID 20619222.
- "Let-7 microRNA inhibits the proliferation of human glioblastoma cells". J Neurooncol 102 (1): 19–24. 2010. doi:10.1007/s11060-010-0286-6. PMID 20607356.
- "A let-7 microRNA-binding site polymorphism in 3'-untranslated region of KRAS gene predicts response in wild-type KRAS patients with metastatic colorectal cancer treated with cetuximab monotherapy". Ann Oncol 22 (1): 104–9. 2010. doi:10.1093/annonc/mdq315. PMID 20603437.
- "Let-7 family miRNAs regulate estrogen receptor alpha signaling in estrogen receptor positive breast cancer". Breast Cancer Res Treat 127 (1): 69–80. 2010. doi:10.1007/s10549-010-0972-2. PMID 20535543.
- "MicroRNA-98 and let-7 Regulate Expression of Suppressor of Cytokine Signaling-4 in Biliary Epithelial Cells in Response to Cryptosporidium parvum Infection". J Infect Dis 202 (1): 125–35. 2010. doi:10.1086/653212. PMID 20486857.
- "Mutations in the let-7 binding site – a mechanism of RAS activation in juvenile myelomonocytic leukemia?". Haematologica 95 (9): 1616. 2010. doi:10.3324/haematol.2010.024984. PMID 20460640.
- "MicroRNA let-7 suppresses nasopharyngeal carcinoma cells proliferation through downregulating c-Myc expression". J Cancer Res Clin Oncol 137 (3): 415–422. 2010. doi:10.1007/s00432-010-0898-4. PMID 20440510.
- "The let-7 family of microRNAs inhibits Bcl-xL expression and potentiates sorafenib-induced apoptosis in human hepatocellular carcinoma". J Hepatol 52 (5): 698–704. 2010. doi:10.1016/j.jhep.2009.12.024. PMID 20347499.
- "Overexpression of Dicer as a Result of Reduced let-7 microRNA Levels Contributes to Increased Cell Proliferation of Oral Cancer Cells". Genes Chromosomes Cancer 49 (6): 549–59. 2010. doi:10.1002/gcc.20765. PMID 20232482.
- "Analysis of deep sequencing microRNA expression profile from human embryonic stem cells derived mesenchymal stem cells reveals possible role of let-7 microRNA family in downstream targeting of Hepatic Nuclear Factor 4 Alpha". BMC Genomics 11 (Suppl 1): S6. 2010. doi:10.1186/1471-2164-11-S1-S6. PMID 20158877.
- "LIN28 alters cell fate succession and acts independently of the let-7 microRNA during neurogliogenesis in vitro". Development 137 (6): 891–900. 2010. doi:10.1242/dev.042895. PMID 20179095.
- "Genetic modulation of the Let-7 microRNA binding to KRAS 3'-untranslated region and survival of metastatic colorectal cancer patients treated with salvage cetuximab-irinotecan". Pharmacogenomics J 10 (5): 458–64. 2010. doi:10.1038/tpj.2010.9. PMID 20177422.
- "Loss of let-7 binding sites resulting from truncations of the 3' untranslated region of HMGA2 mRNA in uterine leiomyomas". Cancer Genet Cytogenet 196 (2): 119–23. 2010. doi:10.1016/j.cancergencyto.2009.09.021. PMID 20082846.
- "Lin28-let7 modulates radiosensitivity of human cancer cells with activation of K-Ras". Int J Radiat Oncol Biol Phys 76 (1): 5–8. 2010. doi:10.1016/j.ijrobp.2009.08.028. PMID 20005451.
- "Correlation of overexpression of HMGA1 and HMGA2 with poor tumor differentiation, invasion, and proliferation associated with let-7 down-regulation in retinoblastomas". Hum Pathol 41 (4): 493–502. 2010. doi:10.1016/j.humpath.2009.08.022. PMID 20004941.
- "Regression of murine lung tumors by the let-7 microRNA". Oncogene 29 (11): 1580–7. 2010. doi:10.1038/onc.2009.445. PMID 19966857.
- "Effects of let-7 microRNA on Cell Growth and Differentiation of Papillary Thyroid Cancer". Transl Oncol 2 (4): 236–41. 2009. doi:10.1593/tlo.09151. PMID 19956384.
- "Let-7 microRNAs are developmentally regulated in circulating human erythroid cells". J Transl Med 7: 98. 2009. doi:10.1186/1479-5876-7-98. PMID 19939273.
- "The let-7 target gene mouse lin-41 is a stem cell specific E3 ubiquitin ligase for the miRNA pathway protein Ago2". Nat Cell Biol 11 (12): 1411–20. 2009. doi:10.1038/ncb1987. PMID 19898466.
- "An epigenetic switch involving NF-κB, Lin28, let-7 microRNA, and IL6 links inflammation to cell transformation". Cell 139 (4): 693–706. 2009. doi:10.1016/j.cell.2009.10.014. PMID 19878981.
- "A feedback circuit involving let-7-family miRNAs and DAF-12 integrates environmental signals and developmental timing in Caenorhabditis elegans". Proc Natl Acad Sci U S A 106 (44): 18668–73. 2009. doi:10.1073/pnas.0908131106. PMID 19828440. Bibcode: 2009PNAS..10618668H.
- "The role of let-7 in cell differentiation and cancer". Endocr Relat Cancer 17 (1): F19–36. 2010. doi:10.1677/ERC-09-0184. PMID 19779035.
- "Lin28 recruits the TUTase Zcchc11 to inhibit let-7 maturation in embryonic stem cells". Nature Structural & Molecular Biology 16 (10): 1021–5. 2009. doi:10.1038/nsmb.1676. PMID 19713958.
- "LIN-28 and the poly(U) polymerase PUP-2 regulate let-7 microRNA processing in Caenorhabditis elegans". Nature Structural & Molecular Biology 16 (10): 1016–20. 2009. doi:10.1038/nsmb.1675. PMID 19713957.
- "Up-regulation of miR-200 and let-7 by natural agents leads to the reversal of epithelial-mesenchymal transition in gemcitabine-resistant pancreatic cancer cells". Cancer Res 69 (16): 6704–12. 2009. doi:10.1158/0008-5472.CAN-09-1298. PMID 19654291.
- "Transcription of the C. elegans let-7 microRNA is temporally regulated by one of its targets, hbl-1". Dev Biol 334 (2): 523–34. 2009. doi:10.1016/j.ydbio.2009.07.012. PMID 19627983.
- "Ribosomal protein RPS-14 modulates let-7 microRNA function in Caenorhabditis elegans". Dev Biol 334 (1): 152–60. 2009. doi:10.1016/j.ydbio.2009.07.011. PMID 19627982.
- "Let-7 repression leads to HMGA2 overexpression in uterine leiomyosarcoma". J Cell Mol Med 13 (9B): 3898–905. 2009. doi:10.1111/j.1582-4934.2008.00541.x. PMID 19602040.
- "HuR recruits let-7/RISC to repress c-Myc expression". Genes Dev 23 (15): 1743–8. 2009. doi:10.1101/gad.1812509. PMID 19574298.
- "Identification of novel HMGA2 fusion sequences in lipoma: evidence that deletion of let-7 miRNA consensus binding site 1 in the HMGA2 3' UTR is not critical for HMGA2 transcriptional upregulation". Genes Chromosomes Cancer 48 (8): 673–8. 2009. doi:10.1002/gcc.20674. PMID 19431195.
- "A let-7 microRNA-binding site polymorphism in the KRAS 3′ UTR is associated with reduced survival in oral cancers". Carcinogenesis 30 (6): 1003–7. 2009. doi:10.1093/carcin/bgp099. PMID 19380522.
- "let-7 microRNA reduces tumor growth". Cell Cycle 8 (12): 1823. 2009. doi:10.4161/cc.8.12.8639. PMID 19377282.
- "MicroRNA let-7 Regulates 3T3-L1 Adipogenesis". Mol Endocrinol 23 (6): 925–31. 2009. doi:10.1210/me.2008-0298. PMID 19324969.
- "let-7 MicroRNA transfer in pancreatic cancer-derived cells inhibits in vitro cell proliferation but fails to alter tumor progression". Hum Gene Ther 20 (8): 831–44. 2009. doi:10.1089/hum.2008.134. PMID 19323605.
- "Let-7 and miR-200 microRNAs: Guardians against pluripotency and cancer progression". Cell Cycle 8 (6): 843–52. 2009. doi:10.4161/cc.8.6.7907. PMID 19221491.
- "Lin-28B transactivation is necessary for Myc-mediated let-7 repression and proliferation". Proc Natl Acad Sci U S A 106 (9): 3384–9. 2009. doi:10.1073/pnas.0808300106. PMID 19211792. Bibcode: 2009PNAS..106.3384C.
- "Frequent overexpression of HMGA1 and 2 in gastroenteropancreatic neuroendocrine tumours and its relationship to let-7 downregulation". Br J Cancer 100 (3): 501–10. 2009. doi:10.1038/sj.bjc.6604883. PMID 19156147.
- "Raf kinase inhibitory protein suppresses a metastasis signalling cascade involving LIN28 and let-7". The EMBO Journal 28 (4): 347–58. 2009. doi:10.1038/emboj.2008.294. PMID 19153603.
- "Overexpression of HMGA2 relates to reduction of the let-7 and its relationship to clinicopathological features in pituitary adenomas". Mod Pathol 22 (3): 431–41. 2009. doi:10.1038/modpathol.2008.202. PMID 19136928.
- "let-7 Overexpression Leads to an Increased Fraction of Cells in G2/M, Direct Down-regulation of Cdc34, and Stabilization of Wee1 Kinase in Primary Fibroblasts". J Biol Chem 284 (11): 6605–9. 2009. doi:10.1074/jbc.C900002200. PMID 19126550.
- "The let-7 microRNA target gene, Mlin41/Trim71 is required for mouse embryonic survival and neural tube closure". Cell Cycle 7 (24): 3935–42. 2008. doi:10.4161/cc.7.24.7397. PMID 19098426.
- "Lin28 mediates the terminal uridylation of let-7 precursor MicroRNA". Mol Cell 32 (2): 276–84. 2008. doi:10.1016/j.molcel.2008.09.014. PMID 18951094.
- "A SNP in a let-7 microRNA complementary site in the KRAS 3′UTR Increases Non-Small Cell Lung Cancer Risk". Cancer Res 68 (20): 8535–40. 2008. doi:10.1158/0008-5472.CAN-08-2129. PMID 18922928.
- "A novel biochemical method to identify target genes of individual microRNAs: Identification of a new Caenorhabditis elegans let-7 target". RNA 14 (11): 2440–51. 2008. doi:10.1261/rna.1139508. PMID 18824511.
- "The let-7 microRNA interfaces extensively with the translation machinery to regulate cell differentiation". Cell Cycle 7 (19): 3083–90. 2008. doi:10.4161/cc.7.19.6778. PMID 18818519.
- "A search for conserved sequences in coding regions reveals that the let-7 microRNA targets Dicer within its coding sequence". Proc Natl Acad Sci U S A 105 (39): 14879–84. 2008. doi:10.1073/pnas.0803230105. PMID 18812516. Bibcode: 2008PNAS..10514879F.
- "The let-7 family of microRNAs". Trends Cell Biol 18 (10): 505–16. 2008. doi:10.1016/j.tcb.2008.07.007. PMID 18774294.
- "let-7: Developmental timing conserved through evolution". Curr Biol 18 (16): R707–8. 2008. doi:10.1016/j.cub.2008.07.013. PMID 18727906.
- "Identification of specific let-7 microRNA binding complexes in Caenorhabditis elegans". RNA 14 (10): 2104–14. 2008. doi:10.1261/rna.551208. PMID 18719242.
- "let-7 regulates Dicer expression and constitutes a negative feedback loop". Carcinogenesis 29 (11): 2073–7. 2008. doi:10.1093/carcin/bgn187. PMID 18700235.
- "let-7 microRNAs in development, stem cells and cancer". Trends Mol Med 14 (9): 400–9. 2008. doi:10.1016/j.molmed.2008.07.001. PMID 18674967.
- "Enjoy the Silence: The Story of let-7 MicroRNA and Cancer". Curr Genomics 8 (4): 229–33. 2007. doi:10.2174/138920207781386933. PMID 18645597.
- "Mouse let-7 miRNA populations exhibit RNA editing that is constrained in the 5′-seed/ cleavage/anchor regions and stabilize predicted mmu-let-7a:mRNA duplexes". Genome Res 18 (10): 1571–81. 2008. doi:10.1101/gr.078246.108. PMID 18614752.
- "A feedback loop comprising lin-28 and let-7 controls pre-let-7 maturation during neural stem-cell commitment". Nat Cell Biol 10 (8): 987–93. 2008. doi:10.1038/ncb1759. PMID 18604195.
- "A let-7 MicroRNA-sensitive vesicular stomatitis virus demonstrates tumor-specific replication". Mol Ther 16 (8): 1437–43. 2008. doi:10.1038/mt.2008.130. PMID 18560417.
- "Drosophila let-7 microRNA is required for remodeling of the neuromusculature during metamorphosis". Genes Dev 22 (12): 1591–6. 2008. doi:10.1101/gad.1671708. PMID 18559475.
- "Clinical significance of high mobility group A2 in human gastric cancer and its relationship to let-7 microRNA family". Clin Cancer Res 14 (8): 2334–40. 2008. doi:10.1158/1078-0432.CCR-07-4667. PMID 18413822.
- "Identification of let-7-regulated oncofetal genes". Cancer Res 68 (8): 2587–91. 2008. doi:10.1158/0008-5472.CAN-08-0264. PMID 18413726.
- "Antiproliferative effects by Let-7 repression of high-mobility group A2 in uterine leiomyoma". Mol Cancer Res 6 (4): 663–73. 2008. doi:10.1158/1541-7786.MCR-07-0370. PMID 18403645.
- "let-7 microRNA expression and the distinction between nonmucinous and mucinous bronchioloalveolar carcinomas". Lung Cancer 60 (2): 307. 2008. doi:10.1016/j.lungcan.2008.02.010. PMID 18395292.
- "Do cells let-7 determine stemness?". Cell Stem Cell 2 (1): 8–9. 2008. doi:10.1016/j.stem.2007.12.003. PMID 18371414.
- "Upregulation of the let-7 microRNA with precocious development in lin-12/Notch hypermorphic C. elegans mutants". Dev Biol 316 (2): 191–9. 2008. doi:10.1016/j.ydbio.2007.12.046. PMID 18334253.
- "Suppression of non-small cell lung tumor development by the let-7 microRNA family". Proc Natl Acad Sci U S A 105 (10): 3903–8. 2008. doi:10.1073/pnas.0712321105. PMID 18308936. Bibcode: 2008PNAS..105.3903K.
- "Solution structure of a let-7 miRNA:lin-41 mRNA complex from C. elegans". Nucleic Acids Res 36 (7): 2330–7. 2008. doi:10.1093/nar/gkn088. PMID 18296482.
- "let-7 regulates self renewal and tumorigenicity of breast cancer cells". Cell 131 (6): 1109–23. 2007. doi:10.1016/j.cell.2007.10.054. PMID 18083101.
- "Regulation of the Drosophila lin-41 homologue dappled by let-7 reveals conservation of a regulatory mechanism within the LIN-41 subclade". Dev Dyn 237 (1): 196–208. 2008. doi:10.1002/dvdy.21396. PMID 18069688.
- "Let-7 prevents early cancer progression by suppressing expression of the embryonic gene HMGA2". Cell Cycle 6 (21): 2585–90. 2007. doi:10.4161/cc.6.21.4845. PMID 17957144.
- "Human TRIM71 and its nematode homologue are targets of let-7 microRNA and its zebrafish orthologue is essential for development". Mol Biol Evol 24 (11): 2525–34. 2007. doi:10.1093/molbev/msm195. PMID 17890240.
- "microRNAs and regeneration: let-7 members as potential regulators of dedifferentiation in lens and inner ear hair cell regeneration of the adult newt". Biochem Biophys Res Commun 362 (4): 940–5. 2007. doi:10.1016/j.bbrc.2007.08.077. PMID 17765873.
- "let-7 microRNA expression is reduced in bronchioloalveolar carcinoma, a non-invasive carcinoma, and is not correlated with prognosis". Lung Cancer 58 (3): 392–6. 2007. doi:10.1016/j.lungcan.2007.07.013. PMID 17728006.
- "P68 RNA helicase unwinds the human let-7 microRNA precursor duplex and is required for let-7-directed silencing of gene expression". J Biol Chem 282 (45): 32773–9. 2007. doi:10.1074/jbc.M705054200. PMID 17724023.
- "Let-7 microRNA-mediated mRNA deadenylation and translational repression in a mammalian cell-free system". Genes Dev 21 (15): 1857–62. 2007. doi:10.1101/gad.1566707. PMID 17671087.
- "Characterization and expression patterns of let-7 microRNA in the silkworm (Bombyx mori)". BMC Dev Biol 7: 88. 2007. doi:10.1186/1471-213X-7-88. PMID 17651473.
- "The tumor suppressor microRNA let-7 represses the HMGA2 oncogene". Genes Dev 21 (9): 1025–30. 2007. doi:10.1101/gad.1540407. PMID 17437991.
- "The C. elegans pumilio homolog, puf-9, is required for the 3'UTR mediated repression of the let-7 microRNA target gene, hbl-1". Dev Biol 305 (2): 551–63. 2007. doi:10.1016/j.ydbio.2007.02.040. PMID 17412319.
- "Misexpression of the Caenorhabditis elegans miRNA let-7 is sufficient to drive developmental programs". Cold Spring Harb Symp Quant Biol 71: 21–7. 2006. doi:10.1101/sqb.2006.71.018. PMID 17381276.
- "Post-transcriptional regulation of the let-7 microRNA during neural cell specification". FASEB J 21 (2): 415–26. 2007. doi:10.1096/fj.06-6130com. PMID 17167072.
- "The mir-84 and let-7 paralogous microRNA genes of Caenorhabditis elegans direct the cessation of molting via the conserved nuclear hormone receptors NHR-23 and NHR-25". Development 133 (23): 4631–41. 2006. doi:10.1242/dev.02655. PMID 17065234.
- "let-7 microRNA functions as a potential growth suppressor in human colon cancer cells". Biol Pharm Bull 29 (5): 903–6. 2006. doi:10.1248/bpb.29.903. PMID 16651716.
- "Reciprocal Expression of lin-41 and the microRNAs let-7 and mir-125 During Mouse Embryogenesis". Dev Dyn 234 (4): 1046–54. 2005. doi:10.1002/dvdy.20599. PMID 16247770.
- "Post-embryonic expression of C. elegans microRNAs belonging to the lin-4 and let-7 families in the hypodermis and the reproductive system". Dev Dyn 234 (4): 868–77. 2005. doi:10.1002/dvdy.20572. PMID 16217741.
- "Regulatory mutations of mir-48, a C. elegans let-7 family MicroRNA, cause developmental timing defects". Dev Cell 9 (3): 415–22. 2005. doi:10.1016/j.devcel.2005.08.002. PMID 16139229.
- "The let-7 MicroRNA family members mir-48, mir-84, and mir-241 function together to regulate developmental timing in Caenorhabditis elegans". Dev Cell 9 (3): 403–14. 2005. doi:10.1016/j.devcel.2005.07.009. PMID 16139228.
- "Regulation by let-7 and lin-4 miRNAs results in target mRNA degradation". Cell 122 (4): 553–63. 2005. doi:10.1016/j.cell.2005.07.031. PMID 16122423.
- "Inhibition of translational initiation by Let-7 MicroRNA in human cells". Science 309 (5740): 1573–6. 2005. doi:10.1126/science.1115079. PMID 16081698. Bibcode: 2005Sci...309.1573P.
- "The temporal patterning microRNA let-7 regulates several transcription factors at the larval to adult transition in C. elegans". Dev Cell 8 (3): 321–30. 2005. doi:10.1016/j.devcel.2004.12.019. PMID 15737928.
- "Substrate requirements for let-7 function in the developing zebrafish embryo". Nucleic Acids Res 32 (21): 6284–91. 2004. doi:10.1093/nar/gkh968. PMID 15585662.
- "Trans-splicing and polyadenylation of let-7 microRNA primary transcripts". RNA 10 (10): 1586–94. 2004. doi:10.1261/rna.7122604. PMID 15337850.
- "The C. elegans microRNA let-7 binds to imperfect let-7 complementary sites from the lin-41 3′UTR". Genes Dev 18 (2): 132–7. 2004. doi:10.1101/gad.1165404. PMID 14729570.
- "Human let-7 stem–loop precursors harbor features of RNase III cleavage products". Nucleic Acids Res 31 (22): 6593–7. 2003. doi:10.1093/nar/gkg855. PMID 14602919.
- "The time of appearance of the C. elegans let-7 microRNA is transcriptionally controlled utilizing a temporal regulatory element in its promoter". Dev Biol 259 (2): 364–79. 2003. doi:10.1016/S0012-1606(03)00202-1. PMID 12871707.
- "Expression of the 22 nucleotide let-7 heterochronic RNA throughout the Metazoa: a role in life history evolution?". Evol Dev 5 (4): 372–8. 2003. doi:10.1046/j.1525-142X.2003.03044.x. PMID 12823453.
- "The expression of the let-7 small regulatory RNA is controlled by ecdysone during metamorphosis in Drosophila melanogaster". Dev Biol 244 (1): 170–9. 2002. doi:10.1006/dbio.2002.0594. PMID 11900466.
- "A cellular function for the RNA-interference enzyme Dicer in the maturation of the let-7 small temporal RNA". Science 293 (5531): 834–8. 2001. doi:10.1126/science.1062961. PMID 11452083.
- "The lin-41 RBCC gene acts in the C. elegans heterochronic pathway between the let-7 regulatory RNA and the LIN-29 transcription factor". Mol Cell 5 (4): 659–69. 2000. doi:10.1016/S1097-2765(00)80245-2. PMID 10882102.
- Lin, S; Li, H; Mu, H; Luo, W; Li, Y; Jia, X; Wang, S; Jia, X et al. (Jul 10, 2012). "Let-7b regulates the expression of the growth hormone receptor gene in deletion-type dwarf chickens.". BMC Genomics 13: 306. doi:10.1186/1471-2164-13-306. PMID 22781587.
- Shen, Y; Wollam, J; Magner, D; Karalay, O; Antebi, A (Dec 14, 2012). "A steroid receptor-microRNA switch regulates life span in response to signals from the gonad.". Science 338 (6113): 1472–6. doi:10.1126/science.1228967. PMID 23239738. Bibcode: 2012Sci...338.1472S.
- Repetto, E; Briata, P; Kuziner, N; Harfe, BD; McManus, MT; Gherzi, R; Rosenfeld, MG; Trabucchi, M (2012). "Let-7b/c enhance the stability of a tissue-specific mRNA during mammalian organogenesis as part of a feedback loop involving KSRP.". PLOS Genetics 8 (7): e1002823. doi:10.1371/journal.pgen.1002823. PMID 22844247.
External links
