Chemistry:Chromium(III) iodide
| |
| Names | |
|---|---|
| IUPAC name
Chromium(III) iodide
| |
| Other names
Chromium triiodide, chromic iodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| CrI3 | |
| Molar mass | 432.7095 g·mol−1 |
| Appearance | black solid |
| Density | 5.32 g/cm3[1] |
| Melting point | > 600 °C (1,112 °F; 873 K) |
| Soluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Chromium(III) iodide, also known as chromium triiodide, is an inorganic compound with the formula CrI
3. It is a black solid that is used to prepare other chromium iodides.[2]
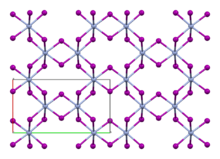
Like the isomorphous chromium(III) chloride (CrCl
3), chromium(III) iodide exhibits a cubic-closest packing arrangement in a double-layer crystal lattice. In this structure, chromium exhibits octahedral coordination geometry.[3]
Preparation and properties
Chromium triiodide is prepared by the direct reaction of chromium metal with an excess of iodine. The reaction is conducted at 500 °C:
- 2 Cr + 3 I
2 → 2 CrI
3
To obtain high purity samples, the product is thermally decomposed at 700 °C to sublime out chromium(II) iodide. The diiodide is then reiodinated.[2]
Chromium triiodide is stable in contact with oxygen and moisture, but at temperatures approaching 200 °C it reacts with oxygen and releases iodine. Like CrCl
3, the triiodide exhibits slow solubility in water owing to the kinetic inertness of Cr(III). Addition of small amounts of chromous iodide accelerates the dissolving process.[2]
Chromium triiodide can also be prepared as nanoplatelets[clarification needed] from the alkoxide Cr(OC(CH
3)(C(CH
3)
3)
2)
3.[4]
Chromium triiodide was one of the first materials which was discovered to be a magnetic two-dimensional material that has great potentials for spintronics devices.[5]
References
- ↑ Perry, Dale L. (2011). Handbook of Inorganic Compounds, Second Edition. Boca Raton, Florida: CRC Press. p. 123. ISBN 978-1-43981462-8. https://books.google.com/books?id=SFD30BvPBhoC. Retrieved 2014-01-10.
- ↑ 2.0 2.1 2.2 Gregory, N. W.; Handy, L. L. (1957). "Chromium (III) Iodide". Inorganic Syntheses. 5. 128–130. doi:10.1002/9780470132364.ch34. ISBN 9780470132364.
- ↑ Gregory, N. W.; Handy, L. L. (1952). "Structural Properties of Chromium(III) Iodide and Some Chromium(III) Mixed Halides". J. Am. Chem. Soc. 74 (4): 891–893. doi:10.1021/ja01124a009.
- ↑ De Siena, Michael C.; Creutz, Sidney E.; Regan, Annie; Malinowski, Paul; Jiang, Qianni; Kluherz, Kyle T.; Zhu, Guomin; Lin, Zhong et al. (2020-03-11). "Two-Dimensional van der Waals Nanoplatelets with Robust Ferromagnetism". Nano Letters 20 (3): 2100–2106. doi:10.1021/acs.nanolett.0c00102. ISSN 1530-6984. PMID 32031382. https://doi.org/10.1021/acs.nanolett.0c00102.
- ↑ Huang, B. (2017). "Layer-dependent ferromagnetism in a van der Waals crystal down to the monolayer limit". Nature 546 (7657): 270–273. doi:10.1038/nature22391. PMID 28593970. Bibcode: 2017Natur.546..270H.
| HI | He | ||||||||||||||||
| LiI | BeI2 | BI3 | CI4 | NI3 | I2O4, I2O5, I4O9 |
IF, IF3, IF5, IF7 |
Ne | ||||||||||
| NaI | MgI2 | AlI3 | SiI4 | PI3, P2I4 |
S | ICl, ICl3 |
Ar | ||||||||||
| KI | CaI2 | Sc | TiI4 | VI3 | CrI3 | MnI2 | FeI2 | CoI2 | NiI2 | CuI | ZnI2 | Ga2I6 | GeI2, GeI4 |
AsI3 | Se | IBr | Kr |
| RbI | SrI2 | YI3 | ZrI4 | NbI5 | Mo | Tc | Ru | Rh | Pd | AgI | CdI2 | InI3 | SnI4, SnI2 |
SbI3 | TeI4 | I | Xe |
| CsI | BaI2 | HfI4 | TaI5 | W | Re | Os | Ir | Pt | AuI | Hg2I2, HgI2 |
TlI | PbI2 | BiI3 | Po | AtI | Rn | |
| Fr | RaI2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | Ce | Pr | Nd | Pm | SmI2 | Eu | Gd | TbI3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | ThI4 | Pa | UI3, UI4 |
Np | Pu | Am | Cm | Bk | Cf | EsI3 | Fm | Md | No | Lr | |||
 |
