Biology:TNF receptor superfamily
| TNFR/NGFR cysteine-rich region | |||||||||
|---|---|---|---|---|---|---|---|---|---|
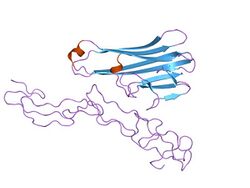 Structure of the soluble human 55 kd TNF receptor-human TNF beta complex.[1] | |||||||||
| Identifiers | |||||||||
| Symbol | TNFR_c6 | ||||||||
| Pfam | PF00020 | ||||||||
| InterPro | IPR011614 | ||||||||
| PROSITE | PDOC00561 | ||||||||
| SCOP2 | 1tnr / SCOPe / SUPFAM | ||||||||
| Membranome | 6 | ||||||||
| |||||||||
The tumor necrosis factor receptor superfamily (TNFRSF) is a protein superfamily of cytokine receptors characterized by the ability to bind tumor necrosis factors (TNFs) via an extracellular cysteine-rich domain.[2][3] With the exception of nerve growth factor (NGF), all TNFs are homologous to the archetypal TNF-alpha.[4] In their active form, the majority of TNF receptors form trimeric complexes in the plasma membrane. Accordingly, most TNF receptors contain transmembrane domains (TMDs), although some can be cleaved into soluble forms (e.g. TNFR1), and some lack a TMD entirely (e.g. DcR3). In addition, most TNF receptors require specific adaptor protein such as TRADD, TRAF, RIP and FADD for downstream signalling. TNF receptors are primarily involved in apoptosis and inflammation, but they can also take part in other signal transduction pathways, such as proliferation, survival, and differentiation. TNF receptors are expressed in a wide variety of tissues in mammals, especially in leukocytes.[4]
The term death receptor refers to those members of the TNF receptor superfamily that contain a death domain, such as TNFR1, Fas receptor, DR4 and DR5.[4] They were named after the fact that they seemed to play an important role in apoptosis (programmed cell death), although they are now known to play other roles as well.[5]
In the strict sense, the term TNF receptor is often used to refer to the archetypal members of the superfamily, namely TNFR1 and TNFR2, which recognize TNF-alpha.
Members
There are 27 family members, numerically classified as TNFRSF#, where # denotes the member number, sometimes followed a letter.[2]
| Type | Protein (member #) | Synonyms | Gene | Ligand(s) |
|---|---|---|---|---|
| 1 | Tumor necrosis factor receptor 1 (1A) | CD120a | TNFRSF1A | TNF (cachectin) |
| Tumor necrosis factor receptor 2 (1B) | CD120b | TNFRSF1B | ||
| 3 | Lymphotoxin beta receptor (3) | CD18 | LTBR | Lymphotoxin beta (TNF-C) |
| 4 | OX40 (4) | CD134 | TNFRSF4 | OX40L |
| 5 | CD40 (5) | Bp50 | CD40 | CD154 |
| 6 | Fas receptor (6) | Apo-1, CD95 | FAS | FasL |
| Decoy receptor 3 (6B) | TR6, M68 | TNFRSF6B | FasL, LIGHT, TL1A | |
| 7 | CD27 (7) | S152, Tp55 | CD27 | CD70, Siva |
| 8 | CD30 (8) | Ki-1, TNR8 | TNFRSF8 | CD153 |
| 9 | 4-1BB (9) | CD137 | TNFRSF9 | 4-1BB ligand |
| 10 | Death receptor 4 (10A) | TRAILR1, Apo-2, CD261 | TNFRSF10A | TRAIL |
| Death receptor 5 (10B) | TRAILR2, CD262 | TNFRSF10B | ||
| Decoy receptor 1 (10C) | TRAILR3, LIT, TRID, CD263 | TNFRSF10C | ||
| Decoy receptor 2 (10D) | TRAILR4, TRUNDD, CD264 | TNFRSF10D | ||
| 11 | RANK (11A) | CD265 | TNFRSF11A | RANKL |
| Osteoprotegerin (11B) | OCIF, TR1 | TNFRSF11B | ||
| 12 | TWEAK receptor (12A) | Fn14, CD266 | TNFRSF12A | TWEAK |
| 13 | TACI (13B) | IGAD2, CD267 | TNFRSF13B | APRIL, BAFF, CAMLG |
| BAFF receptor (13C) | CD268 | TNFRSF13C | BAFF | |
| 14 | Herpesvirus entry mediator (14) | ATAR, TR2, CD270 | TNFRSF14 | LIGHT |
| 16 | Nerve growth factor receptor (16) | p75NTR, CD271 | NGFR | NGF, BDNF, NT-3, NT-4 |
| 17 | B-cell maturation antigen (17) | TNFRSF13A, CD269, BCMA | TNFRSF17 | BAFF |
| 18 | Glucocorticoid-induced TNFR-related (18) | AITR, CD357 | TNFRSF18 | GITR ligand |
| 19 | TROY (19) | TAJ, TRADE | TNFRSF19 | unknown |
| 21 | Death receptor 6 (21) | CD358 | TNFRSF21 | |
| 25 | Death receptor 3 (25) | Apo-3, TRAMP, LARD, WS-1 | TNFRSF25 | TL1A |
| 27 | Ectodysplasin A2 receptor (27) | XEDAR | EDA2R | EDA-A2 |
References
- ↑ Banner DW; D'Arcy A; Janes W et al. (May 1993). "Crystal structure of the soluble human 55 kd TNF receptor-human TNF beta complex: implications for TNF receptor activation". Cell 73 (3): 431–45. doi:10.1016/0092-8674(93)90132-A. PMID 8387891.
- ↑ Jump up to: 2.0 2.1 "The TNF and TNF receptor superfamilies: integrating mammalian biology". Cell 104 (4): 487–501. 2001. doi:10.1016/S0092-8674(01)00237-9. PMID 11239407.
- ↑ "The intriguing biology of the tumour necrosis factor/tumour necrosis factor receptor superfamily: players, rules and the games". Immunology 115 (1): 1–20. 2005. doi:10.1111/j.1365-2567.2005.02143.x. PMID 15819693.
- ↑ Jump up to: 4.0 4.1 4.2 Gravestein, LA; Borst, J (December 1998). "Tumor necrosis factor receptor family members in the immune system.". Seminars in Immunology 10 (6): 423–34. doi:10.1006/smim.1998.0144. PMID 9826575.
- ↑ Ashkenazi, A.; Dixit, VM (1998). "Death Receptors: Signaling and Modulation". Science 281 (5381): 1305–8. doi:10.1126/science.281.5381.1305. PMID 9721089.
Further reading
- "Death receptors and their ligands in atherosclerosis". Arterioscler Thromb Vasc Biol 28 (10): 1694–702. 2008. doi:10.1161/ATVBAHA.107.155143. PMID 18669890.
- Hatano, E. (2007). "Tumor necrosis factor signaling in hepatocyte apoptosis". J Gastroenterol Hepatol 22: S43–44. doi:10.1111/j.1440-1746.2006.04645.x. PMID 17567463.
External links
- Tumor+Necrosis+Factor+Receptor at the US National Library of Medicine Medical Subject Headings (MeSH)
 |

