Biology:Cilium
The cilium (pl.: cilia; from la cilium 'eyelash'; in Medieval Latin and in anatomy, cilium) is a short hair-like membrane protrusion from many types of eukaryotic cell.[1][2] (Cilia are absent in bacteria and archaea.) The cilium has the shape of a slender threadlike projection that extends from the surface of the much larger cell body.[2] Eukaryotic flagella found on sperm cells and many protozoans have a similar structure to motile cilia that enables swimming through liquids, but they are longer than cilia and have a different undulating motion.[3][4]
There are two major classes of cilia: motile and non-motile cilia, each with two subtypes, giving four types in all.[5] A cell will typically have one primary cilium or many motile cilia.[6] The structure of the cilium core, called the axoneme, determines the cilium class. Most motile cilia have a central pair of single microtubules surrounded by nine pairs of double microtubules called a 9+2 axoneme. Most non-motile cilia have a 9+0 axoneme that lacks the central pair of microtubules. Also lacking are the associated components that enable motility including the outer and inner dynein arms, and radial spokes.[7] Some motile cilia lack the central pair, and some non-motile cilia have the central pair, hence the four types.[5][7]
Most non-motile cilia, termed primary cilia or sensory cilia, serve solely as sensory organelles.[8][9] Most vertebrate cell types possess a single non-motile primary cilium, which functions as a cellular antenna.[10][11] Olfactory neurons possess a great many non-motile cilia. Non-motile cilia that have a central pair of microtubules are the kinocilia present on hair cells.[5]
Motile cilia are found in large numbers on respiratory epithelial cells – around 200 cilia per cell, where they function in mucociliary clearance, and also have mechanosensory and chemosensory functions.[12][13][14] Motile cilia on ependymal cells move the cerebrospinal fluid through the ventricular system of the brain. Motile cilia are also present in the oviducts (fallopian tubes) of female (therian) mammals, where they function in moving egg cells from the ovary to the uterus.[13][15] Motile cilia that lack the central pair of microtubules are found in the cells of the embryonic primitive node; termed nodal cells, these nodal cilia are responsible for the left-right asymmetry of bilaterians.[16]
Structure
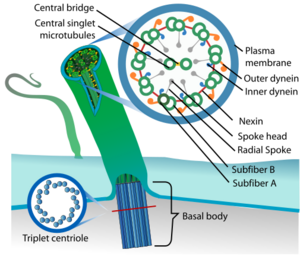
Cilia can be between one and five micrometers in length.[17] A cilium is assembled and built from a basal body on the cell surface. From the basal body, the ciliary rootlet forms ahead of the transition plate and transition zone where the earlier microtubule triplets change to the microtubule doublets of the axoneme.
Basal body
The foundation of the cilium is the basal body, a modified mother centriole on the cell surface.[18] Mammalian basal bodies consist of a barrel of nine triplet microtubules, subdistal appendages and nine strut-like structures, known as distal appendages, which attach the basal body to the membrane at the base of the cilium. Two of each of the basal body's triplet microtubules extend during growth of the axoneme to become the doublet microtubules.
Ciliary rootlet
The ciliary rootlet is a cytoskeleton-like structure that originates from the basal body at the proximal end of a cilium. Rootlets are typically 80-100 nm in diameter and contain cross striae distributed at regular intervals of approximately 55-70 nm. A prominent component of the rootlet is rootletin a coiled coil rootlet protein coded for by the CROCC gene.[19]
Transition zone
To achieve its distinct composition, the proximal-most region of the cilium consists of a transition zone, also known as the ciliary gate, that controls the entry and exit of proteins to and from the cilium.[20][21][22] At the transition zone, Y-shaped structures connect the ciliary membrane to the underlying axoneme. Control of selective entry into cilia may involve a sieve-like function of transition zone. Inherited defects in components of the transition zone cause ciliopathies, such as Joubert syndrome. Transition zone structure and function is conserved across diverse organisms, including vertebrates, Caenorhabditis elegans, Drosophila melanogaster and Chlamydomonas reinhardtii. In mammals, disruption of the transition zone reduces the ciliary abundance of membrane-associated ciliary proteins, such as those involved in Hedgehog signal transduction, compromising Hedgehog-dependent embryonic development of digit number and central nervous system patterning.
Axoneme
Inside a cilium is a microtubule-based cytoskeletal core called the axoneme. The axoneme of a primary cilium typically has a ring of nine outer microtubule doublets (called a 9+0 axoneme), and the axoneme of a motile cilium has, in addition to the nine outer doublets, two central microtubule singlets (called a 9+2 axoneme). This is the same axoneme type of the flagellum. The axoneme in a motile cilium acts as a scaffold for the inner and outer dynein arms that move the cilium, and provides tracks for the microtubule motor proteins of kinesin and dynein.[2][23][24] The transport of ciliary components is carried out by intraflagellar transport (IFT) which is similar to the axonal transport in a nerve fibre. Transport is bidirectional and cytoskeletal motor proteins kinesin and dynein transport ciliary components along the microtubule tracks; kinesin in an anterograde movement towards the ciliary tip and dynein in a retrograde movement towards the cell body. The cilium has its own ciliary membrane enclosed within the surrounding cell membrane.[25]
Types
Non-motile cilia
In animals, non-motile primary cilia are found on nearly every type of cell, blood cells being a prominent exception.[2] Most cells only possess one, in contrast to cells with motile cilia, an exception being olfactory sensory neurons, where the odorant receptors are located, which each possess about ten cilia. Some cell types, such as retinal photoreceptor cells, possess highly specialized primary cilia.[26]
Although the primary cilium was discovered in 1898, it was largely ignored for a century and considered a vestigial organelle without important function.[27][2] Recent findings regarding its physiological roles in chemosensation, signal transduction, and cell growth control, have revealed its importance in cell function. Its importance to human biology has been underscored by the discovery of its role in a diverse group of diseases caused by the dysgenesis or dysfunction of cilia, such as polycystic kidney disease,[28] congenital heart disease,[29] mitral valve prolapse,[30] and retinal degeneration,[31] called ciliopathies.[32][33] The primary cilium is now known to play an important role in the function of many human organs.[2][10] Primary cilia on pancreatic beta cells regulate their function and energy metabolism. Cilia deletion can lead to islet dysfunction and type 2 diabetes.[34]
Cilia are assembled during the G1 phase and are disassembled before mitosis occurs.[35][11] Disassembly of cilia requires the action of aurora kinase A.[36] The current scientific understanding of primary cilia views them as "sensory cellular antennae that coordinate many cellular signaling pathways, sometimes coupling the signaling to ciliary motility or alternatively to cell division and differentiation."[37] The cilium is composed of subdomains[clarification needed] and enclosed by a plasma membrane continuous with the plasma membrane of the cell. For many cilia, the basal body, where the cilium originates, is located within a membrane invagination called the ciliary pocket. The cilium membrane and the basal body microtubules are connected by distal appendages (also called transition fibers). Vesicles carrying molecules for the cilia dock at the distal appendages. Distal to the transition fibers form a transition zone where entry and exit of molecules is regulated to and from the cilia. Some of the signaling with these cilia occur through ligand binding such as Hedgehog signaling.[38] Other forms of signaling include G protein-coupled receptors including the somatostatin receptor 3 in neurons.[39]

Modified non-motile cilia
In the inner ear, kinocilia that are termed as specialized primary cilia, or modified non-motile cilia are found on the hair cells. They possess the 9+2 axoneme of the motile cilia, but lack the inner dynein arms that give movement. They do move passively following the detection of sound, allowed by the outer dynein arms.[40][41]
Motile cilia

Mammals also have motile cilia or secondary cilia that are usually present on a cell's surface in large numbers (multiciliate), and beat in coordinated metachronal waves.[42] Multiciliated cells are found lining the respiratory tract where they function in mucociliary clearance sweeping mucus containing debris away from the lungs.[13] Each cell in the respiratory epithelium has around 200 motile cilia.[12]
In the reproductive tract, smooth muscle contractions help the beating of the cilia in moving the egg cell from the ovary to the uterus.[13][15]
In the ventricles of the brain ciliated ependymal cells circulate the cerebrospinal fluid.
The functioning of motile cilia is strongly dependent on the maintenance of optimal levels of periciliary fluid bathing the cilia. Epithelial sodium channels (ENaCs) are specifically expressed along the entire length of cilia in the respiratory tract, and fallopian tube or oviduct that apparently serve as sensors to regulate the periciliary fluid.[13][43]
Modified motile cilia
Motile cilia without the central pair of singlets (9+0) are found in early embryonic development. They are present as nodal cilia on the nodal cells of the primitive node. Nodal cells are responsible for the left-right asymmetry in bilateral animals.[16] While lacking the central apparatus there are dynein arms present that allow the nodal cilia to move in a spinning fashion. The movement creates a current flow of the extraembryonic fluid across the nodal surface in a leftward direction that initiates the left-right asymmetry in the developing embryo.[12][44]
Motile, multiple, 9+0 cilia are found on the epithelial cells of the choroid plexus. Cilia also can change structure when introduced to hot temperatures and become sharp. They are present in large numbers on each cell and move relatively slowly, making them intermediate between motile and primary cilia. In addition to 9+0 cilia that are mobile, there are also solitary 9+2 cilia that stay immobile found in hair cells.[41]
Nodal cilia

Nodal cells have a single cilium called a monocilium. They are present in the very early development of the embryo on the primitive node. There are two areas of the node with different types of nodal cilia. On the central node are motile cilia, and on the peripheral area of the node the nodal cilia are modified motile.[44]
The motile cilia on the central cells rotate to generate the leftward flow of extracellular fluid needed to initiate the left-right asymmetry.[44]
Cilia versus flagella
The motile cilia on sperm cells and many protozoans enables swimming through liquids and are traditionally referred to as "flagella".[3] As these protrusions are structurally identical to motile cilia, attempts at preserving this terminology include making a distinction by morphology ("flagella" are typically longer than ordinary cilia and have a different undulating motion)[4] and by number.[45]
Microorganisms
Ciliates are eukaryotic microorganisms that possess motile cilia exclusively and use them for either locomotion or to simply move liquid over their surface. A Paramecium for example is covered in thousands of cilia that enable its swimming. These motile cilia have been shown to be sensory as well.[46]
Ciliogenesis
Cilia are formed through the process of ciliogenesis. An early step is docking of the basal body to the growing ciliary membrane, after which the transition zone forms. The building blocks of the ciliary axoneme, such as tubulins, are added at the ciliary tips through a process that depends partly on intraflagellar transport (IFT).[47][48] Exceptions include Drosophila sperm and Plasmodium falciparum flagella formation, in which cilia assemble in the cytoplasm.[49]
At the base of the cilium where it attaches to the cell body is the microtubule organizing center, the basal body. Some basal body proteins such as CEP164, ODF2,[50] and CEP170,[51] are required for the formation and the stability of the cilium.
In effect, the cilium is a nanomachine composed of perhaps more than 600 proteins in molecular complexes, many of which also function independently as nanomachines. Flexible linkers allow the mobile protein domains connected by them to recruit their binding partners and induce long-range allostery via protein domain dynamics.[37]
Function
The dynein in the axoneme – axonemal dynein forms bridges between neighbouring microtubule doublets. When ATP activates the motor domain of dynein, it attempts to walk along the adjoining microtubule doublet. This would force the adjacent doublets to slide over one another if not for the presence of nexin between the microtubule doublets. And thus the force generated by dynein is instead converted into a bending motion.[52][53]
Sensing the extracellular environment
Some primary cilia on epithelial cells in eukaryotes act as cellular antennae, providing chemosensation, thermosensation and mechanosensation of the extracellular environment.[54][10] These cilia then play a role in mediating specific signalling cues, including soluble factors in the external cell environment, a secretory role in which a soluble protein is released to have an effect downstream of the fluid flow, and mediation of fluid flow if the cilia are motile.[54]
Some epithelial cells are ciliated, and they commonly exist as a sheet of polarized cells forming a tube or tubule with cilia projecting into the lumen. This sensory and signalling role puts cilia in a central role for maintaining the local cellular environment and may be why ciliary defects cause such a wide range of human diseases.[33]
In the embryo, nodal cilia are used to direct the flow of extracellular fluid. This leftward movement is to generate left-right asymmetry across the midline of the embryo. Central cilia coordinate their rotational beating while the immotile cilia on the sides sense the direction of the flow.[44][55][56] Studies in mice suggest a biophysical mechanism by which the direction of flow is sensed.[57]
Axo-ciliary synapse
With axo-ciliary synapses, there is communication between serotonergic axons and primary cilia of CA1 pyramidal neurons that alters the neuron's epigenetic state in the nucleus – "a way to change what is being transcribed or made in the nucleus" via this signalling distinct from that at the plasma membrane which also is longer-term.[58][59]
Clinical significance
Ciliary defects can lead to a number of human diseases.[33][60] Defects in cilia adversely affect many critical signaling pathways essential to embryonic development and to adult physiology, and thus offer a plausible hypothesis for the often multi-symptom nature of diverse ciliopathies.[32][33] Known ciliopathies include primary ciliary dyskinesia, Bardet–Biedl syndrome, polycystic kidney and liver disease, nephronophthisis, Alström syndrome, Meckel–Gruber syndrome, Sensenbrenner syndrome and some forms of retinal degeneration.[32][54] Genetic mutations compromising the proper functioning of cilia, ciliopathies, may cause chronic disorders such as primary ciliary dyskinesia (PCD), nephronophthisis, and Senior–Løken syndrome. In addition, a defect of the primary cilium in the renal tubule cells may lead to polycystic kidney disease (PKD). In another genetic disorder called Bardet–Biedl syndrome (BBS), the mutant gene products are the components in the basal body and cilia.[32]
Defects in cilia cells are linked to obesity and often pronounced in type 2 diabetes. Several studies already showed impaired glucose tolerance and reduction in the insulin secretion in the ciliopathy models. Moreover, the number and length of cilia was decreased in the type 2 diabetes models.[61]
Epithelial sodium channels (ENaCs) that are expressed along the length of cilia regulate periciliary fluid level. Mutations that decrease the activity of ENaCs result in multisystem pseudohypoaldosteronism, that is associated with fertility problems.[13] In cystic fibrosis that results from mutations in the chloride channel CFTR, ENaC activity is enhanced leading to a severe reduction of the fluid level that causes complications and infections in the respiratory airways.[43]
Since the flagellum of human sperm has the same internal structure of a cilium, ciliary dysfunction may be responsible for male infertility as well.[62]
There is an association of primary ciliary dyskinesia with left-right anatomic abnormalities such as situs inversus (a combination of findings is known as Kartagener syndrome), and situs ambiguus (also known as Heterotaxy syndrome).[63] These left-right anatomic abnormalities can also result in congenital heart disease.[64]
It has been shown that proper cilial function is responsible for the normal left-right asymmetry in mammals.[65]
The diverse outcomes caused by ciliary dysfunction may result from alleles of different strengths that compromise ciliary functions in different ways or to different extents. Many ciliopathies are inherited in a Mendelian manner, but specific genetic interactions between distinct functional ciliary complexes, such as transition zone and BBS complexes, can alter the phenotypic manifestations of recessive ciliopathies.[66][67] Some mutations in transition zone proteins can cause specific serious ciliopathies.[68]
Extracellular changes
Reduction of cilia function can also result from infection. Research into biofilms has shown that bacteria can alter cilia. A biofilm is a community of bacteria of either the same or multiple species of bacteria. The cluster of cells secretes different factors which form an extracellular matrix. Cilia in the respiratory system is known to move mucus and pathogens out of the airways. It has been found that patients with biofilm positive infections have impaired cilia function. The impairment may present as decreased motion or reduction in the number of cilia. Though these changes result from an external source, they still effect the pathogenicity of the bacteria, progression of infection, and how it is treated.[69]
The transportation of the immature egg cell, and the embryo to the uterus for implantation depends on the combination of regulated smooth muscle contractions, and ciliary beating. Dysfunction in this transportation can result in an ectopic pregnancy where the embryo is implanted (usually) in the fallopian tube before reaching its proper destination of the uterus. Many factors can affect this stage including infection and menstrual cycle hormones. Smoking (causing inflammation), and infection may reduce the numbers of cilia, and the ciliary beat may be affected by hormonal changes.[15][70]
Primary cilia in pancreatic cells
The pancreas is a mixture of highly differentiated exocrine and endocrine cells. Primary cilia are present in exocrine cells, which are centroacinar duct cells.[71][34] Endocrine tissue is composed of different hormone-secreting cells. Insulin-secreting beta cells and glucagon-secreting alpha cells are highly ciliated.[72][73]
See also
References
- ↑ "Definition of CILIUM" (in en). https://www.merriam-webster.com/dictionary/cilium.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Gardiner, Mary Beth (September 2005). "The Importance of Being Cilia". HHMI Bulletin 18 (2). http://www.hhmi.org/sites/default/files/Bulletin/2005/September/sept2005_fulltext.pdf. Retrieved 26 July 2008.
- ↑ 3.0 3.1 "Cilia, flagella, and microtubules". The Journal of Cell Biology 91 (3 Pt 2): 125s–130s. December 1981. doi:10.1083/jcb.91.3.125s. PMID 6459327.
- ↑ 4.0 4.1 Alberts, Bruce (2015). Molecular biology of the cell (6 ed.). New York, NY. pp. 941–942. ISBN 978-0-8153-4464-3.
- ↑ 5.0 5.1 5.2 Falk, N; Lösl, M; Schröder, N; Gießl, A (11 September 2015). "Specialized Cilia in Mammalian Sensory Systems". Cells 4 (3): 500–19. doi:10.3390/cells4030500. PMID 26378583.
- ↑ Wheatley, DN (September 2021). "Primary cilia: turning points in establishing their ubiquity, sensory role and the pathological consequences of dysfunction". Journal of Cell Communication and Signaling 15 (3): 291–297. doi:10.1007/s12079-021-00615-5. PMID 33970456.
- ↑ 7.0 7.1 Fisch, C; Dupuis-Williams, P (June 2011). "Ultrastructure of cilia and flagella - back to the future!". Biology of the Cell 103 (6): 249–70. doi:10.1042/BC20100139. PMID 21728999.
- ↑ Prevo, B; Scholey, JM; Peterman, EJG (September 2017). "Intraflagellar transport: mechanisms of motor action, cooperation, and cargo delivery". The FEBS Journal 284 (18): 2905–2931. doi:10.1111/febs.14068. PMID 28342295.
- ↑ Elliott, Kelsey H.; Brugmann, Samantha A. (1 March 2019). "Sending mixed signals: Cilia-dependent signaling during development and disease". Developmental Biology 447 (1): 28–41. doi:10.1016/j.ydbio.2018.03.007. ISSN 1095-564X. PMID 29548942.
- ↑ 10.0 10.1 10.2 Singla, Veena; Reiter, Jeremy F. (2006-08-04). "The primary cilium as the cell's antenna: signaling at a sensory organelle". Science 313 (5787): 629–633. doi:10.1126/science.1124534. ISSN 1095-9203. PMID 16888132. Bibcode: 2006Sci...313..629S.
- ↑ 11.0 11.1 Patel, MM; Tsiokas, L (1 November 2021). "Insights into the Regulation of Ciliary Disassembly". Cells 10 (11): 2977. doi:10.3390/cells10112977. PMID 34831200.
- ↑ 12.0 12.1 12.2 Horani, A; Ferkol, T (May 2018). "Advances in the Genetics of Primary Ciliary Dyskinesia". Chest 154 (3): 645–652. doi:10.1016/j.chest.2018.05.007. PMID 29800551.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 "Epithelial sodium channels (ENaC) are uniformly distributed on motile cilia in the oviduct and the respiratory airways". Histochemistry and Cell Biology 137 (3): 339–53. March 2012. doi:10.1007/s00418-011-0904-1. PMID 22207244.
- ↑ Bloodgood, RA (15 February 2010). "Sensory reception is an attribute of both primary cilia and motile cilia". Journal of Cell Science 123 (Pt 4): 505–9. doi:10.1242/jcs.066308. PMID 20144998.
- ↑ 15.0 15.1 15.2 Panelli, DM; Phillips, CH; Brady, PC (2015). "Incidence, diagnosis and management of tubal and nontubal ectopic pregnancies: a review.". Fertility Research and Practice 1: 15. doi:10.1186/s40738-015-0008-z. PMID 28620520.
- ↑ 16.0 16.1 Desgrange, A; Le Garrec, JF; Meilhac, SM (22 November 2018). "Left-right asymmetry in heart development and disease: forming the right loop". Development 145 (22). doi:10.1242/dev.162776. PMID 30467108. https://hal.archives-ouvertes.fr/hal-03094768/file/dev162776.full.pdf.
- ↑ Louvi, A; Grove, EA (24 March 2011). "Cilia in the CNS: the quiet organelle claims center stage.". Neuron 69 (6): 1046–60. doi:10.1016/j.neuron.2011.03.002. PMID 21435552.
- ↑ Ishikawa, T (3 January 2017). "Axoneme Structure from Motile Cilia.". Cold Spring Harbor Perspectives in Biology 9 (1). doi:10.1101/cshperspect.a028076. PMID 27601632.
- ↑ "Rootelin". https://www.uniprot.org/uniprot/Q5TZA2.
- ↑ Garcia, Galo; Raleigh, David R.; Reiter, Jeremy F. (23 April 2018). "How the Ciliary Membrane Is Organized Inside-Out to Communicate Outside-In". Current Biology 28 (8): R421–R434. doi:10.1016/j.cub.2018.03.010. ISSN 1879-0445. PMID 29689227. Bibcode: 2018CBio...28.R421G.
- ↑ Garcia-Gonzalo, Francesc R.; Reiter, Jeremy F. (2017-02-01). "Open Sesame: How Transition Fibers and the Transition Zone Control Ciliary Composition". Cold Spring Harbor Perspectives in Biology 9 (2). doi:10.1101/cshperspect.a028134. ISSN 1943-0264. PMID 27770015.
- ↑ Gonçalves, João; Pelletier, Laurence (April 2017). "The Ciliary Transition Zone: Finding the Pieces and Assembling the Gate". Molecules and Cells 40 (4): 243–253. doi:10.14348/molcells.2017.0054. ISSN 0219-1032. PMID 28401750.
- ↑ "Intraflagellar transport". Nature Reviews. Molecular Cell Biology 3 (11): 813–25. November 2002. doi:10.1038/nrm952. PMID 12415299.
- ↑ "Intraflagellar transport motors in cilia: moving along the cell's antenna". The Journal of Cell Biology 180 (1): 23–29. January 2008. doi:10.1083/jcb.200709133. PMID 18180368.
- ↑ "The ciliary membrane". Current Opinion in Cell Biology 22 (4): 541–46. August 2010. doi:10.1016/j.ceb.2010.03.010. PMID 20399632.
- ↑ Wolfrum, U; Schmitt, A (June 2000). "Rhodopsin transport in the membrane of the connecting cilium of mammalian photoreceptor cells.". Cell Motility and the Cytoskeleton 46 (2): 95–107. doi:10.1002/1097-0169(200006)46:2<95::AID-CM2>3.0.CO;2-Q. PMID 10891855.
- ↑ Satir, Peter (2017). "CILIA: before and after". Cilia 6. doi:10.1186/s13630-017-0046-8. ISSN 2046-2530. PMID 28293419.
- ↑ "News from the cyst: insights into polycystic kidney disease". Journal of Nephrology 21 (1): 14–16. 2008. PMID 18264930. http://www.jnephrol.com/Article.action?cmd=navigate&urlkey=Public_Details&t=JN&uid=9A13E591-2E27-441B-8356-23BF86D9CFB0. Retrieved 1 March 2010.
- ↑ "Heterotaxia, congenital heart disease, and primary ciliary dyskinesia". Circulation 115 (22): 2793–95. June 2007. doi:10.1161/CIRCULATIONAHA.107.699256. PMID 17548739.
- ↑ "Primary cilia defects causing mitral valve prolapse". Sci. Transl. Med. 11 (493). 2019. doi:10.1126/scitranslmed.aax0290. PMID 31118289.
- ↑ Chen, Holly Y.; Kelley, Ryan A.; Li, Tiansen; Swaroop, Anand (2020-07-31). "Primary cilia biogenesis and associated retinal ciliopathies". Seminars in Cell & Developmental Biology 110: 70–88. doi:10.1016/j.semcdb.2020.07.013. ISSN 1096-3634. PMID 32747192.
- ↑ 32.0 32.1 32.2 32.3 "The ciliopathies: an emerging class of human genetic disorders". Annual Review of Genomics and Human Genetics 7: 125–48. 2006. doi:10.1146/annurev.genom.7.080505.115610. PMID 16722803.
- ↑ 33.0 33.1 33.2 33.3 Reiter, Jeremy F.; Leroux, Michel R. (September 2017). "Genes and molecular pathways underpinning ciliopathies". Nature Reviews. Molecular Cell Biology 18 (9): 533–547. doi:10.1038/nrm.2017.60. ISSN 1471-0080. PMID 28698599.
- ↑ 34.0 34.1 Hegyi, P; Petersen, OH (2013). "The exocrine pancreas: the acinar-ductal tango in physiology and pathophysiology.". Reviews of Physiology, Biochemistry and Pharmacology 165: 1–30. doi:10.1007/112_2013_14. ISBN 978-3-319-00998-8. PMID 23881310.
- ↑ "The primary cilium: keeper of the key to cell division". Cell 129 (7): 1255–57. June 2007. doi:10.1016/j.cell.2007.06.018. PMID 17604715.
- ↑ "HEF1-dependent Aurora A activation induces disassembly of the primary cilium". Cell 129 (7): 1351–63. June 2007. doi:10.1016/j.cell.2007.04.035. PMID 17604723.
- ↑ 37.0 37.1 "Structure and function of mammalian cilia". Histochemistry and Cell Biology 129 (6): 687–93. June 2008. doi:10.1007/s00418-008-0416-9. PMID 18365235.
- ↑ Wong, Sunny Y.; Reiter, Jeremy F. (2008). "The primary cilium at the crossroads of mammalian hedgehog signaling". Current Topics in Developmental Biology 85: 225–260. doi:10.1016/S0070-2153(08)00809-0. ISSN 0070-2153. PMID 19147008.
- ↑ "Signaling through the Primary Cilium". Frontiers in Cell and Developmental Biology 6. 2018. doi:10.3389/fcell.2018.00008. PMID 29473038.
- ↑ Wang, D; Zhou, J (2021). "The Kinocilia of Cochlear Hair Cells: Structures, Functions, and Diseases.". Frontiers in Cell and Developmental Biology 9. doi:10.3389/fcell.2021.715037. PMID 34422834.
- ↑ 41.0 41.1 Takeda, Sen; Narita, Keishi (February 2012). "Structure and function of vertebrate cilia, towards a new taxonomy". Differentiation 83 (2): S4–S11. doi:10.1016/j.diff.2011.11.002. PMID 22118931.
- ↑ Benjamin Lewin (2007). Cells. Jones & Bartlett Learning. p. 359. ISBN 978-0-7637-3905-8. https://books.google.com/books?id=2VEGC8j9g9wC&pg=PA359.
- ↑ 43.0 43.1 "Epithelial sodium channel (ENaC) family: Phylogeny, structure-function, tissue distribution, and associated inherited diseases". Gene 579 (2): 95–132. April 2016. doi:10.1016/j.gene.2015.12.061. PMID 26772908.
- ↑ 44.0 44.1 44.2 44.3 Schoenwolf, Gary C. (2015). Larsen's human embryology (Fifth ed.). Philadelphia, PA. p. 64. ISBN 978-1-4557-0684-6.
- ↑ Lindemann, CB; Lesich, KA (15 February 2010). "Flagellar and ciliary beating: the proven and the possible.". Journal of Cell Science 123 (Pt 4): 519–28. doi:10.1242/jcs.051326. PMID 20145000.
- ↑ Valentine, M; Van Houten, J (24 September 2021). "Using Paramecium as a Model for Ciliopathies.". Genes 12 (10): 1493. doi:10.3390/genes12101493. PMID 34680887.
- ↑ "Polarity of flagellar assembly in Chlamydomonas". The Journal of Cell Biology 119 (6): 1605–11. December 1992. doi:10.1083/jcb.119.6.1605. PMID 1281816.
- ↑ "Intraflagellar transport delivers tubulin isotypes to sensory cilium middle and distal segments". Nature Cell Biology 13 (7): 790–98. June 2011. doi:10.1038/ncb2268. PMID 21642982.
- ↑ Of cilia and silliness (more on Behe) – The Panda's Thumb
- ↑ "Odf2-deficient mother centrioles lack distal/subdistal appendages and the ability to generate primary cilia". Nature Cell Biology 7 (5): 517–24. May 2005. doi:10.1038/ncb1251. PMID 15852003.
- ↑ Lamla S (2009-01-22). Functional characterisation of the centrosomal protein Cep170 (Ph.D.). Ludwig-Maximilians-Universität München.
- ↑ Alberts, Bruce (2002). "Molecular Motors". Molecular Biology of the Cell. Garland Science. https://www.ncbi.nlm.nih.gov/books/NBK26888/.[ISBN missing]
- ↑ King, SM (1 November 2016). "Axonemal Dynein Arms.". Cold Spring Harbor Perspectives in Biology 8 (11). doi:10.1101/cshperspect.a028100. PMID 27527589.
- ↑ 54.0 54.1 54.2 "Recent advances in the molecular pathology, cell biology and genetics of ciliopathies". Journal of Medical Genetics 45 (5): 257–67. May 2008. doi:10.1136/jmg.2007.054999. PMID 18178628.
- ↑ Wolpert, Lewis; Tickle, Cheryll; Martinez Arias, Alfonso (2015). Principles of Development (5th ed.). Oxford University Press. p. 227.
- ↑ Cilia function as calcium-mediated mechanosensors that instruct left-right asymmetry, Science, 5 January 2023, Vol 379, Issue 6627, pp. 71-78; DOI: 10.1126/science.abq7317
- ↑ Immotile cilia mechanically sense the direction of fluid flow for left-right determination, Science, 5 January 2023, Vol 379, Issue 6627, pp. 66-71; DOI: 10.1126/science.abq8148
- ↑ Tamim, Baba (4 September 2022). "New discovery: Synapse hiding in the mice brain may advance our understanding of neuronal communication". interestingengineering.com. https://interestingengineering.com/science/new-discovery-synapse-hiding-in-mice-brain.
- ↑ Sheu, Shu-Hsien; Upadhyayula, Srigokul; Dupuy, Vincent; Pang, Song; Deng, Fei; Wan, Jinxia; Walpita, Deepika; Pasolli, H. Amalia et al. (1 September 2022). "A serotonergic axon-cilium synapse drives nuclear signaling to alter chromatin accessibility" (in English). Cell 185 (18): 3390–3407.e18. doi:10.1016/j.cell.2022.07.026. ISSN 0092-8674. PMID 36055200.
- University press release: "Scientists discover new kind of synapse in neurons' tiny hairs" (in en). Howard Hughes Medical Institute via phys.org. https://phys.org/news/2022-09-scientists-kind-synapse-neurons-tiny.html.
- ↑ Braun, Daniela A.; Hildebrandt, Friedhelm (2017-03-01). "Ciliopathies". Cold Spring Harbor Perspectives in Biology 9 (3). doi:10.1101/cshperspect.a028191. ISSN 1943-0264. PMID 27793968.
- ↑ Gerdes, Jantje M.; Christou-Savina, Sonia; Xiong, Yan; Moede, Tilo; Moruzzi, Noah; Karlsson-Edlund, Patrick; Leibiger, Barbara; Leibiger, Ingo B. et al. (2014). "Ciliary dysfunction impairs beta-cell insulin secretion and promotes development of type 2 diabetes in rodents". Nature Communications 5. doi:10.1038/ncomms6308. PMID 25374274. Bibcode: 2014NatCo...5.5308G.
- ↑ "Obstructive azoospermia associated with chronic sinopulmonary infection and situs inversus totalis". Urology 68 (1): 204.e5–7. July 2006. doi:10.1016/j.urology.2006.01.072. PMID 16850538.
- ↑ Worsley, Calum; Weerakkody, Yuranga (1 November 2009). Heterotaxy syndrome. doi:10.53347/rID-7420. https://radiopaedia.org/articles/heterotaxy-syndrome?lang=gb. Retrieved 10 June 2022.
- ↑ "Congenital heart disease and other heterotaxic defects in a large cohort of patients with primary ciliary dyskinesia". Circulation 115 (22): 2814–21. June 2007. doi:10.1161/CIRCULATIONAHA.106.649038. PMID 17515466.
- ↑ "Cilia are at the heart of vertebrate left-right asymmetry". Current Opinion in Genetics & Development 13 (4): 385–92. August 2003. doi:10.1016/S0959-437X(03)00091-1. PMID 12888012.
- ↑ Leitch, Carmen C.; Zaghloul, Norann A.; Davis, Erica E.; Stoetzel, Corinne; Diaz-Font, Anna; Rix, Suzanne; Alfadhel, Majid; Al-Fadhel, Majid et al. (April 2008). "Hypomorphic mutations in syndromic encephalocele genes are associated with Bardet-Biedl syndrome". Nature Genetics 40 (4): 443–448. doi:10.1038/ng.97. ISSN 1546-1718. PMID 18327255.
- ↑ Yee, Laura E.; Garcia-Gonzalo, Francesc R.; Bowie, Rachel V.; Li, Chunmei; Kennedy, Julie K.; Ashrafi, Kaveh; Blacque, Oliver E.; Leroux, Michel R. et al. (November 2015). "Conserved Genetic Interactions between Ciliopathy Complexes Cooperatively Support Ciliogenesis and Ciliary Signaling". PLOS Genetics 11 (11). doi:10.1371/journal.pgen.1005627. ISSN 1553-7404. PMID 26540106.
- ↑ Cavalier-Smith, T (May 2022). "Ciliary transition zone evolution and the root of the eukaryote tree: implications for opisthokont origin and classification of kingdoms Protozoa, Plantae, and Fungi.". Protoplasma 259 (3): 487–593. doi:10.1007/s00709-021-01665-7. PMID 34940909. Bibcode: 2022Prpls.259..487C.
- ↑ "Biofilms in chronic rhinosinusitis: Pathophysiology and therapeutic strategies". World Journal of Otorhinolaryngology – Head and Neck Surgery 2 (4): 219–29. December 2016. doi:10.1016/j.wjorl.2016.03.002. PMID 29204570.
- ↑ "Mechanisms of disease: the endocrinology of ectopic pregnancy". Expert Reviews in Molecular Medicine 14. March 2012. doi:10.1017/erm.2011.2. PMID 22380790.
- ↑ "orpk mouse model of polycystic kidney disease reveals essential role of primary cilia in pancreatic tissue organization". Development 131 (14): 3457–3467. 2004. doi:10.1242/dev.01189. PMID 15226261.
- ↑ "Disruption of IFT results in both exocrine and endocrine abnormalities in the pancreas of Tg737(orpk) mutant mice". Laboratory Investigation 85 (1): 45–64. 2005. doi:10.1038/labinvest.3700207. PMID 15580285.
- ↑ "Electron microscopic observation of the primary cilia in the pancreatic islets". Archivum Histologicum Japonicum 49 (4): 449–457. 1986. doi:10.1679/aohc.49.449. PMID 3545133.
External links
- Brief summary of importance of cilia to many organs in human physiology
- The Ciliary Proteome Web Page at Johns Hopkins
 |
