Chemistry:Uranium(IV) iodide
From HandWiki
 | |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| UI4 | |
| Molar mass | 745.647 g/mol |
| Appearance | black hygroscopic crystals |
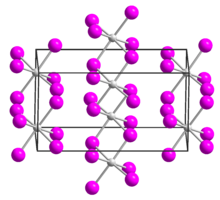
| Structure[1] | |
| monoclinic | |
| C2/c, No. 15 | |
a = 1396.7 pm, b = 847.2 pm, c = 751 pm α = 90°, β = 90.54°, γ = 90°
| |
| Hazards | |
| Main hazards | Radioactive |
| GHS pictograms |   
|
| GHS Signal word | Danger |
| H300, H330, H373, H411 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Uranium(IV) iodide, also known as uranium tetraiodide, is an inorganic chemical compound. It is a salt of uranium in oxidation state +4 and iodine.
Preparation
Uranium tetraiodide can be prepared from the reaction between uranium and an excess of iodine.[2]
Properties
Uranium tetraiodide is a black solid and forms needle-like crystals. Upon heating, it dissociates into uranium triiodide and iodine gas.[2] It crystallizes in the monoclinic crystal system, space group C2/c.[1]
References
- ↑ 1.0 1.1 Levy, J. H.; Taylor, J. C.; Waugh, A. B. (1980). "Crystal structure of uranium(IV) tetraiodide by x-ray and neutron diffraction" (in en). Inorganic Chemistry 19 (3): 672–674. doi:10.1021/ic50205a019. ISSN 0020-1669.
- ↑ 2.0 2.1 Brauer, Georg (1978). Handbuch der Präparativen Anorganischen Chemie. II (3rd ed.). Stuttgart: Ferdinand Enke. p. 1218. ISBN 3-432-87813-3.
| HI | He | ||||||||||||||||
| LiI | BeI2 | BI3 | CI4 | NI3 | I2O4, I2O5, I4O9 |
IF, IF3, IF5, IF7 |
Ne | ||||||||||
| NaI | MgI2 | AlI3 | SiI4 | PI3, P2I4 |
S | ICl, ICl3 |
Ar | ||||||||||
| KI | CaI2 | Sc | TiI4 | VI3 | CrI3 | MnI2 | FeI2 | CoI2 | NiI2 | CuI | ZnI2 | Ga2I6 | GeI2, GeI4 |
AsI3 | Se | IBr | Kr |
| RbI | SrI2 | YI3 | ZrI4 | NbI5 | Mo | Tc | Ru | Rh | Pd | AgI | CdI2 | InI3 | SnI4, SnI2 |
SbI3 | TeI4 | I | Xe |
| CsI | BaI2 | HfI4 | TaI5 | W | Re | Os | Ir | Pt | AuI | Hg2I2, HgI2 |
TlI | PbI2 | BiI3 | Po | AtI | Rn | |
| Fr | RaI2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | Ce | Pr | Nd | Pm | SmI2 | Eu | Gd | TbI3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | ThI4 | Pa | UI3, UI4 |
Np | Pu | Am | Cm | Bk | Cf | EsI3 | Fm | Md | No | Lr | |||
 |
